Chemistry Instrumentation SOLO 3
1/99
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
100 Terms
electrophoresis
migration of charged solutes or particles in a liquid medium under the influence of an electric field
separation technique used primarily for separating and identifying serum proteins in blood
Iontophoresis
migration of small ions
Zone electrophoresis
used to separate macromolecules, such as proteins, in a porous support medium
charged molecules migrate as zones
migration takes place across a support medium
sample is mixed with buffer solution
separated protein zones are generated on the support media known as an electropherogram
media stained with protein specific stain
zones are quantified using a densitometer
ampholyte (zwitterion)
molecule that can become either negatively or positively charged based on the pH of the solution in which it resides
isoelectric point
the pH at which a molecule has a net charge of zero
The ability to separate proteins by electrophoresis is based on what?
the characteristics of amino acids
Direction of migration in electrophoresis
an ampholyte will take a positive charge in a solution more acidic than its isoelectric point and migrates towards the cathode
an ampholyte will take a zero net charge in a solution at which the pH equals the isoelectric point
an ampholyte will take a negative charge in a solution more alkaline than its isoelectric point and migrates toward the anode
What are the five bands proteins are separated into during serum protein electrophoresis?
Albumin (fasstest)
Alpha 1 globulins
Alpha 2 globulins
Beta globulins
Gamma globulins (slowest)
migration inhibition factor phenomenon
at a pH of 8.6, the gamma globulins move toward the cathode, despite the fact they are negatively charged
The rate of migration in serum protein electrophoresis depends on:
net electrical charge of the molecule (larger=faster)
size and shape of the molecule
electrical field strength
properties of the supporting medium
temperature of the operation
Basic components of an electrophoresis apparatus
power supply
two buffer chambers
electrodes in buffer
electrophoretic support media
wick in buffer
chamber lid
Power Supply of an Electrophoretic apparatus
best to use one with a constant current as opposed to xonstant voltage to keep the migration rate relatively constant
constant voltage power supplies cause a rise in current due to production of heat during electrophoresis
rise in heat reduces resistance and increases the electromotive force due to:
Joule heat: flow of current through a medium that offeres resistance
heat causes thermal agitation of ions
water loss due to evaporation causes an increase in ion concentration
temperature of gel must be controlled because heat also has the potential to dentaure proteins
Wick flow of an Electrophoretic apparatus
caused by movement of buffer into the support medium
moisture evaporation from the gel from heat generation during electrophoresis causes movement of the buffer into the gel
gel absorption of buffer to replce the lost moisture affects the migration of sample molecules
using a lid or cover during electrophoresis can prevent some of this evaporation
methods that gernerate excessive heat utilize a cooling system during electrophoresis to prevent wick flow and other damage to sample solutes
Buffer of an Electrophoretic apparatus
carries the applied current
establishes the pH at which electrophoresis is performed
determines the electrical charge on the solute
ionic strength
the sum of all charges present, negative or positive
Increasing ionic strength of the buffer results in:
increases conductance of the support
increases size of the ionic cloud surrounding a charged molecule
yields slower migration rates
yields sharper band separations
Support media of an Electrophoretic apparatus
cellulose acetae (one of the first, used in isoelectric focusing)
agarose gel (serum protein electrophoresis)
polyacrylamide gel (used in PAGE, separates proteins using molecular sieving (m/z and size))
automated systems
What is the importance of the ionic strength of the buffer used in gel electrophoresis?
a heat labile protien will become denatured if the ionic strength of the solution is too high
In electrophoresis, the best support medium to use because it is thermostable, transparent, and eliminates endosmosis due to the lack of charge is?
