MedChem
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
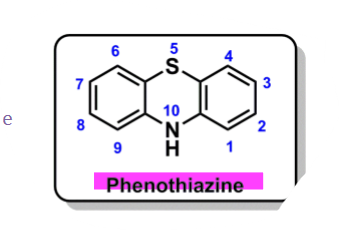
What is the pharmacophore in antipsychotics?
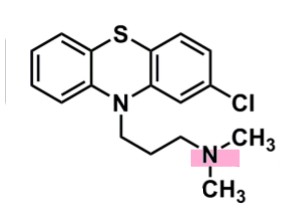
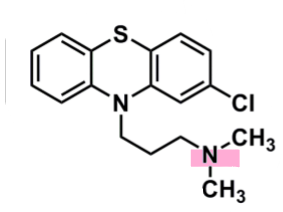
Chlorpromazine
What is important about chlorpromazine?
pharmacophore: phenothiazine
EWG Cl @C2
enhances binding and penetration into CNS
3C spacer to tertiary amine
responsible for pharm activity
metabolites are not active, or very weak
dialkylamines
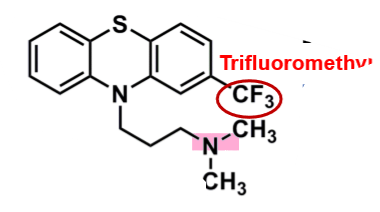
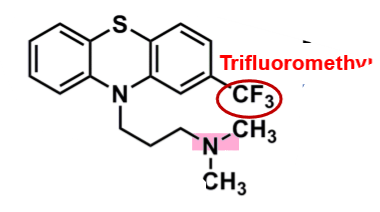
Triflurpromazine
What is important about triflupromazine?
pharmacophore: phenothiazine
trifluoromethyl @C2
more lipophilic → enhances binding affinity and increases EPS
superior potency versus chlorpromazine
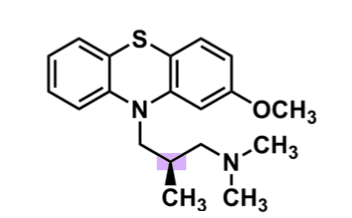
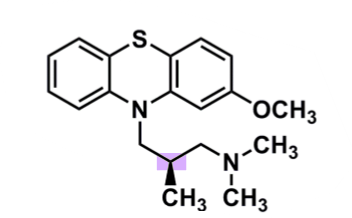
Methotrimeprazine
What is important about methotrimeprazine?
pharmacophore: phenothiazine
EDG @C2
less lipophilic → lower CNS side effects
polar substituent → decreased half-life and potency
methyl group of 3C spacer
R-enantiomer active (also analgesic)
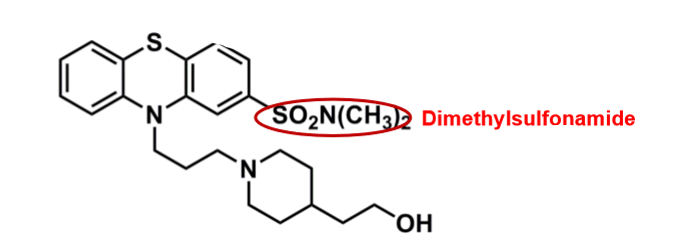
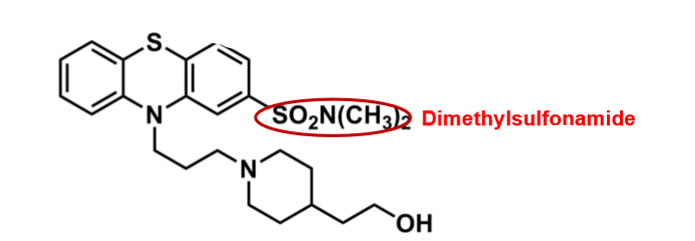
Pipotiazine
What is important about pipotiazine?
