Chemistry - Unit 8 Test General Review
0.0(0)
Studied by 21 peopleCard Sorting
1/68
Earn XP
Description and Tags
Comprehensive review of various concepts/formulas/conversions that may be useful to have well remembered according to the test overview and unit slideshow (not fun!!!)
Last updated 4:44 AM on 3/16/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
69 Terms
1
New cards
Solids (Particle motion)
Particles vibrate
2
New cards
Liquids (Particle motion)
Particles slide past/against each other
3
New cards
Gases (Particle motion)
Particles move very quickly and freely of one another
4
New cards
Solids (IMFs)
Experiences the strongest IMFs
5
New cards
Gases (IMFs)
Experiences the weakest IMFs
6
New cards
Liquids (IMFs)
Experiences IMFs that are stronger than in gases, but weaker than in solids
7
New cards
Solids (Particle distance)
Particles are the closest together; they are packed very tightly together (rough explanation)
8
New cards
Liquids (Particle distance)
Particles are close to one another and have some distance from one another; more particle distance than solids but less than gases (rough explanation)
9
New cards
Gases (Particle distance)
Particles are the farthest apart and can be very far from each other (rough explanation)
10
New cards
Solids (V + S)
Definite/Fixed Volume and Shape
11
New cards
Liquids (V + S)
Fixed/Definite Volume and Indefinite Shape (assumes the shape of the container)
12
New cards
Gases (V + S)
Indefinite Volume and Indefinite Shape (assumes the shape of the container)
13
New cards
Solids (KE)
Lowest (average) kinetic energy when compared to all three states of matter
14
New cards
Gases (KE)
Highest (average) kinetic energy when compared to all three states of matter
15
New cards
Liquids (KE)
Higher (avg) kinetic energy than solids, lower (avg) kinetic energy than gases
16
New cards
Solids

17
New cards
Liquids
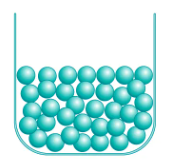
18
New cards
Gases
Image assumes an open container, must be adapted in a closed container
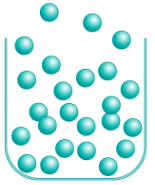
19
New cards
Gases
The **only** compressible state of matter (thereby meaning the **other two** states of matter are **not** compressible!)
20
New cards
Temperature
Average kinetic energy of all particles within a substance; measured with a thermometer
21
New cards
**Intra**molecular Forces
Occurs **within** a molecule; Ionic or Covalent; Chemical Reaction occurs when broken
22
New cards
**Inter**molecular Forces (IMFs)
Weak attractive force **between** molecules; London-Dispersion, Dipole-Dipole, Hydrogen Bond, Ion-Dipole; Physical Change occurs when broken
23
New cards
London-Dispersion Forces
(IMF) Exists in all substances; caused by an unequal distribution of electrons in a molecule or atom, creating a temporary instantaneous dipole at any given moment
24
New cards
Dipole-Dipole Forces
(IMF) **Only** present in **polar covalent** molecules; the unequal sharing of electrons create a permanent dipole which causes this IMF (positive ends are attracted to negative ends)
25
New cards
Hydrogen Bonding
(IMF) Special case of Dipole-Dipole Forces; only occurs when **H** is bonded to a **N**, **O,** or **F**
26
New cards
Ion-Dipole Forces
(IMF) **Only** occurs in mixtures with two different substances, one of which being an ion and the other being polar and covalent; caused by ions dissociating within a polar solvent so that cations are attracted to negative ends of the polar molecule and anions are attracted to positive ends of the polar molecule
27
New cards
Dispersion, Dipole-Dipole, H-Bond, Ion-Dipole
Order of IMFs from **weakest** to **strongest**
28
New cards
IMF Strength
Affected by the distance between particles (Inversely Proportional); Strength of charges (Electronegativity); Dispersion forces are affected by molar mass, easier to polarize when larger; Indirectly affected by temperature due to the Kinetic Molecular Theory
29
New cards
IMFs
Viscosity, cohesion, surface tension, and adhesion are caused by ____.
30
New cards
Kinetic Energy
1/2mv^2 (m = mass, v = velocity) → This pertains to the Kinetic Molecular Theory.