polyacrylamide gel
General operations in a conventional electrophoresis include:
separation
staining detection
quantification
Stianing detection in electrophoresis
serum proteins: amido black coomassie brilliant blue, ponceau S
isoenzymes: formazan, nitrotetrazolium blue
lipoprotein zones: fat red 7b, oil red o, sudan black b
DNA fragments: ethidium brommide (fluorescent)
CSF proteins: silver nitrate
A reliable and accurate quantification of electrophoresis requires:
light of appropriate wavelength, may include visible and U.V. light
linear response of the instrument
transparent background in the strip being scanned
Usefule features of a densitometer include:
ability to scan gels of appropriate length
automatic gain, which chooses the peak of greatest intensity and sets it to full scale
automatic background zeroing to select lowest point in the scan as a baseline
variable wavelength control over the range of 400-700nm
variable slits to allow adjustment of the beam size
appropriate selectrion of cut points between peaks
automatic indexing to move the scanner from one strip or sample channel to the next
ability to measure ultraviolet fluoescence
Endosmosis
preferential movement of water in one direction through an electrophoresis medium
occurs due to selective binding of one type of charge on the surface of the medium
minimal in AGE and PAGE because the surface charge of these gels is low
much more significant in capillary electrophoresis
electrolysis
chemical decomposition produced by passing an electric current through a liquid or solution containing ions
Buffer technical considerations of electrophoresis
highly susceptible to bacterial growth
should be discarded after each use due to electrolysis
Common problems in electrophoresis
discontinuties (dirty applicators)
unequal migration of samples across the gel (dirty electrodes)
distorted protein zones (bent applicators, air bubbles, over application, excessive drying)
irregularities in sample application (excessively wet media)
Unusual bands in electrophoresis can be caused by?
hemolyzed specimens (increase beta globulins)
plasma (fibrinogen band)
albumin (split zone caused by bis-albuminemia or more than one type of albumin)
Zone electrophoresis
produces zones of prroteins
migration depends on the ratio of charge to zise
agarose, cellulose acetate, and polyacrylamide gel used
Isoelectric focusing
separates amphoteric compounds sucha as proteins across a gell possessing a pH gradient
based on separation of different molecules by their charge
proteins migrate across a gel until they reach a zone that mathces the isoelectric point of the protien
most commonly used for abnormal hemoglobin screens
immunofixation electrophoresis
detects specific ypes of immunoglobulins or kappa and lamba chains in serum or urine of patients with monoclonal gammopathies such as multiple myeloma or Waldenstrom’s macroglobulinemia
capillary electrophoresis
uses the basic principles of electrophoresis carried out in a small-bore capillary tube which serves at the electrophoresis chamber
tube is connected to a detector at its terminal end and to a high-voltage power supply via buffer reservoirs
Capillary electrophoresis buffers must:
not interfere with the ability to detect the analyte of interest
maintain the solubility of the analyte
maintain the buffering capacity through the analysis
produce the desired separation
Advantages of capillary electrophoresis
produces efficient heat dissipation
enhances separation efficiency
reduces separation time in some cases to less than 1 minutes
two-dimensional electrophoresis
1st dimension: charge dependent isoelectric focusing
2nd dimension:molecular weight dependent electrophoresis
used to study families of proteins, look for genetic or diseased based differences in proteins, and study the protein content of cells of various types
proteomics
the identification and quantification of proteins and their posttranslational modifications in a given system or systems
osmosis
process that constitutes movement of solvent across a membrane in response to differences in osmotic pressure across the two sides of the membrane
osmotic pressure
pressure required to stop osmosis through a semipermeable membrane between a solution and pure solvent
governs the movement of solvent across membranes that separate two solutions
osmole
1 gram molecular weight of a substance divided by the number of particles into which it dissociates
osmolality
expresses concentrations related to mass of solvent
osmolarity
expresses concentrations per volume of solution
major osmotic substances in normal plsama
sodium
chloride
glucose
urea (BUN)
Osmolality formula
mOsm/kg= 2[Na(mmol/L)]+glucose[mg/dl]/18 + urea[mg/dl]/2.8
osmometry
a technique that measures the concentration of dissolved solute particles in a solution that contribute to the osmotic pressure
dependent on the number of particles in solution rather than size or charge
