Levels of Protein Structure
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
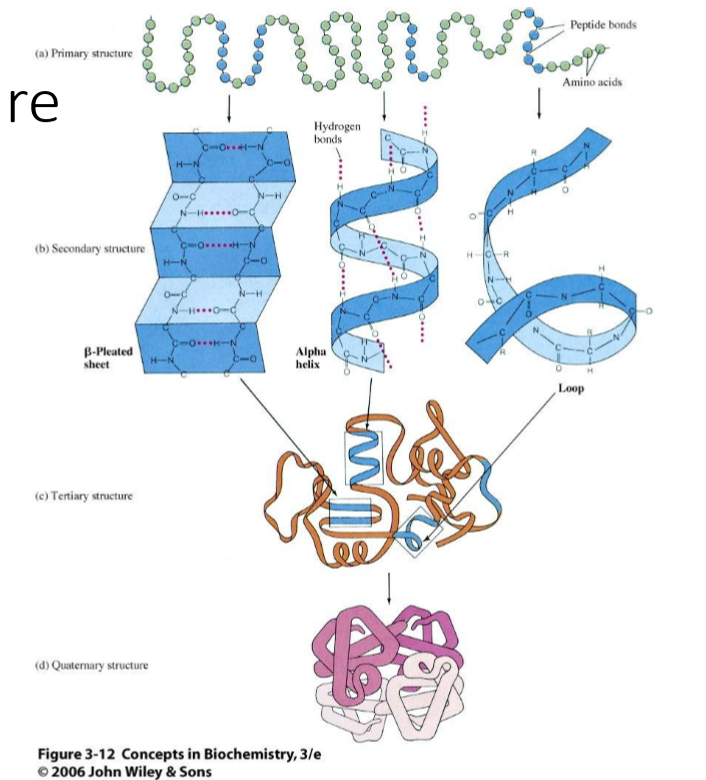
Briefly describe each level of protein structure and what interactions hold them together
primary - sequence of amino acids; covalent peptide bonds between amino acids
secondary - areas of contigous local structure; hydrogen bonds between H of amine and Oyxgen of carbonyl
tertiary - overall 3D shape, noncontinugous sections arranged; hydrophobic affect, sometimes ionic or disulfide
quaternary - multiple polypeptide chains coming together
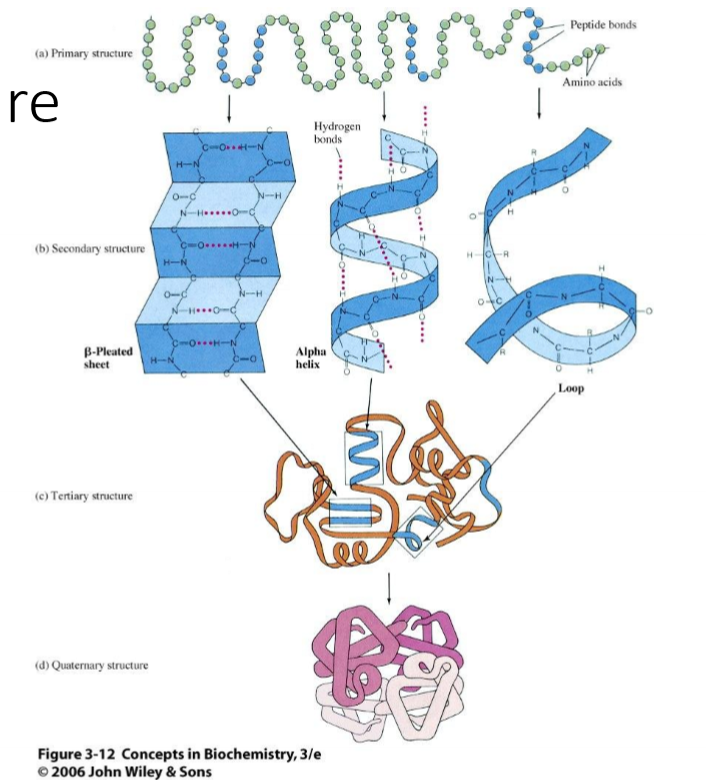
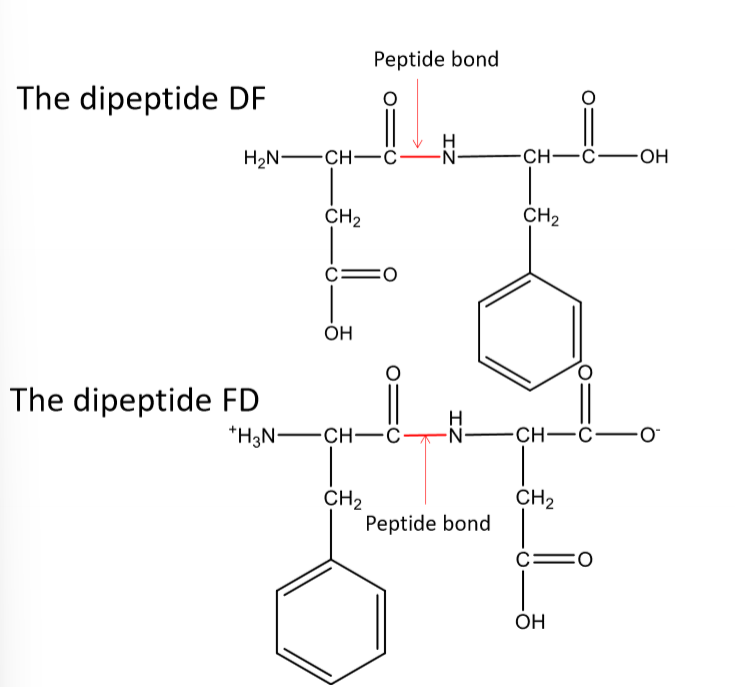
