Exam 3 Chem 2
1/100
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
101 Terms
Strong Acids
Hydrochloric Acid- HCl
Hydrobromic Acid- HBr
Hydroiodic Acid- HI
Nitric Acid- HNO3
Perchloric Acid- HClO4
Sulfuric Acid- H2SO4
Chloric Acid- HClO3
Buffer stops working when..
If too much acid is added → all A⁻ is used up
If too much base is added → all HA is used up
Strong Bases
Group 1:
lithium hydroxide- LiOH
sodium hydroxide- NaOH
potassium hydroxide- KOH
rubidium hydroxide- RbOH
cesium hydroxide- CsOH
Group 2:
calcium hydroxide- Ca(OH)2
strontium hydroxide- Sr(OH)2
barium hydroxide- Ba(OH)2
ammonium NH4 charge
⁺
acetate C₂H₃O₂
⁻
arsenate AsO₃
³⁻
carbonate CO₃
²⁻
chromate CrO₄
²⁻
chlorate ClO₃
⁻
nitrate NO₃
⁻
nitrite NO₂
⁻
perchlorate ClO₄
⁻
phosphate PO₄
³⁻
sulfate SO₄/sulfite SO₃
²⁻
Silver Ag and Zinc Zn
+ and 2+
If Q < Ksp= more soluble
Solution is unsaturated and will not form a precipitate
Change in G is negative (spontaneous, reaction moves forward with high reactant concentration to reach equilibrium)
If Q > Ksp (lower Ksp)
Solution is saturated and will form a precipitate
Ksp= less soluble
Positive change in G
Decreasing pH increases…
the solubility of salts with weak bases (react with H+ to make a weak acid)
pH formula
pH = −log[H⁺]
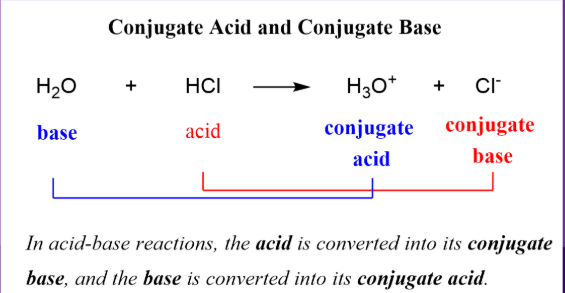
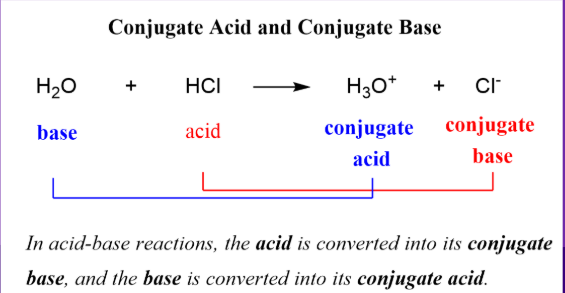
Bronsted-Lowry
Acids will always donate a proton, while bases will always accept a proton
This means that when you are looking at an acid-base reaction, the starting material WITH an H will be your acid, and it will not have an H in the products because it DONATES it
The starting material WITHOUT an H will be your base, and it will have an H in the products because it ACCEPTS it.
NH3+H2O→← NH4+ + OH-
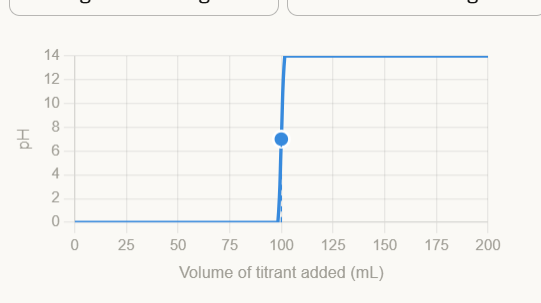
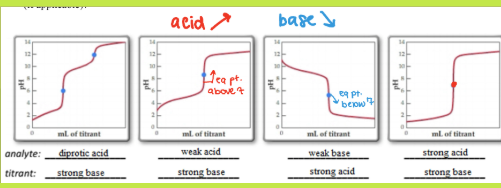
Strong Acid + Strong Base Graph
Equivalence point at pH = 7 (neutral). The salt formed (e.g. NaCl) does not hydrolyze — neither ion reacts with water. The curve is symmetric and steep at the equivalence point.