expressed in units of mOsm/kg or /L
includes osmolarity and osmolality
A solute dissolved in solvent causes the following changes in colligative properties:
increased osmotic pressure
lowered vapor pressure
increases boiling point
lowered freezing point
Freezing point osmometers
measurement of the freezing point of a solution by supercooling the specimen to -7C
vapor pressure osmometers
measurement at the specimen’s dew point
correlation shows more variation at freezing point than dew point so freezing point osmometers more commonly used
factors that affect osmo readings
lipemic specimens (dont freeze well)
prescence of lactic acid (increase freezing point)
volatile substances such as ethanol (decrease freezing point_
Chromatography
a group of separation techniques that sepaate analytes by differential distribution between a stationay phase and a mobile phase
stationary phase in chromatography
a solid or liquid phase that interacts with components of the mobile pahse
mobile phase in chromatography
a gas ro liquid that flows in a chromatographic system and carries the sample past the stationary phase
As mobile phase flows past the stationary phase, the solutes may:
reside only on the stationary phase (no separation)
reside only in the mobile phase (migration with the mobile phase)
distribute between the two phases (differential migration)
Basis of separation
those solutes with high affinity for the stationary phase reside in the stationary phase and migrate slower
those solutes with low affinity for the stationary phase reside mostly in the mobile phase and migrate faster
strongly bound solutes subsequently are displaced from the stationary pahse by changing the physical or chemical nature of the mobie phase
planar chromatography
separation technique in which the stationary phase is on a thin support such as paper or solid surface
used in identification of drugs in urine, analysis of amino acid and analysis of lipids in amniotic fluid
migration of a solute is expressed by its retention facotr value, the ratio of solute migration to solvent front migration
includes paper and thin layer chromatography
paper chromatography
the stationary phase consists of a layer of water or a polar solvent coated onto the fibers of a sheet of paper
separation takes place between the nonpolar mobile phase and the polar stationary phase
thin layer chromatography
the stationary phase consists of a thin layer of particles of a material such as silica gel that is uniformly spread on a glass plate, plastic sheet, or aluminum sheet
when the thin layer consists of particles of a small diameter, the technique is known as high-performance thin layer chromatogrpahy (HPTLC)
Retention factor formula
distance from application point to solute center/ distance from application point to mobile pahse front
What phase of chromatography carries the sample?
mobile phase
Column chromatography
separation technique in which the stationary phase is packed into a tube or is coated on the inner surface of the tube
includes gas and liquid chromatography
gas chromatography
gas mobile pahse or carrier gas is used to carry a mixture of volatile solutes through a column containing the stationary phase
carrier gas is normally nitrogen, argon or helium
to be analyzed, a compound must be volatile or be made volatile
liquid chromatography
separation is based on distribution of solutes between a liquid mobile phase and a stationary phase
ligh performance liquid chromatography (HPLC) uses small diameter particles
Detector in column chromatography
a device that responds to the prescence of analyte in the mobile phase, the magnitude of which is used to identify and quantify analytes
flame ionization detector most used in GC
produces an electronic signal that is plotted as a funcion of time, distance or volume
results in a chromatogram
chromatogram
a plot of detector resopnse used in column chromatography to the prescence of analyte in the mobile phase as a function of time or mobie phase volume
retention time
time interval between specimen injection and solute reaching the detector
column chromatography Resolution (Rs)
measure of chromatographic separation
requires that two peaks have different elution times for the peak centers and sifficiently narrow bandwidths to eliminate or minimize overlap
incomplete separation occurs when the calculated value is <0.8
baseline separation is obtained when the value is >1.25
Improved resolution is achieved by enhancing column efficiency by:
using smaller particles
changing flow rate
using a longer column
minimizing dead volume (volume of tubing, connectors, etc)
Enhanced resolution is achieved by changing the selectivity of separation by altering factors such as:
the composition of the mobile phase
stationary phase
temperature
which component of a gas chromatograph introduces an aliquot of sample to be analyzed into the column?
