Entropy and the 2nd Law
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
What does spontaneous change refer to
Refers to change that doesn’t require an external source of energy.
In a spontaneous process what happens to matter and energy
Becomes more disordered
What is the symbol for entropy and what does it measure
Entropy , S, measures the disorder of matter and energy.
State an equation for entropy relating it to micro states
S = k ln W
K is the Boltzmann constant
W is number of microstates
This equation is true for a single molecule, to do it for one mole you must replace k with the constant R as ( k*NA = R)
State the second Law of thermodynamics
The second law of thermodynamics refers to how the entropy of the universe tends to increase.
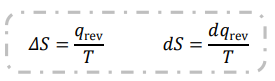
Give equation linking heat exchanged reversibly with delta entropy
d notation for a tiny change
qrev refers to heat exchanged reversibly
T for temperature
What does entropy being a state function and molar entropy being an intensive property mean
S is a state function (independent of route taken)
Sm is an intensive property (independent of quantity of the substance).
State equation for entropy of fusion and its definition
Entropy of fusion = increase of entropy when melting a solid substance
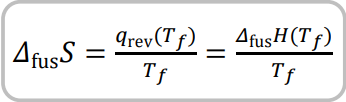
State equation for entropy of vaporisation and its definition
Entropy of vaporization = increase of entropy upon vaporization of a liquid
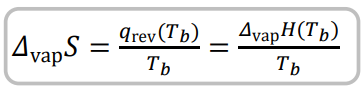
State Troutan’s Rule, name three exceptions and why
Δ vap H / Tb is approximately the same for all liquids, about +85 J K-1 mol-1 .” (But there are exceptions.)
Hg, due to diatomic formation
NH3, due to H bonds
H2O, due to H bonds
All are due to beginning in a more stable formation so change in entropy is great as it goes from highly ordered to highly disordered.
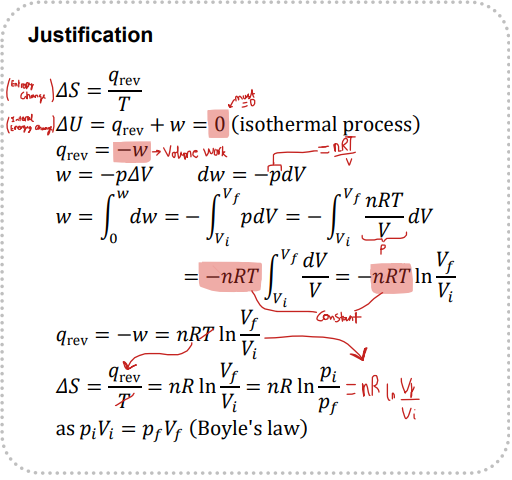
State the equation linking entropy with a change in volume
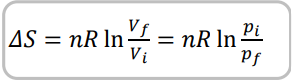
Give the proof for entropy changes due to volume changing
State the equation for change in entropy when there is a change in temperature
If temperature is changing we need to use an integral
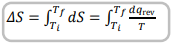
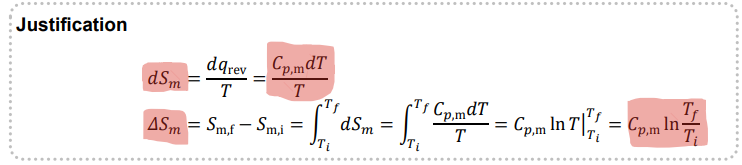
State the equation for change in entropy when there is a change in temperature, for a perfect gas

Give the justification for entropy change when temperature is changing with a perfect gas
State the equation relating Specific heat capacity at constant pressure with Specific heat capacity at constant volume, giving its justification
C.p.m = C.v.m + R
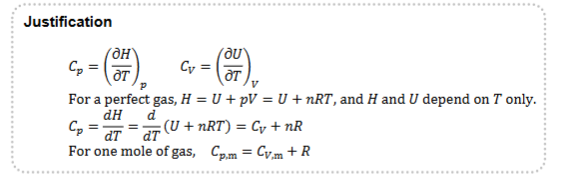
State values of R for Cp and Cv when monatomic

State values of R when Cv and Cp are polyatomic