Weak Acid + Strong Base Graph (look at equivalence point)
Equivalence point at pH > 7 because the conjugate base (e.g. CH₃COO⁻) is basic and hydrolyzes water. The ½ equivalence point is especially important: [acid] = [conjugate base], so the Henderson–Hasselbalch log term = 0, and pH = pKa.
![<p><span>Equivalence point at pH > 7 because the conjugate base (e.g. CH₃COO⁻) is basic and hydrolyzes water. The ½ equivalence point is especially important: [acid] = [conjugate base], so the Henderson–Hasselbalch log term = 0, and pH = pKa.</span></p>](https://assets.knowt.com/user-attachments/b6377448-6537-4c21-a1c1-403082eb5add.png)
Strong Acid + Weak Base graph
Equivalence point at pH < 7 because the conjugate acid (e.g. NH₄⁺) is acidic and donates a proton to water. At the ½ equivalence point, pH = pKa of the conjugate acid (= 14 − pKb of the base).
pH=pKa/pOH=pKb
Weak Acid-Strong Base titration: occurs at the half-equivalence point, the point in the buffer region where exactly half of the initial acid has been converted to its conjugate base, [A-]=[HA]
Strong Acid-Weak Base titration: occurs at the half-equivalence point, the point in the buffer region where exactly half of the weak base is neutralized, base [B] = [BH+] conj. acid
To find pH of weak base and strong acid use what formula?
Ka = Kw (1.0×10⁻¹⁴/)/Kb to find its pH
Lewis
Acid: Electron Pair acceptor
Base: Electron Pair donor
BF3+ :NH3 (Base) → F3B-NH3
Arrhenius
Acid: release H+
Base: release OH-
Acid Example: HCl (Hydrochloric acid)
When dissolved in water, it dissociates to release H+ ions.
Base Example: NaOH (Sodium hydroxide)
When dissolved in water, it dissociates to release OH- ions.
[OH-] → [H+] or [H+] → [OH-]
1 × 10^-14 / [OH-]= [H+] or 1 × 10^-14 / [H+]= [OH-]
[OH-] or [H+] → pH or pOH
-log[OH-]= pOH or -log[H+]= pH
pOH → pH or pH → pOH
14-pH= pOH or 14-pOH= pH
pOH or pH → [OH-] or [H+]
10^-pOH or 10^-pH
Autoionization of Water
H2O(l)⇌H+(aq)+OH−(aq)
Kw=[H+][OH−]=1.0×10−14 at 25°C
Neutral water
[H+]=[OH−]=1.0×10−7
and your pH & pOH would be 7
In a conjugate acid-base pair:
Ka×Kb=Kw (1.0×10−14)
pKa+pKb=pKw (14)
Large Ka
Strong acid (nearly complete ionization)
Small Ka (< 1)
Weak acid (partial ionization)
Strong acids/bases
Completely dissociate in water
Weak acids/bases
Partially dissociate; equilibrium exists between the undissociated species and its ions.
Water is not included in K because
pure liquid
Buffers resist pH change due to
presence of both weak acid or base with its conjugate.
Adding acid/base shifts equilibrium but
barely affects pH in buffer systems.
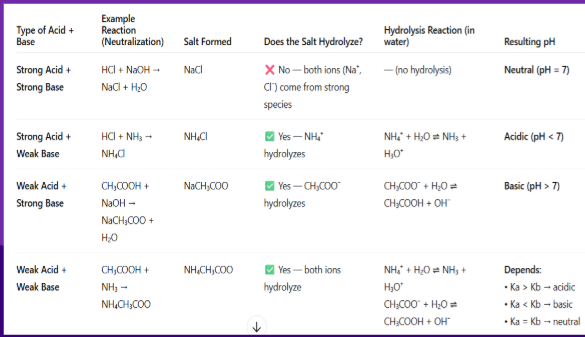
Salt Hydrolysis
When ions in a salt react with water
Some salts do hydrolyze in water; others do not.