injector
Chromatographic separations are classified by the chemical or physical mechanism used to separate the solute and include:
ion exchange
partition
adsorption
size exclusion
affinity mechanisms
ion exhange chromatography
based on an exchange of ions between a charged stationary surface and ions of the opposite charge in mobile phase
cation exchange particles and anion exchane particles
displacement of ions bound to the stationary phase depends on competition by other ions in the mobile phase
retention is decreased either by higher ionic strength, adjustment of pH to decrease the charge of analytes, or adjustment of pH to decrease the charge of the stationary phase
cation-exchange particles
contain negatively charged groups in the stationary phase that bind positively charged components in the mobile phase
anion-exchange particles
containe positively charged groups in the stationary phase that bind negatively charged components in the mobile phase
partition chromatography
differential distribution of solutes between two immiscible liquids is the basis for separation by partitiona chromatography
one of the immiscible liquids serves as the stationary phase
as the mobile phase carries the solute through the column, separation occurs
classified as gas-liquid chromatography or liquid-liquid chromatography
normal phase liquid-liquid chromatography
stationary phase is polar and the mobile phase is relatively nonpolar
reversed phase liquid-liquid chromatography
the stationary phase is nonpolar and the mobile phase is relatively polar
adsorption chromatography
based on differential adsorption of solutes on the surface of the stationary phase
hydrogen bonding and hydrophobic interactions mediate separation
retention depends on the surface area of the stationary phase and the affinity of the solutes for the stationary phase
In GC, used to separate low molecular weight compounds and compounds that are normally gases at room temperature
size exclusion chromatography
gel-filtration chromatography
separates solutes on the basis of their molecular size in solution
a variety of materials are used as stationary phase for size exclusion
beads have pores that allow small molecules to enter and to be retained to a greater extent than large molecules that are excluded from pores
Affinity chromatography
one component of a highly-specific molecular interaction pairs is immobilized in a stationary phase and is used to capture molecules form the mobile phase
interaction pairs are strong, highly specific, and may include enzyme inhibitor, hormone receptor, antigen-antibody, or aptamer-ligand
What is the method used for unknown identification of analytes in column chromatography?
comparison of retention time
mass spectrometry
works by ionizing a target molecule into ions of a specific mass-charge ratio
separates and measures those ions
produces a mass spectrum
mass spectrum
the relative abundance of each ion is plotted as a function of its mass-charge ratio
mass chromatogram