pharmacophore: phenothiazine
polar dimethylsulfonamide @C2
decreases CNS penetration, EPS and half-life
sulfonamide, has risk of allergy
cyclic amine (piperidine) + alcohol group
more polar
better binding affinity to D2 receptor
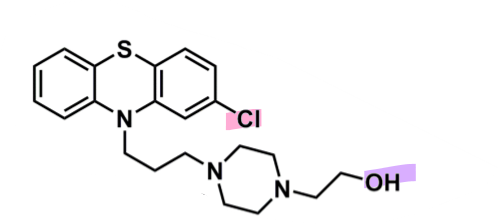
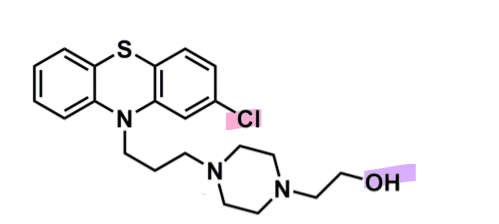
Perphenazine
What is important about perphenazine?
pharmacophore: phenothiazine
EWG chlorine @ C2
piperazine + alcohol after 3C spacer
more lipophilic so less CNS side effects
enhances binding affinity to D2
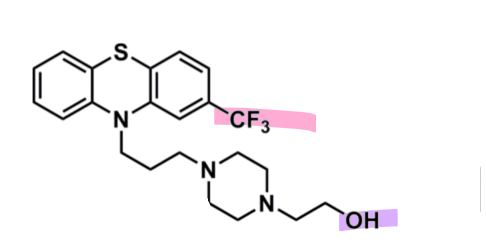
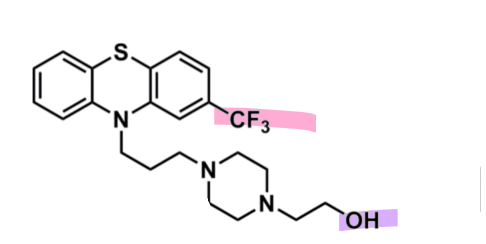
Fluphenazine
What is important about fluphenazine?
pharmacophore: phenothiazine
EWG trifluoromethyl @ C2
superior potency
piperazine + alcohol after 3C spacer
more lipophilic so less CNS side effects
enhances binding affinity to D2
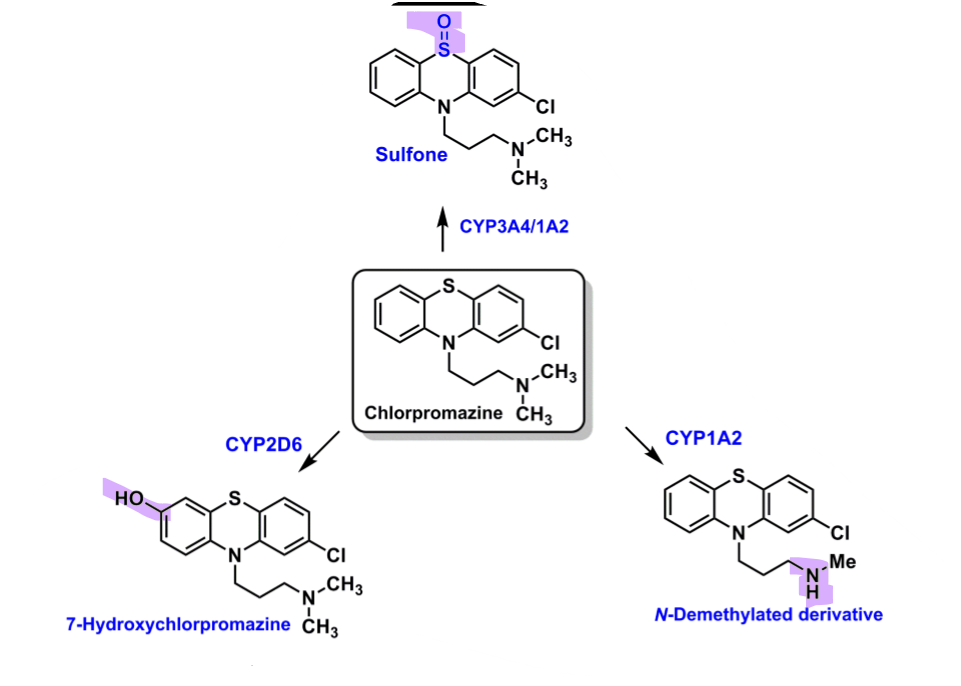
What are the in vivo metabolites of chlorpromazine?