Only salts derived from weak acids or weak bases undergo hydrolysis because their conjugate ions can react with water
Salt hydrolysis: If a cation in reaction with water produces a weak base and H3O+…
pH= acidic, low
Salt hydrolysis: If a anion in reaction with water produces a weak acid and produces OH-…
pH= basic, high
Salt hydrolysis: If a cation or anion produces a strong acid or base…
no hydrolysis takes place
When a salt (metal and non-metal // ionic compound) dissociates it looks like
AB→A⁺+B⁻, and if A⁺ or B⁻ can react with water, hydrolysis occurs.
To see if it can occur, we combine A⁺ and B⁻ with H2O and see what is formed

Salt Hydrolysis examples
Weak Acid expression
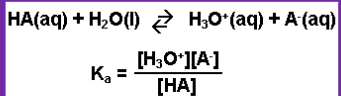
Weak Base expression

Larger Ka and Kb
stronger acid or base
Ka/Kb are inversely proportional to…
pKa and pKb
lower (smaller) pKa indicates a stronger acid
lower (smaller pKb indicates a stronger base
Percent Ionization
measures the ionization of an acid or base
Stronger acid → larger Ka → higher % ionization
More dilute → higher % ionization (Le Châtelier: equilibrium shifts right)
Percent Ionization of Acid
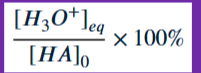
Percent Ionization of Base
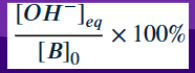
Polyprotic Acids:
Are capable of donating as many protons as they contain
Typically, only the first donation of proton in used when solving for pH
Each dissociation step has a DIFFERENT Ka value
Ka1 > Ka2 > Ka3, understand that the first H will have the highest Ka value when donated
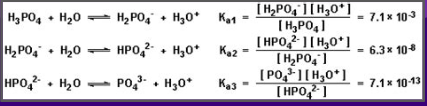
Amphoteric:
Molecules that can act as both a base and as an acid
Common Example: Water (H2O)
Most include polyprotic acids where there is a proton and a negative charge - HPO4 (2-) → acid= PO4 (3-) + base= H2PO4(-) and H2PO4(-) → acid= HPO4(2-) + base= H3PO4
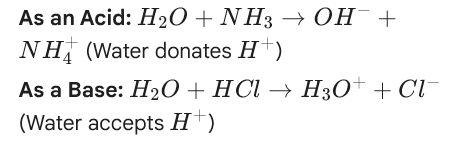
Buffers
Buffers help prevent changes in pH when adding something to the solution
Consist of a conjugate weak acid and base pair
Buffer Capacity
Buffer capacity refers to the amount of strong acid or base that can be added before a drastic change in pH occurs
more moles of buffer= higher buffer capacity
Finding Buffer Capacity
When given choices, eliminate choices that are not buffers
meaning, the pair must be a conjugate weak acid/base pair
Find the smallest number within each pair and cross out the larger one
Compare the smallest numbers. The largest number of this group has the largest buffer capacity.
Make sure add total concentration too (if smallest eliminate that choice)
Check ratio closest to 1:1 is ideal
Limiting amount is the smaller concentration
How to get pKa/PKb fro Ka and Kb
-log(Ka) or -log(Kb)
Titration curve
½ equivalence point→ [HA] = [A-]
Equivalence point→ mol weak acid present=mol OH- added
At the equivalence point, pH does not equal 7 due to the weak acid equilibrium in water ( A + H2O →← HA + OH- soln. becomes basic)
![<ul><li><p>½ equivalence point→ [HA] = [A-]</p></li><li><p>Equivalence point→ mol weak acid present=mol OH- added</p></li><li><p>At the equivalence point, pH does not equal 7 due to the weak acid equilibrium in water ( A + H2O →← HA + OH- soln. becomes basic)</p></li></ul><p></p>](https://assets.knowt.com/user-attachments/41dbc300-df0d-4007-8d23-192359c50914.png)
Henderson-Hasselbalch equation
NEED TO HAVE A BUFFER TO USE THIS EQUATION (use pH= -log[H], etc.)