abundance pltted at a function of time
mass analysis
the process by which ions are identified according to m/z ratios
resolution (MS)
the mass of a given compound divided by the width of the corresponding peak
“full width at half max”
mass accuracy
how accurate the instrument is by comparing measured mass assignment for an ion to the expected mass
sensitivity
what concentrations of an analyte can be detected
ionization sources
electron ionization
electrospray ionization (ESI)
Atmospheric Pressure Chemical Ionization (APCI)
Matrix Assisted Laser Desorption Ionization (MALDI)
Electrospray ionization
applicable to a wide range of analytes
minimal fragmentation of molecular ion
high molecular weights can be analyzed
high sensitivity
ionization suppression (competition for the ions to leave the droplet)
mobile phase composition can affect sensitivity
Atmospheric Pressure Chemical Ionization (APCI)
low polarity molecules can be ionized
wide dynamic range
mass sensitive detection dependency
higher buffer concentration allowed
analytes must be thermally stable
analytes must be volatile
MALDI
applicable to a wide range of analytes, especially for large biomolecules
sample in solid state
more sophisticated workflows
fast analysis
high background for low m/z ratio region from matrix effects
limited matrix selection for different compound types
Mass analyzer types
quadrupole
ion trap
tandem mass spectrometry (MS/MS)
high resolution MS
time of flight
orbitrap
Quadrupole
four parallel rods in which the opposite pair of rods are connected to the same DC and RF voltage
rapidly changing polarity and magnitude of voltage applied to filter by m/z
full scan or selected ion monitoring
Ion trap
trap and store ions over time for greater sensitivity
electric field is adjusted to selectively eject ions based on m/z
Scanning modes
full scan- detects all ions
selected ion monitoring- detects ions of one selected m/z
product ion scans- select ions of one m/z and fragments ions, all ion fragments pass through
selected reaction monitoring- select ions of one m/z, fragments ions, fragments of only one m/z allowed to pass through
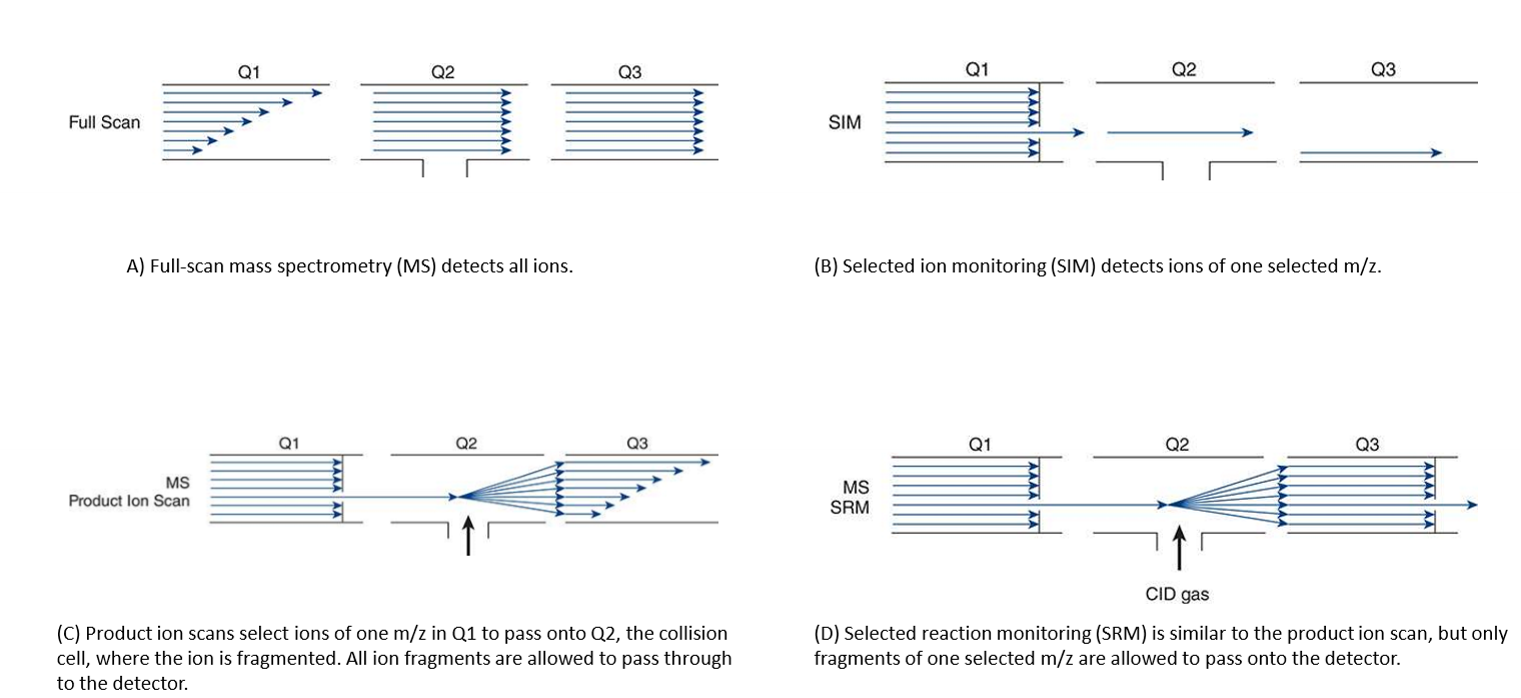
Tandem mass spectrometry
important technique in clinical and analytical labs
quantitative analysis, compound identification, structural characterization
high resolution MS
can measure large number of analytse simultaneously in complex matrices
calculaate mass to 0.001-0.0001 Da
time of flight
flight time- time it takes to traverse the flight tube
commonly used with continuous flow or pulsed ion sources
high mass accuracy and resolution
MS detectors
series of dynodes with increasing potential
signal amplifies to the order of 1 mil or greater
includes discrete, continuous and microchannel
faraday cup
ion abundance is so high that it would saturate the output of an electron multiplier
clinical application of MS
small molecule analysis (toxocology, Vit D, Testosterone, immunosuppressants)
proteomics (shotgun approach)
pathogen identification
point of care