3A4/1A2
sulfone
NO activity
associated with CV toxicity
2D6
oxidation of benzene to hydroxy
some activity
1A2
secondary amine
inactive
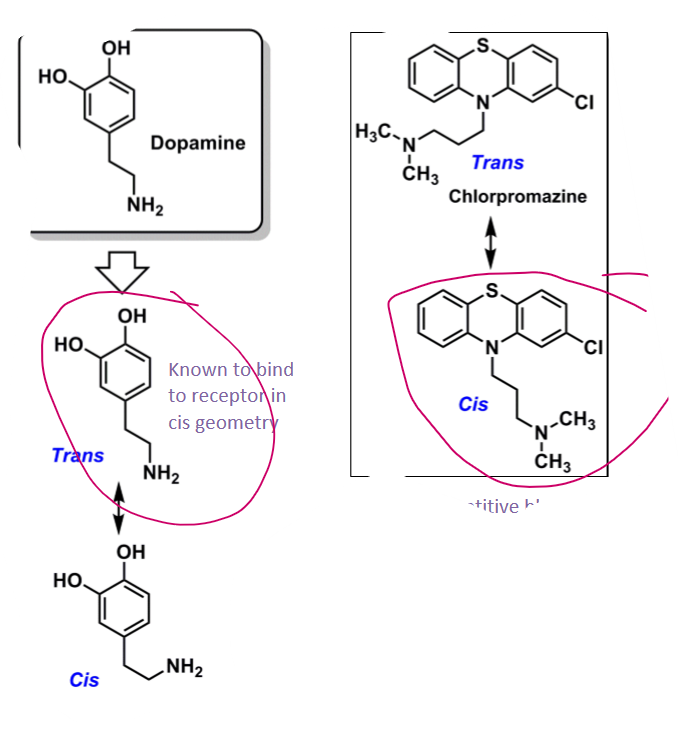
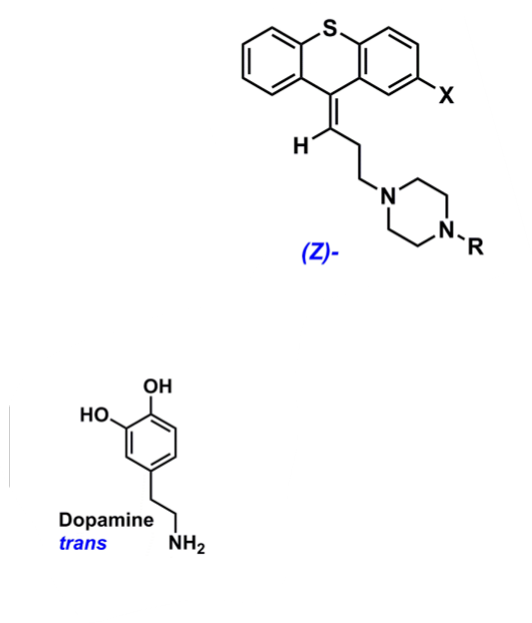
What is the bioactive conformation of phenothiazines?
cis-conformation of phenothiazines have similar geometry as trans-dopamine (active conformation)
will act as competitive blocker and prevent DA from biding
EWG @ C2 will promote bioactive conformation
protonated amine can interact with EWG and cation pi interaction lock in Cis conformation
What is the SAR of phenothiazines?
ring A
EWG @C2/3 increase D2 receptor binding
polar @C2/3 decrease CNS effects
C1/4 substitution decrease activity
ring B - @N10
need 3C spacer; anything else decreased activity
tertiary amine better activity
piperazine > piperidine > aliphatic amine
hydroxypiperazines > alkyl piperidine
ring C
unsubstituted at all positions
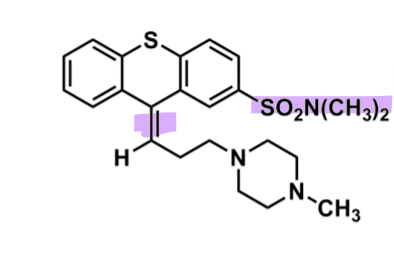
Thiothixene
What is important about thiothixene?