31
New cards
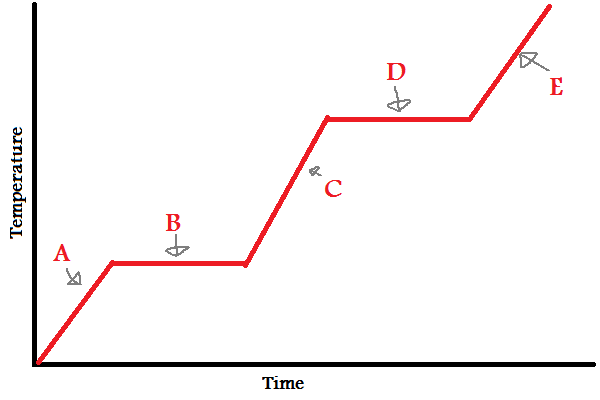
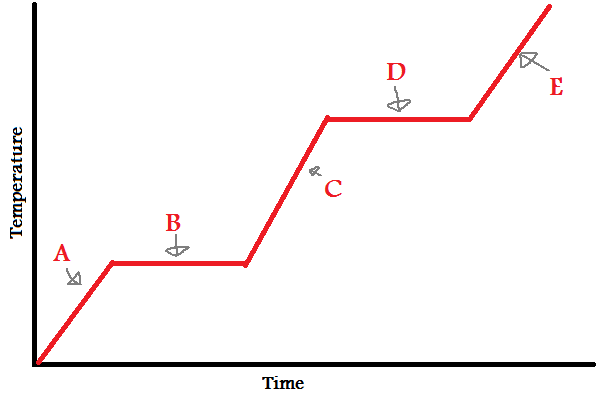
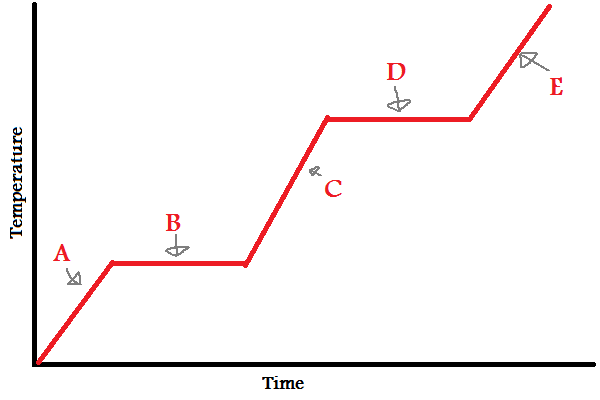
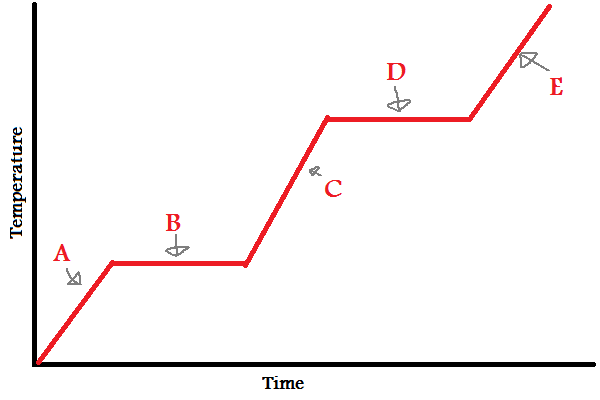
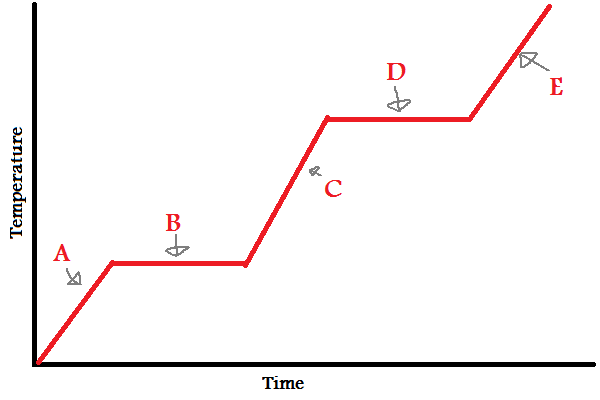
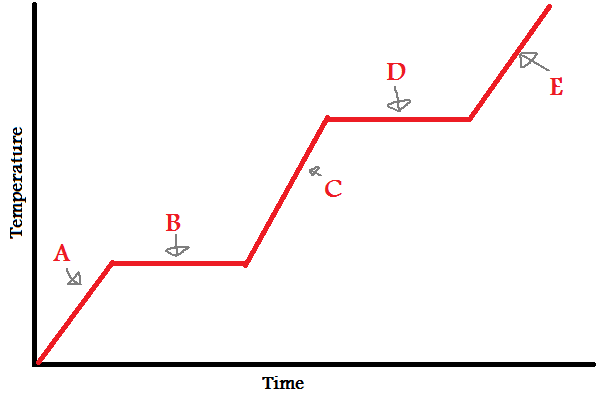
Solid
What does (A) indicate?