Captures the buffer region
![<ul><li><p>NEED TO HAVE A BUFFER TO USE THIS EQUATION (use pH= -log[H], etc.)</p></li><li><p>Captures the buffer region</p></li></ul><p></p>](https://assets.knowt.com/user-attachments/e7ce1a69-8380-40fb-87d2-9861ddaf4ec1.png)
When to add and subtract from [A] and [HA]
If you ADD a strong base to a buffer solution, then you add the mole amount to the base and subtract the mole amount from the acid
If you ADD a strong acid to a buffer solution, then you add the mole amount to the acid and subtract the mole amount from the base
MAKE SURE TO USE STOICHIOMETRY
If you add .2 mole of Ba(OH)2, you would add .4 moles of OH
When adding strong acid to a buffer:
the acid will react with the base to form more of the conjugate acid
When solving:
acid moles subtracts from the conjugate base
acid adds moles to the acid
When adding strong base to a buffer:
Base will react with the acid to form more of the conjugate base
When solving:
Base moles subtract from acid moles
Base moles add to conjugate base moles
Titrations
Determine the concentration of an unknown acid or base by reacting it with a known solution
Titrant
solution of known concentration
Analyte
solution of unknown concentration
Equivalence Point
moles of acid = moles of base
Phase 1 (Before titrant is added- before any of your second material added)
at this point all you have in your solution is water and the original acid or base.
If it’s a strong acid or strong base:
Fully dissociated, just use concentration to find pH/pOH.
We can use the [H]->pH and [OH]->[pOH]->[pH]
If it’s a weak acid/base:
Separate the weak acid as reactants into ions as products
Set up an ICE table with Ka or Kb
Your ICE table formula will be the dissociation of the weak acid or base
Your initial amount of acid or base will be the given molarity, and your products will be 0
From there, you can get your H concentration, then go to [H]->pH
![<ul><li><p><span style="background-color: transparent;"><strong> at this point all you have in your solution is water and the original acid or base. </strong></span></p><ul><li><p><span style="background-color: transparent;"><strong>If it’s a strong acid or strong base:</strong></span></p><ul><li><p><span style="background-color: transparent;"><strong>Fully dissociated, just use concentration to find pH/pOH.</strong></span></p></li><li><p><span style="background-color: transparent;"><strong>We can use the [H]->pH and [OH]->[pOH]->[pH]</strong></span></p></li></ul></li></ul></li></ul><p><span style="background-color: transparent;"><strong>If it’s a weak acid/base:</strong></span></p><ul><li><p>Separate the weak acid as reactants into ions as products </p></li><li><p><span style="background-color: transparent;"><strong>Set up an ICE table with Ka or Kb</strong></span></p></li><li><p><span style="background-color: transparent;"><strong>Your ICE table formula will be the dissociation of the weak acid or base</strong></span></p></li><li><p><span style="background-color: transparent;"><strong>Your initial amount of acid or base will be the given molarity, and your products will be 0</strong></span></p></li></ul><ul><li><p><span style="background-color: transparent;"><strong>From there, you can get your H concentration, then go to [H]->pH</strong></span></p></li></ul><p></p>](https://assets.knowt.com/user-attachments/a1249317-e3ea-4817-ac45-e044fd37f686.png)
4 Phases of a titration: Phase 2 (Buffer region)
BUFFER REGION ONLY HAPPENS WHEN YOU ARE DEALING WITH A STARTING WEAK ACID or WEAK BASE
At this point, your weak acid or base will react with an ion from your strong acid / strong base
If it is a weak base and your titrant is a STRONG acid, then HA + OH- → A- + H2O
If it is a weak acid and your titrant is a STRONG base, then B + H+ → BH+ + H2O
Do a BCA table
We use a BCA table here because the reaction goes to completion, unlike an ice table, where the reactant and products will be at equilibrium
get MOLE amounts, not molarity -> need mole amts for HH equation
Using a BCA table means that whichever reactant has LESS moles, you can just use that as your (x) value and subtract that from the other reactants and add it to the product’s side
You will see that when you do this step, your reactant of H or OH will be 0 and you will have left over HA and A- or B and BH
You can plug these values in for HH (25.00mL of 0.100M CH3COOH titrated with 0.100 M NaOH, Ka= 1.8×10^-5 (pKa=4.74))

Phase 3 (equivalence point)
At this point in the reaction / table, all of your original acids and bases have been turned into their conjugates
If strong acid and strong base titration:
pH will be 7
If weak acid and strong base titration: A- + H2O →←HA + OH-
All original HA has been converted to A⁻
Now, all the A⁻ undergoes hydrolysis (reacts with water):
You would use an ICE table to find the amount of OH -> pOH -> pH
If weak base and strong acid titration: BH+ + H2O →← B + H3O+
All original B has been converted to BH+
Now, all the BH undergoes hydrolysis (reacts with water):
You would use an ICE table to find the amount of OH -> pOH -> pH
USE Kw (1.0×10^-14)= Ka x Kb
25 + 25 because 0.002500/0.1000= 0.02500 L= 25mL

Phase 4 (excess strong base or acid)
pH is determined by the excess of strong acid or base in solution.