pharmacophore: thioxanthene
lower binding affinity vs phenothiazines
@C10 - double bonded to first C in Z-isomer better activity
piperazine
sulfonylamide @C2
more polar, decreases half-life and EPS
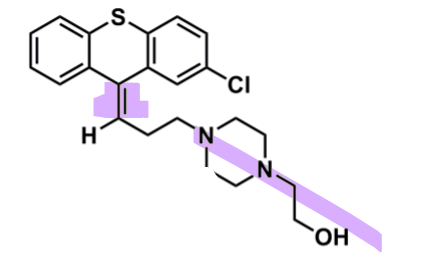
Zuclopenthixol
What is important zuclopenthixol?
pharmacophore: thioxanthene
EWG Cl @C2
@C10 - double bonded to first C in Z-isomer better activity
piperazine + hydroxy → better binding
can be converted to a prodrug ester
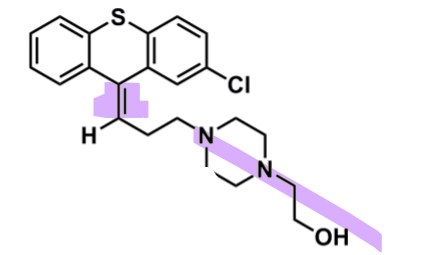
What is the bioactive conformation of thioxanthines?
Z-isomer overlaps with trans-dopamine conformation
piperazine and halogen on the same side
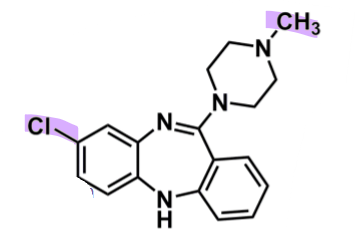
Clozapine
What is important about clozapine?
pharmacophore: dibenzodiazepines
faster dissociation (less tight binding)
piperazine w/ tertiary amine
enhances binding affinity
secondary weak activity
EWG Cl @C2 → enhances binding affinity
central ring
can have positive charge and oxidized species → leading to agranulocytosis
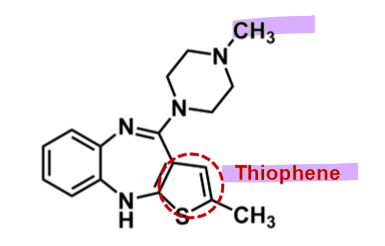
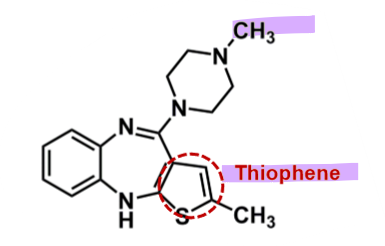
Olanzapine
What is important about olanzapine?
pharmacophore: dibenzodiazepine
similar structure to clozapine → 1 benzene change to thiophene
risk of oxidation of central ring lower → reduced risk of agranulocytosis
enhanced bioavailability and half-life
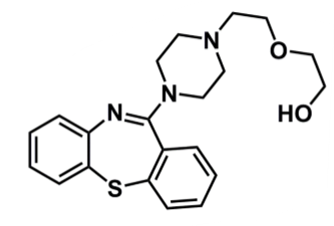
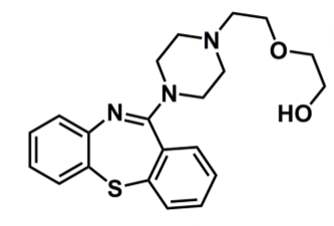
Quetiapine
What is important about quetiapine?