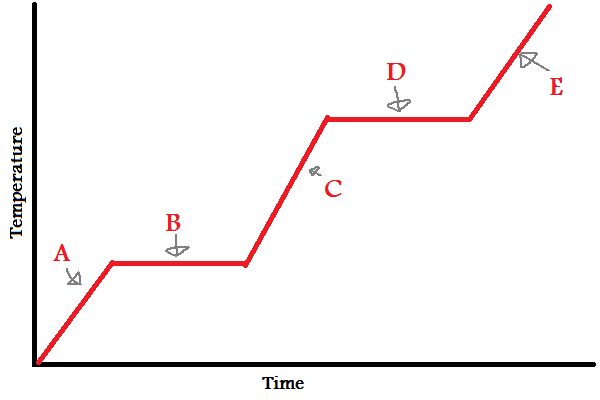
32
New cards
Melting/Freezing
What does (B) indicate?
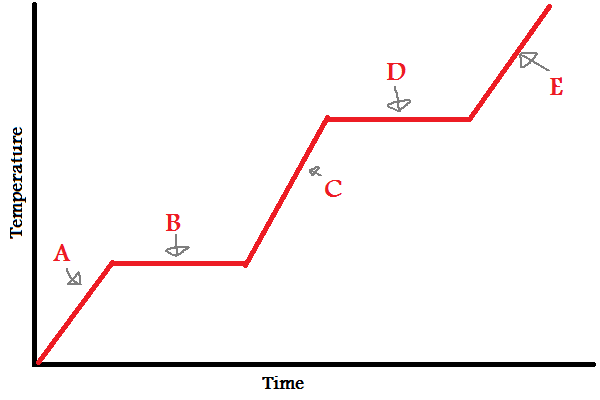
33
New cards
Liquid
What does (C) indicate?
34
New cards
Vaporizing/Condensing
What does (D) indicate?
35
New cards
Gas
What does (E) indicate?
36
New cards
A, B, C, D, E
In which letters is the substance increasing in **energy** (both kinetic and potential)?
37
New cards
A, C, E
In which letters is the substance increasing in **kinetic energy**?
38
New cards
IMFs
During (B) and (D), since the line is flat, what is the energy used to overcome?
39
New cards
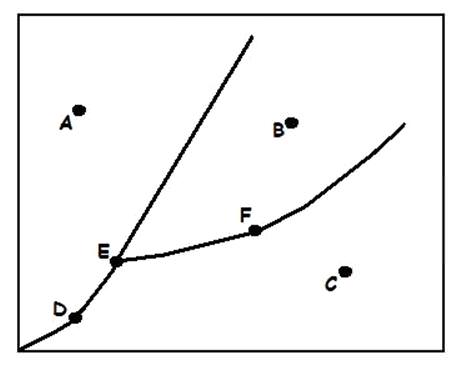
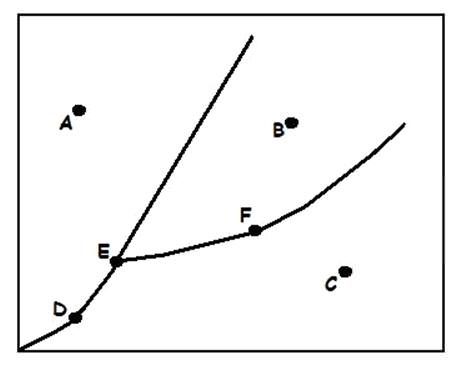
Liquid
What does (A) indicate?
40
New cards
Solid
What does (B) indicate?
