Soft Matter
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
Give the equation for stress for a Hookean solid. What does this say about the behaviour?
σ = Gγ or stress = elastic modulus X strain (G and σ → units = Pa). Applied stress → instantaneous strain, strain is directly proportional to stress. Perfect spring and does not permanently deform.
Give the equation for the stress of a Newtonian liquid. What does this say about its behaviour?
Stress (Pa) = viscosity (Pa s) x shear rate (s-1). Shows a delayed, irreversible response. Higher viscosity = bigger the delay.

What are the 3 types of Non-Newtonian fluids?
Shear-thinning fluids: viscosity decreases with increasing strain rate.
Shear thickening fluids: viscosity increases with increasing strain rate.
Bingham plastic: viscosity appears infinite until a certain stress is applied
Give the equation for stress for a viscoelastic material
Both elastic and viscous response.
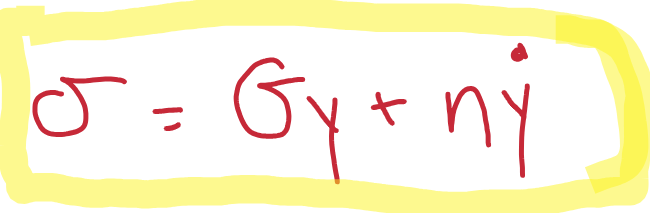
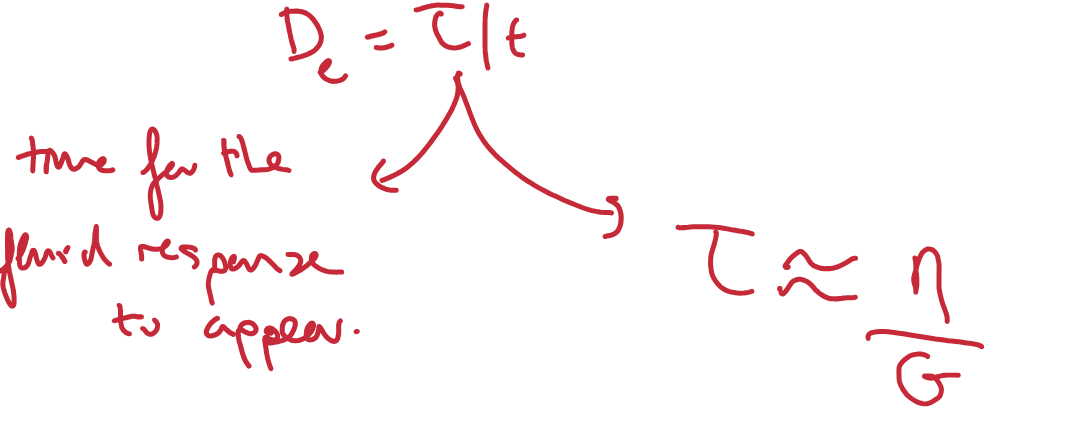
What is the Deborah number (give equation)?
= The characteristic timescale of a material/relaxation time of a system.
What does the Deborah number show?
D(e) » 1, elastic behaviour dominates. Relaxes much slower than the experiment time.
D(e) « 1, viscous behaviour dominates. Relaxation time is short compared to experiment time.
Outline the stress relaxation test and creep test.
Stress relaxation test: strain is held constant, monitors decay of stress as a function of time. Over time stress drops as material starts to flow.
Creep test: stress is held constant, monitors strain increase. Rapid increase at first and slows over time as material starts to flow.

Outline what each value means in this equation for dynamic experiments
w = frequency of deformation.
G’ = storage modulus: accounts for the energy that is stored in deformation i.e. elastic behaviour.
G” = loss modulus: accounts for energy lost in dissipation.
What is the loss tangent? What do the values show?
tan(δ) = loss tangent = G”/G’.
tan(0) = 0 →G”= 0, perfect solid behaviour.
tan(pi/2) = infinity →G’ = 0, perfect Newtonian behaviour.
tan(pi/4) = 1 → G” = G’, viscoelastic. → Used to work out relaxation time.
Outline the Maxwell Model and state the equation that corresponds to it.
Spring and dashpot are in series. Same stress through each element with linear addition of strain.
Good for stress relaxation modelling.
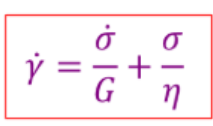
Outline the Voigt Model and state the equation that corresponds to it.
Spring and dashpot in parallel. Same strain through both elements, linear addition of stress.
Good for creep modelling.