Describe primary strucutre, inlcuding:
what changes happen to the atoms
how the esequnece is written
and how the partial double-bond character of peptide affects orientation
lose H on OH of carboxylic acid on first amino acid and lose H of amine as N joins with the carbonyl
written N terminus to C terminus
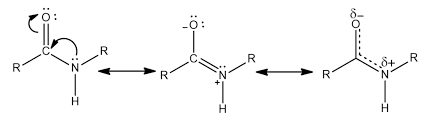
partial double bond character of peptide
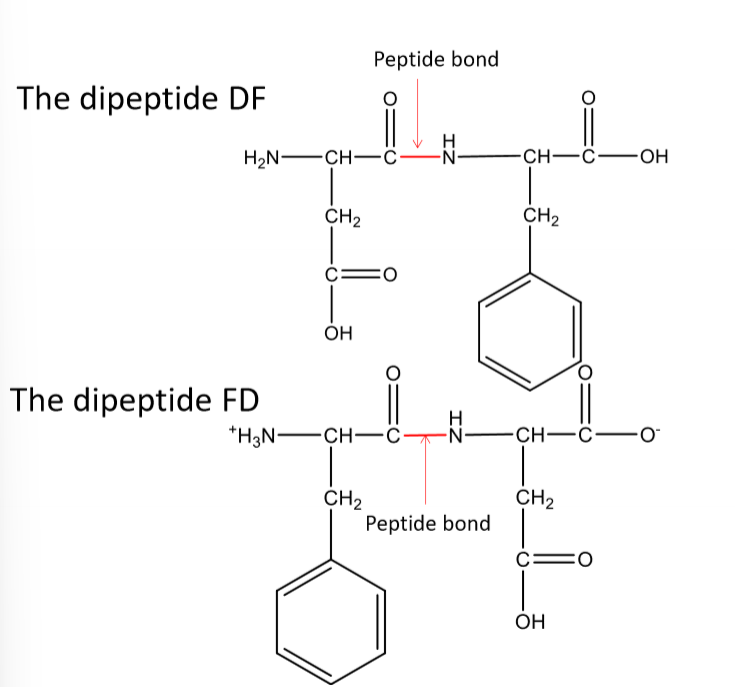
What direction do amino acid chains run?
N terminus to C terminus
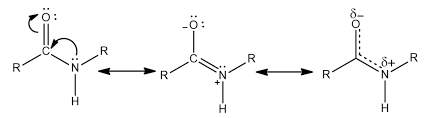
What is the partial double-bond character of a peptide bond and what does it cause?