41
New cards
Gas
What does (C) indicate?
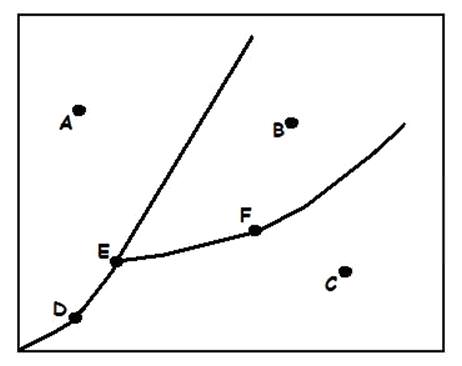
42
New cards
Sublimation/Deposition Line
What does (C) indicate?
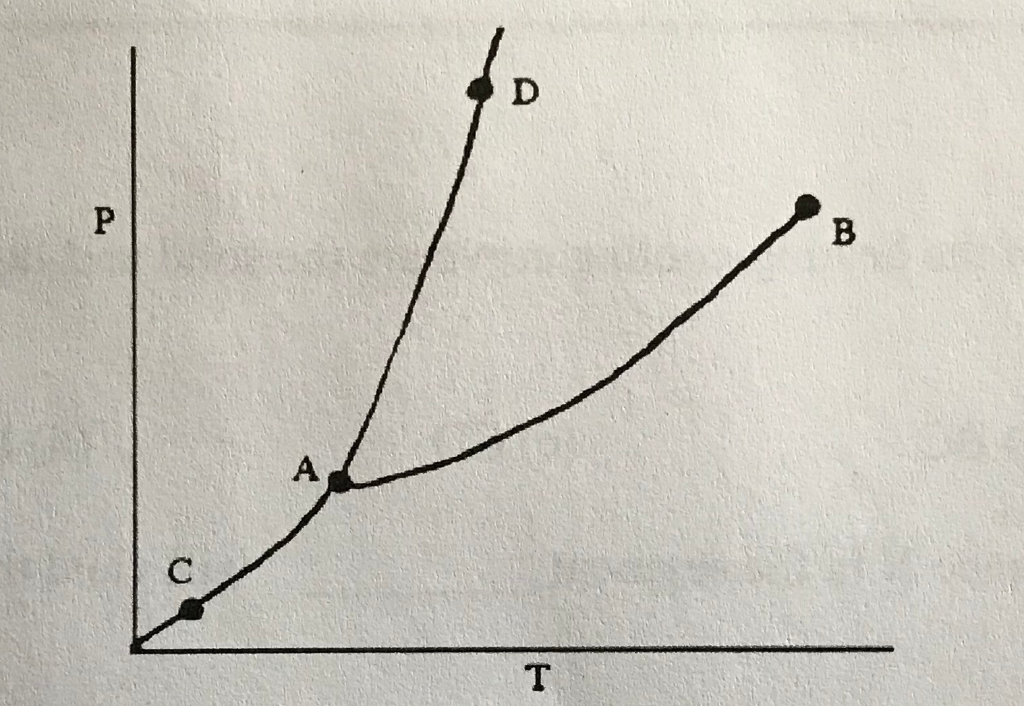