pharmacophore: dibenzothiazepine
similar structure to clozapine
addition of ester → polar agent, conjugation (phase II) reaction can occur so faster elimination (shorter half-life)
change sulphur vs N in central ring → can undergo oxidation to sulphone or sulfoxide which is not active
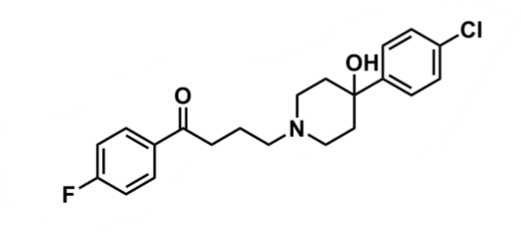
Haloperidol
What is important about haloperidol?
pharmacophore: butyrophenone
4 carbons +benzene + ketone
piperidone + hydroxyl → needed to bind
hydroxyl can be converted to ester prodrug
chlorobenzene → lipophilic and enhances penetration into brain
better binding to D2 and slow dissociation
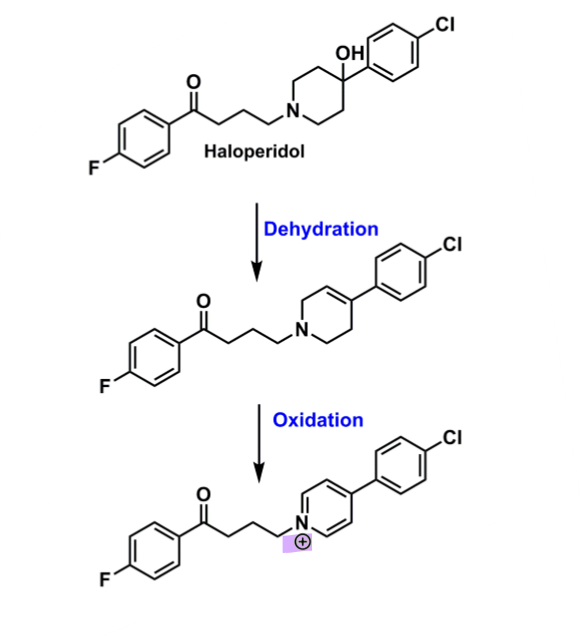
What is the risk of one of haloperidol’s metabolites?
Can produce an aromatic leading to a positive charge on the N of piperidine ring via dehydration + oxidation
positive charge can is a toxic metabolite of DA producing cells in brain
= neurotoxic
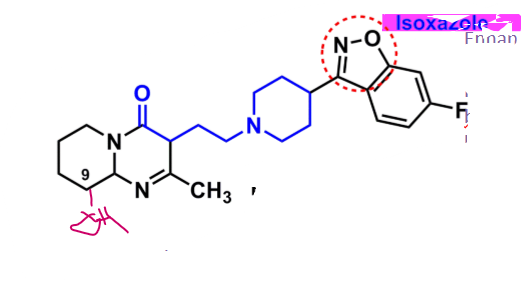
Risperidone
What is important about risperidone?
derivative: butyrophenone-like
isoxazole
enhances binding to 5-HT
increases oral bioavailability and half-life
C9 can undergo oxidation to hydroxy → equal activity
is paliperidone
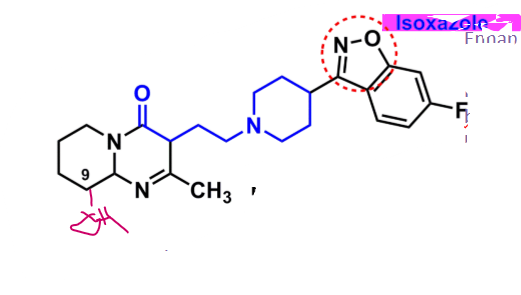
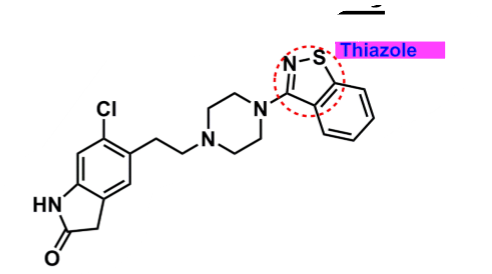
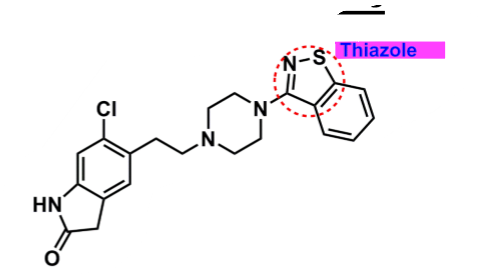
Ziprasidone
What is important about ziprasidone?
