Experiment to measure the specific latent heat of vapourisation
1/8
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No study sessions yet.
9 Terms
Draw diagram
…
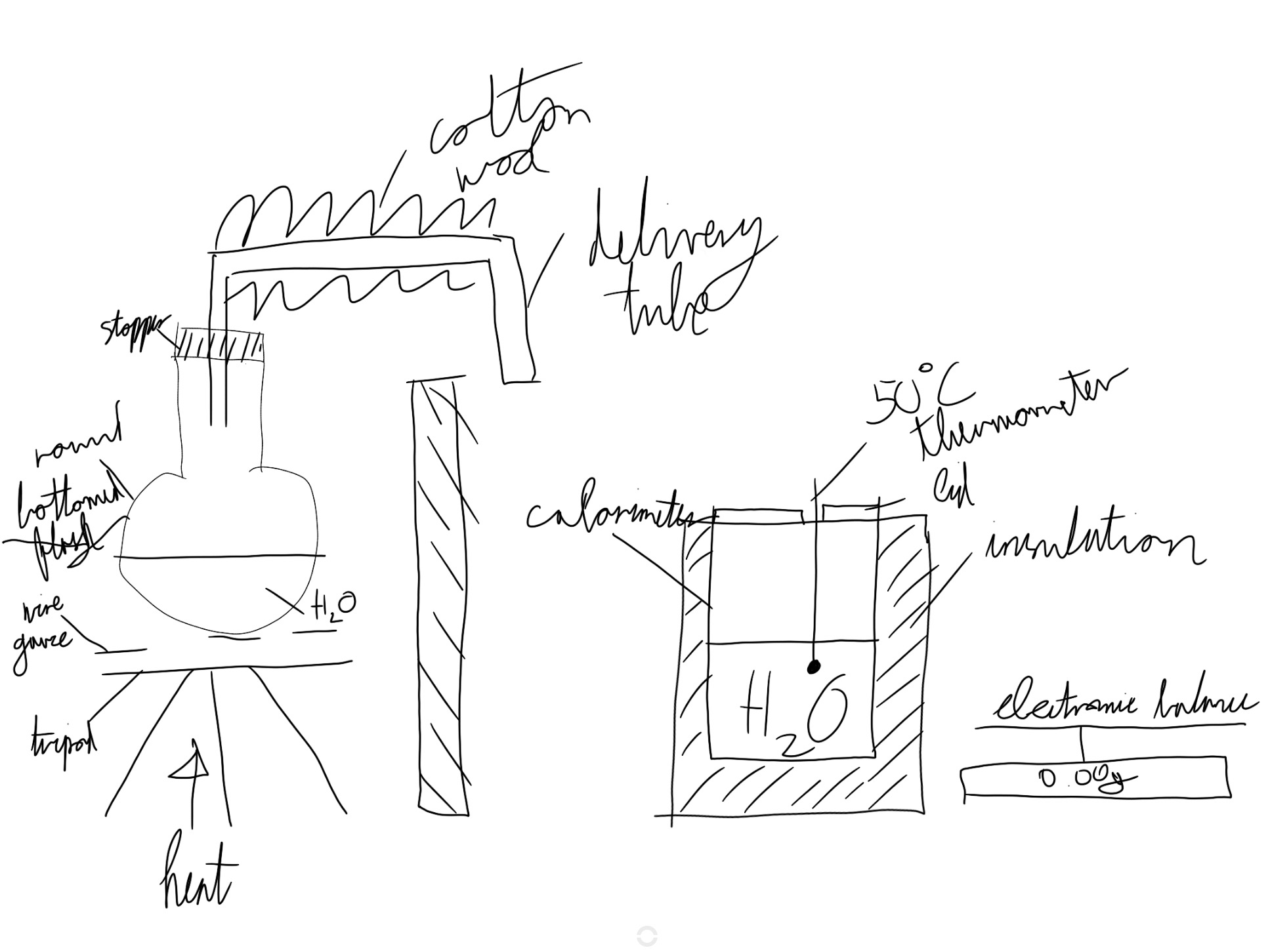
Step 1
Find the mass of a clean dry empty calorimeter (Mc)
Step 2
Half fill with wasted and find the new mass (Mc + w)
Step 3
By subtraction find the mass of the water (Mw)
Step 4
Measure the initial temperature of the water in the can (θi)
Step 5
When the steam has been issuing freely for ten minutes dry the delivery tube and insert it into the water in the can
Step 6
Measure the final temperature (theta f) and the ways of the calorimeter + the water + the steam (Mc +w+ s)
Step 7
By subtraction find the mass of the steam.
Step 8
Calculate the specific latent heat of vapourisation of water water from (Ms)(lvapourisation) + Ms(Cw)(delta theta s) = (Mw)(Cw) (delta theta w) + Mc(Cc) (delta theta c)