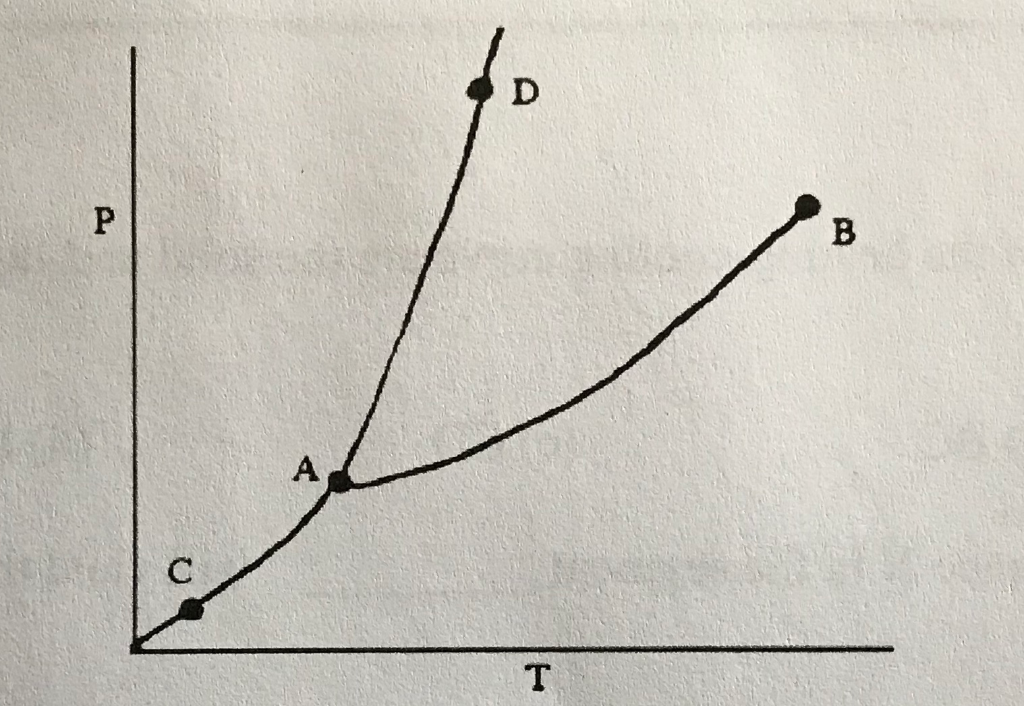
43
New cards
Triple Point
What does (A) indicate?
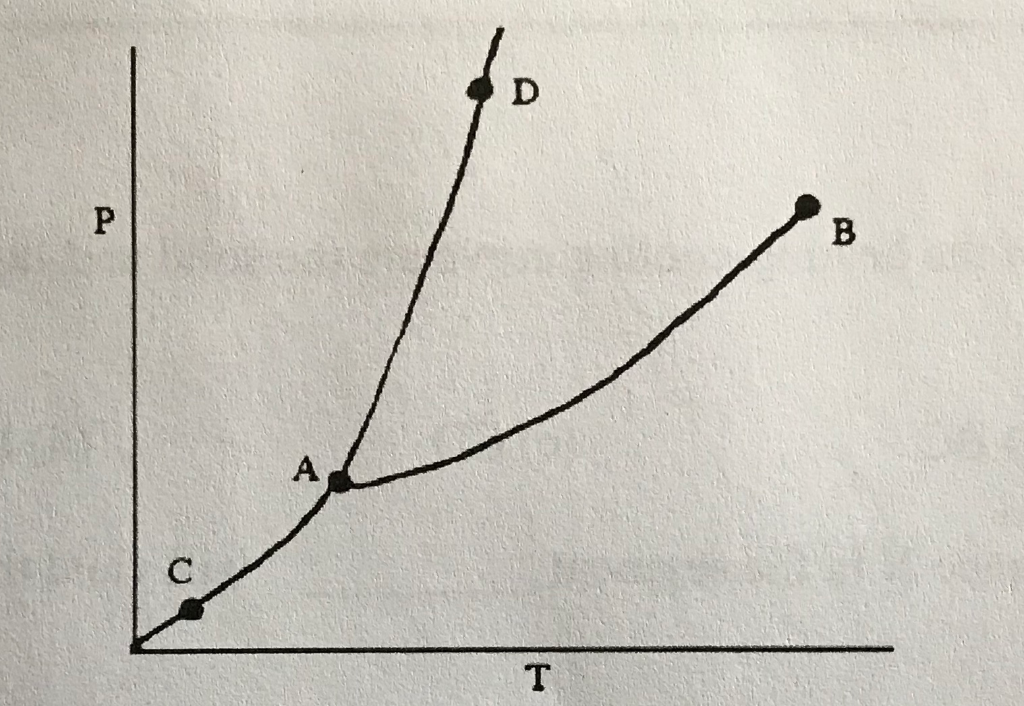
44
New cards
Melting/Freezing Line
What does (D) indicate?