derivative: butyrophenone-like
thiazole
enhances binding
can undergo oxidation → lower bioavailability and half-life
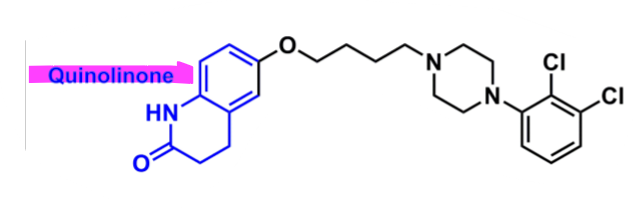
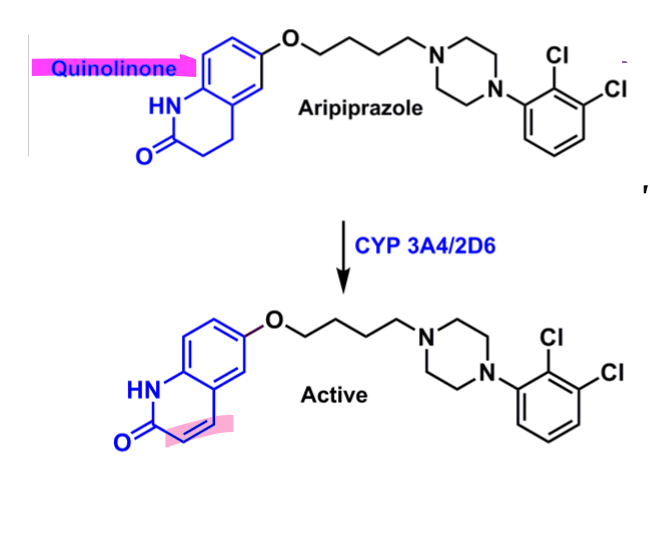
Aripiprazole
What is important about aripiprazole?
butyrophenone-like derivative
quinolinone → enhances binding
convert to 1 double bond → more active and longer half-life (48-68h)
dichlorobenzene
enhances binding affinity
better bioavailability and half-life
more lipophilic and more into brain
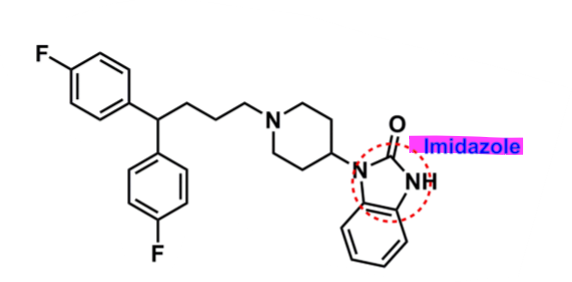
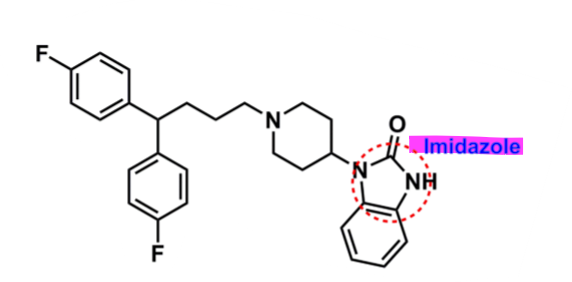
Pimozide
What is important about pimozide?
diphenylbutylpiperidine derivative
diphenol w/ fluorine
lipophilic → enhances half-life (55h)
binds to cholinergic receptors
imidazole for activity
binds to D2 and 5-HT2A