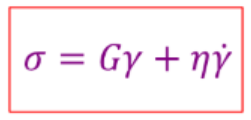
Describe a random-flight polymer and give equation for determining the average end-to-end distance.
Direction of each segment is random relative to the previous. No enthalpic contributions.
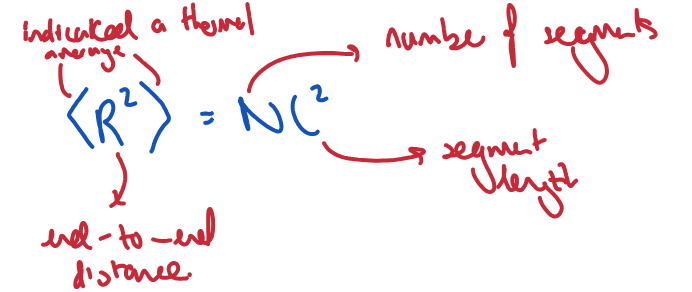
What is the most probable end to end distance?
R = 0, i.e. when the chain ends touch - polymers segments can adopt many more conformations, favouring entropy.
What is the radius of gyration? Give equation.
= measure of the average (r.m.s) distance of a chain segment from the centre of mass of the polymer.
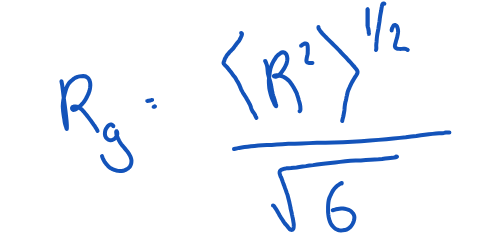
What are the two types of constraints that need to be taken into account for real polymers?
short-range correlations: geometric constraints between a monomer and its neighbours. Fixed bond angles t/f cannot take any angle.
long-range correlations: chain segments cannot occupy the same space.
Outline the characteristic ratio and give the equation for the real polymer end-to-end distance.
Tells you how much larger a real polymer coil is compared to the random flight polymer. Typically 5-10.
C∞l = distance it takes for the polymer to forget its original direction (correlation length).

What is the excluded volume effect?
Since segments cannot intersect → reduced number of accessible configurations → entropic loss → free energy penalty for real polymers.
What are the two competing effects in a self-avoiding polymer?
Excluded volume effect acts to expand the polymer chain to reduce the free energy.
Elasticity entropy acts to keep chain ends close together to reduce coil size.
→ Equilibrium between the two effects.
How do coil sizes scale in a self-avoiding polymer vs a random flight polymer?
Self avoiding: R is proportional to N^0.6
Random flight: R is proportional to N^0.5
Outline what the Flory Huggins parameter shows
= Chi. Characterises the strength of polymer-polymer interactions compared to the polymer-solvent interactions.
Chi < ½ for a goof solvent → expanded coil, R is proportional to N^0.6. Maximises polymer-solvent contacts.
Chi = ½ for a theta solvent, Random coil with segment length C∞l. R scales as N^0.5. Polymer is ideal dimensions.
Chi > ½ for a poor solvent, collapsed globule, R scales as N^0.33. Polymer contracts to reduce polymer-solvent constacts
What can we say about a polymer in dilute solutions, and the viscosity of the solution?
Polymer chains do not overlap - each molecules is considered its isolated. Viscosity increases linearly with polymer concentration (greater viscous drag).
Outline the critical concentration, C*
Where the coils start to overlap (semi-dilute solution). At this point viscosity increases rapidly with concentration of polymer as the chains become entangled and non-Newtonian behaviour seen (shear-thinning).
For semi-dilute solutions, how does viscosity change with increasing shear rate?
As the shear-rate increases, the random coil orientations begin to align, reducing viscous drag and intermolecular entanglements, lowering the effective viscosity.
When are colloids stable?
When the repulsive forces between two particles are sufficient to balance the attractive interactions trying to cause aggregation.
Define: flocculation, coagulation, sedimentation, creaming
Flocculation: reversible aggregation
Coagulation: irreversible aggregation
Sedimentation: coagulated aggregate is denser than the medium
Creaming: coagulated aggregate is less dense than the medium.
What is the decay length (in terms of coagulation). What does a short decay length show?
= the mean height at which the number density or particles fall to 1/e (37%) of their value at the bottom (sedimentation) or top (creaming). Short decay length (alpha) suggests particles interact over large distance.
Outline terminal velocity for a colloidal particle for a small and large particle.
Constant speed of a colloidal particle when the buoyant force = drag force.
For small particles (100nm) → v is slow → creaming happens on a much slower timescale → metastable.