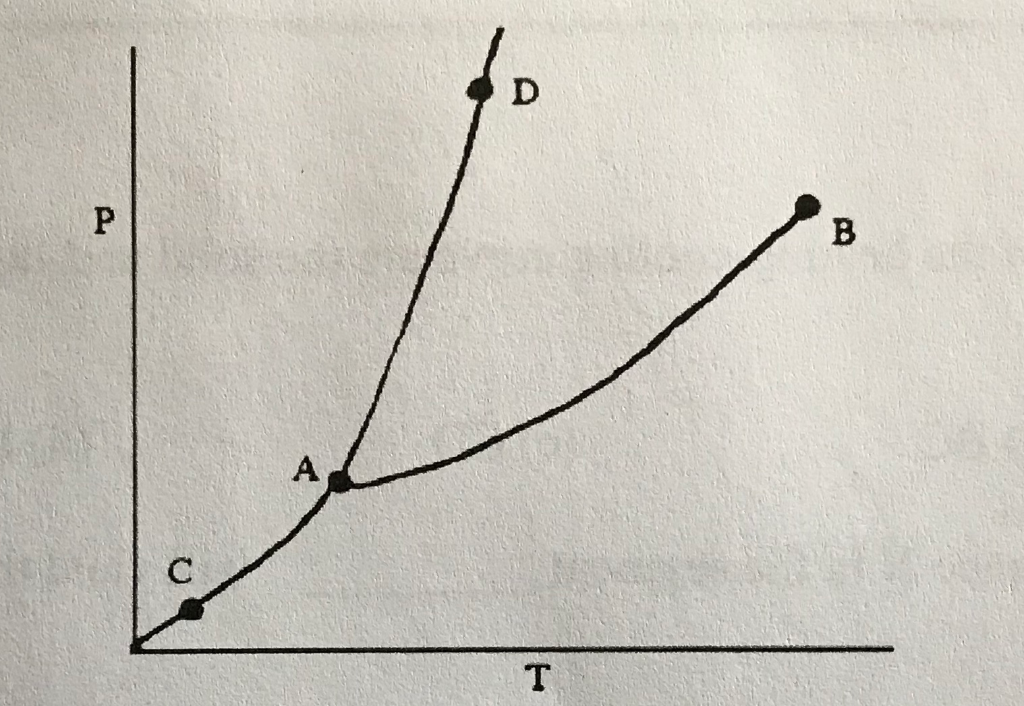
45
New cards
Vaporization/Condensation Line
What does (B) indicate?
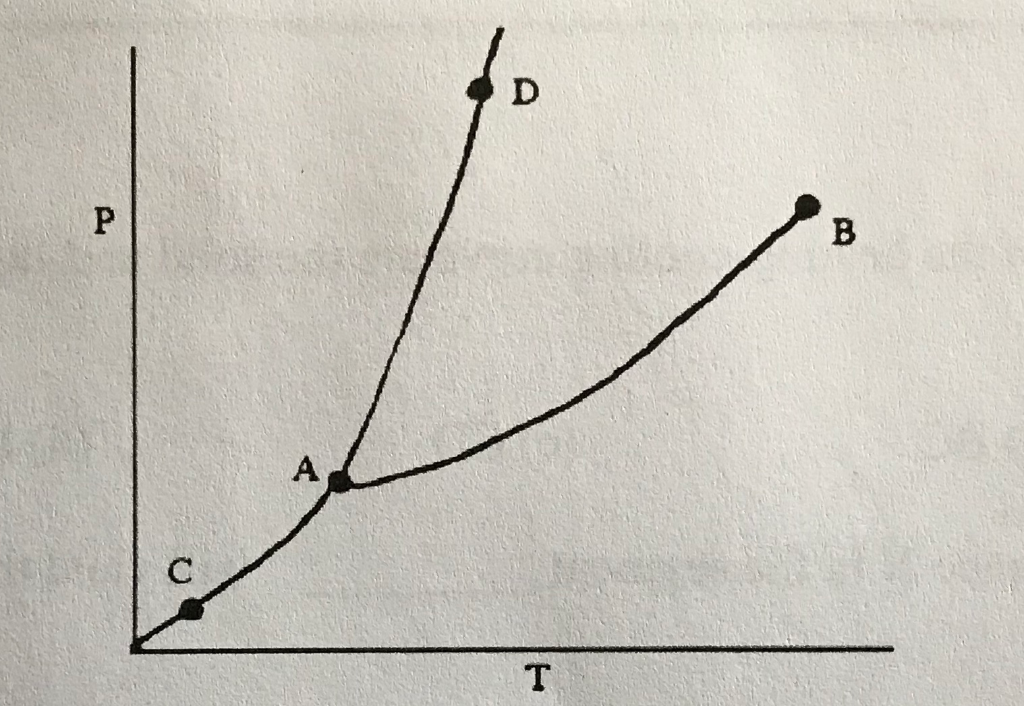
46
New cards
Critical Point
If the point is at the end of the Vaporization/Condensation line as it is with (B), what does the point also indicate?