resonance form of amide created in peptide bond
partial double bond limits rotation on the Nitrogen to only the Hydrogen and R groups
creates a more planar molecule
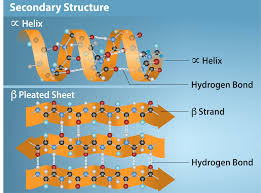
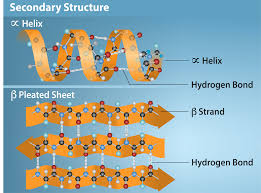
Describe secondary structure, including:
what they are
what interactions hold them together
types and their general info
H bonds between contiguous amino acids
maintained by Hydrogen bonds
alpha helix - coil or spring; right handed helix with side chains facing outward
beta sheets - extended structure with 2 strands, backbone in same place and side chains alteranting between above or below plane
beta turns - 4 amino acids long, change direction of polypeptide chain by 180 degrees, often between beta sheets
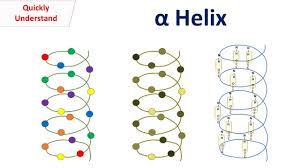
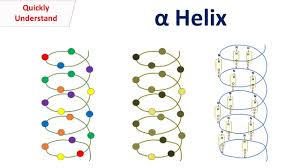
Describe alpha helixes, including their hydrogen bonding, left/right handedness, and common amino acids
most stable secondary structure
each peptide involved in 2 hydrogen bonds (increases stability), neutralizes polarity in each bond and occurs only on backbone
hydrogen bond between adjacent NH (h donor) and carbonyl (H acceptor) of each peptide bond on backbone
coil or spring shaped
Right handed helix (coils to the right)
side chains face outwards
Prole (unflexible) is a helix breaker at end of alpha helix; glycine sometimes
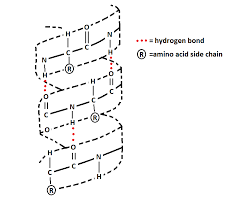
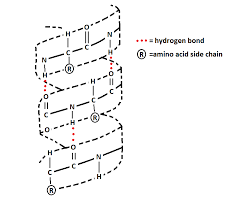
Describe hydrogen bonding on alpha helixes
2 hydrogen bonds on each amino acid
H bond between NH (h donor) and carbonyl (H acceptor) of adjacent amino acids
neutralizes polarity of protein core
h-bonding occurs ONLY on backbone
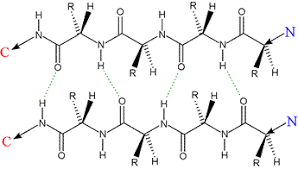
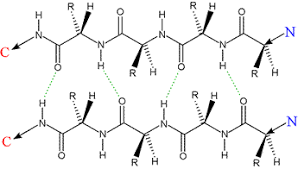
Describe beta sheets, including their structure, hydrogen bonding, orientation, common uses, and types
extended structure with wide space per aa
2 stranded, held together by Hydrogen bond between the backbone NH (h donor) and carbonyl (H acceptor) of other strand
both strands of backboen in same plane, side chains alternate betwen above and below the plane
can form a “barrel” for a pore for ions to pass through; requires a nonpolar and polar side side of molecule (done by alternating Polar and nonpolar amino acids in sequence)
antiparallel, parallel, and mixed
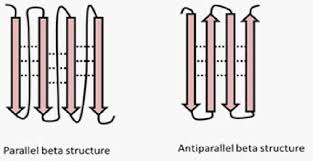
Describe the types of beta sheets
antiparallel - run N to C, then C to N, etc. ; stronger hydrogen bonds
parallel - run N to C, go back to do some other stuff, N to C, etc. Weaker h bonds
mixed - parallel and antiparallel; run N to C, C to N, go back to do some stuff, C to N, etc.
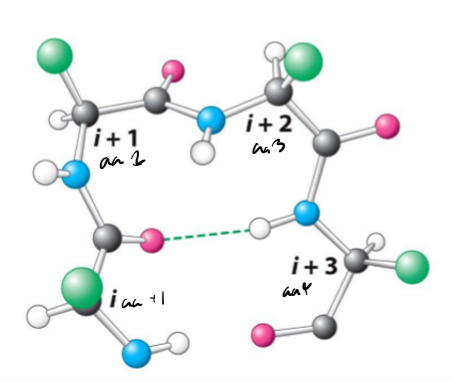
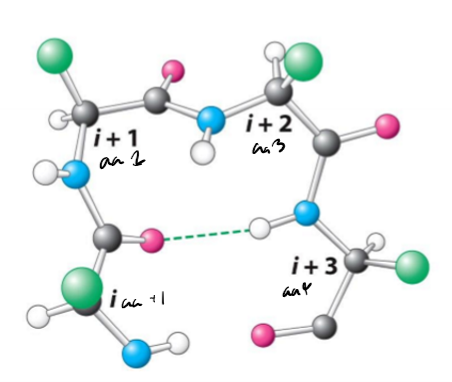
Describe beta turns, inclduing their length, hydrogen bonds, uses, and common amino acids
4 amino acids long
Hydrogen bonds between carbonyl of amino acid and NH of next amino acid on backbone
change direction of polypeptide chain by 180, often between beta sheets
often have proline (naturally has a turn) and glycine (conformationally flexible)
Briefly describe tertiary structure, including what it is, what interactions drive it, and how its organized
overall folding/3D shape
driven by hydrophobic effect
organizes so nonpolar side chains are inside and polar side chains are outside to reduce interactions with H2O and increase entropy
What is the most stable secondary protein structure?
alpha helix
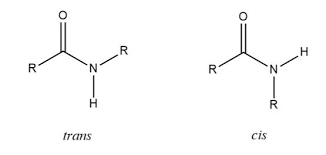
In primary protein structure, what isomer of the partial double bond at the amide is more stable?
trans isomer (R group and amino acid opposite)