For larger particles (1 micron), v is larger
Name one way we can speed up sedmintation.
Increase the gravitational force using centrifuge to destabilise colloidal suspension.
Name 3 ways to prevent sedimentation/creaming.
Stirring (input energy).
Increase viscosity.
Structure the fluid so it is elastic until shear is applied (gels, clay particles).
How does the VDW interaction energy change when going from lange-range interactions between colloids to short range (D « R)?
F = -A/12piD² to F = -AR/12D. Gone from F scaling as 1/D² to 1/D.
Define the Hamaker constant
Determines the effective strength of the VDW interactions between colloids.
Around 10^-20 to 10^-19 J. Larger A = greater interaction.
Explain the 2 different ways polymers can cause flocculation
Bridging flocculation: low coverages of adsorbed polymer bridge two colloids together.
Depletion flocculation: for non-adsorbing polymers. Polymer chains are excluded from the region between the particles by osmotic pressure.
Explain how steric stabilisation effect of polymers keeps colloidal particles apart.
For higher coverages. Confining an adsorbed polymer between two colloidal particles reduces the number of conformations → entropic loss. For grafted or adsorbed polymers. Occurs when tails of polymer chains are long so that attractive force do not dominate.
How are polymer brushes a good example of steric stabilisation?
Dense grafted polymers → extension of polymers due to excluded volume effect + osmotic force. Opposed by elastic force (trying to keep chain ends together). Best in good solvents.
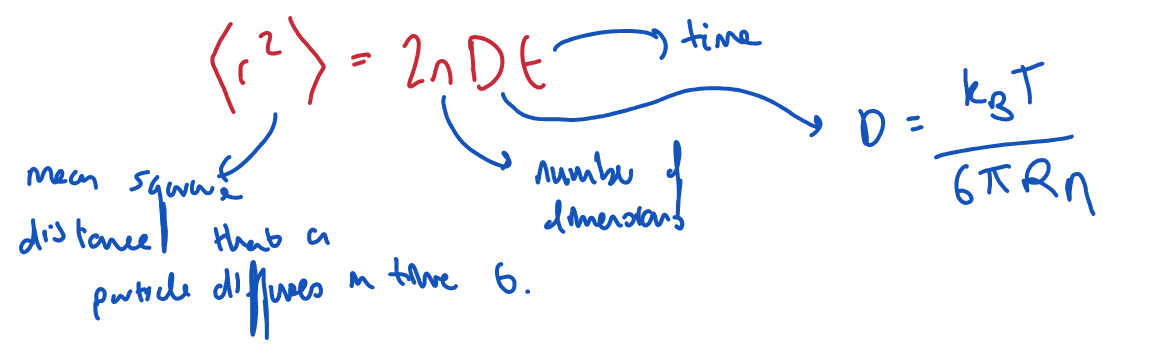
Give the equation for the distance a particle diffuses under Brownian motion.
Define the electrical double layer
Which two factors impact ion distribution in the electrical double layer?
State the Debye-Huckel equation.
For surface potential.
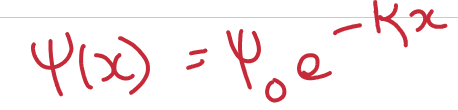
Define the Debye length. How does it change with: valency, ion concentration.
The decay length of the potential = the mean distance of the excess counterions from the surface.
Debye length scales as I^-0.5. Debye length decreases with higher valency or higher concentration of ions.
Outline DLVO theory
Total potential energy = repulsive potential energy (double layer repulsion) + attractive VDW potential energy.
Case3: High electrolyte concentration. If the potential energy curve has a deep attractive minimum at small separations, the system is unstable and association will occur (flocculation or coagulation).
Case 2: Intermediate electrolyte concentration. Potential energy curve has a small secondary minimum separated from the primary minimum by a barrier. If the barrier is large compared to kBT, reversible flocculation can occur -> particles form weakly bound aggregates or flocs.
Case 1: Low electrolyte concentration. No secondary minimum and the potential energy barrier is large compared to kBT, few particles can fall into the primary minimum so the dispersion is kinetically stable.
What is the Schulze Hardy rule?
As the valency increases → rapid fall off of critical coagulation concentration (concentration of electrolytes at which coagulation becomes rapid)
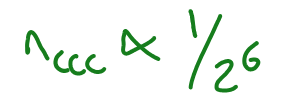
Define surfactants and there effects on colloids and microemulsions
= Amphiphilic molecules, adsorb at air-water interfaces, reducing the surface tension.
Stabilises microemulsions thermodynamically.
Stabilises colloids kinetically.