47
New cards
Pressure/Temperature
In a phase change diagram, what are the two labels on the axes? (Y axis first, then X)
48
New cards
Endothermic
Requires energy input to make them happen (IMFs are being overcome)
49
New cards
Exothermic
Releases energy as they happen (IMFs are becoming significant)
50
New cards
Endothermic
Endothermic or Exothermic? (Melting, Vaporization, Sublimation)
51
New cards
Exothermic
Endothermic or Exothermic? (Freezing, Condensation, Deposition)
52
New cards
Boyle’s Law
(P1V1 = P2V2) At constant mass and temperature, the pressure and volume of a gas are **inversely proportional**.
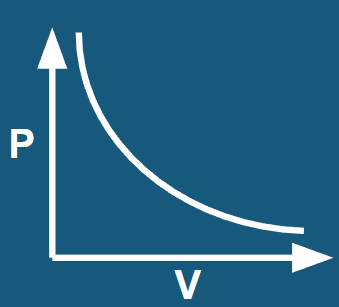
53
New cards
Charles’ Law
(V1/T1 = V2/T2) At constant mass and pressure, the volume and absolute temperature (K) of a gas are **directly proportional**.
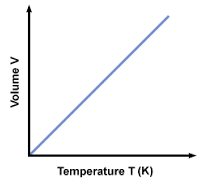
54
New cards
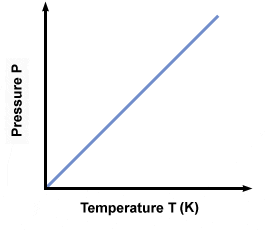
Gay-Lussac’s Law
(P1/T1 = P2/T2) At constant mass and volume, the pressure and absolute temperature (K) of a gas are **directly proportional**.
55
New cards
Combined Gas Law
(P1V1/T1 = P2V2/T2) → **Important formula to know**, it replaces knowing each of the three Gas Law formulas individually.
56
New cards
STP
Standard Temperature and Pressure → **1 atm** and **0 degrees C** (**273 K**)
57
New cards
Molar Volume
**At STP**, the volume occupied by **1 mol** **of gas** is **22.4 L**. → When doing Stoichiometry, 1 mol of gas **at STP** = 22.4 L
58
New cards
Ideal Gas Law
(PV = nRT) → **Important formula to know**, while there are other important uses, it also has to be used during Stoichiometry when you are **not** at **STP**.
59
New cards
R (atm)
(Universal Gas Constant) → R = 0.0821 L ⋅ atm/mol ⋅ K
60
New cards
R (kPa)
(Universal Gas Constant) → R = 8.315 L ⋅ kPa/mol ⋅ K
61
New cards
Ideal Gas
A gas which is behaving according to the Kinetic Molecular Theory → Allows for the Ideal Gas Law to be used. → Gas behavior is most ideal at low pressures, high temperatures, and in nonpolar atoms/molecules.
62
New cards
Real Gas
A gas which does not follow the Kinetic Molecular Theory, typically occurs when a gas gets close to condensing or turning into a solid. This happens at low temperatures or high pressure. → Unlike ideal gases, particles in a **real gas** have significant volume, have IMFs that attract each other, and have collisions that are inelastic (loss of KE).
63
New cards
Absolute Zero
The lowest possible temperature at which all molecular motion stops (KE = 0). → 0 K
64
New cards
Celsius to Kelvin
T (Celsius) + 273 = T (Kelvin) → Exact conversion factor, so use the amount of decimals of the given Celsius temperature for sig figs
65
New cards
atm to Pa
1 atm = 101,325 Pa (can be rounded to 101,300)
66
New cards
atm to kPa
1 atm = 101.325 kPa (can be rounded to 101.3)
67
New cards
atm to mmHg
1 atm = 760 mmHg
68
New cards
atm to Torr
1 atm = 760 Torr
69
New cards
atm to psi
1 atm = 14.7 psi