Calculate moles of titrant added – moles reacted = excess moles.
Divide by total volume → find [H⁺] or [OH⁻].
Convert to pH or pOH.
![<ul><li><p><span style="background-color: transparent;"><strong>pH is determined by the excess of strong acid or base in solution.</strong></span></p><ul><li><p><span style="background-color: transparent;"><strong>Calculate moles of titrant added – moles reacted = excess moles.</strong></span></p></li><li><p><span style="background-color: transparent;"><strong>Divide by total volume → find [H⁺] or [OH⁻].</strong></span></p></li><li><p><span style="background-color: transparent;"><strong>Convert to pH or pOH.</strong></span></p></li></ul></li></ul><p></p>](https://assets.knowt.com/user-attachments/59a7b3ad-e87c-4a9e-b447-86685f370230.png)
Estimate the pH at 10.00 mL NaOH added
0.01000L (10mL)×0.1000M=0.001000mol
Reaction:
HA remaining: 0.002500mol−0.001000mol=0.001500
A⁻ formed: 0.001000
Use Henderson–Hasselbalch:
pH=4.74+log(0.00100/0.001500)= 4.56
After 30.00 mL NaOH added
This is past equivalence.
Excess OH⁻:
0.03000−0.02500=0.00500 L
0.00500L x 0.1000M=0.00500mol OH-
Total Volume:
25.00+30.00=55.00 mL=0.05500 L
[OH-]= 0.000500mol/0.05500L= 9.09×10^-3
(-log) → pOH= 2.04 → pH= 11.96
Molar Solubility (x)
Moles of a solid that dissolve in 1 L of water at saturation.
The solubility product constant (Ksp) of calcium fluoride (CaF₂) at 25 °C is 3.9 x 10^-11. Find the molar solubility (x) of CaF₂ in pure water.
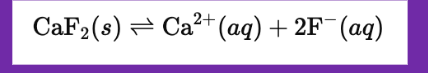
Common Ion Effect
Adding a common ion shifts equilibrium (changes initial ICE values).
Applies to weak acids/bases and solubility.
Usually decreases solubility (shifts left).

Effect of pH on Solubility
If the anion is a weak acid’s conjugate base, it reacts with H₃O⁺ → removes ion → shifts right → increases solubility.

All Brønsted–Lowry acids are
Lewis acids (not all Lewis acids are Brønsted–Lowry)
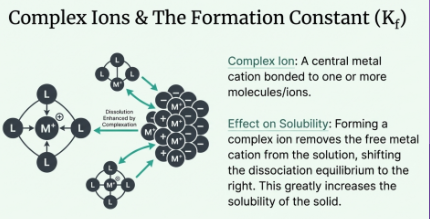
Complex Ion Equilibria
Metal + Lewis base → Complex ion
Some complexes react with water → acidic
Kf = how strongly the complex forms
Overall solubility: Koverall = Ksp × Kf → forming a complex increases solubility
K rules
When reactions are combined, their K values multiply:
K1 × K2 × K3 = Kt (overall equilibrium constant)If you reverse a reaction → invert K: (1/K)
If you multiply or divide coefficients by a factor → raise K to that power: Knew =K^n
Solubility (g/L): = grams of solute that dissolve per liter.
Relates to molar solubility through the compound’s molar mass, pH and Solubility
Insoluble bases dissolve better in acidic solutions (H₃O⁺ reacts with OH⁻)
Insoluble acids dissolve in basic solutions (OH⁻ neutralizes H₃O⁺ or acidic H⁺ ions).
Insoluble salts tend to dissolve better in acidic pH environments → salts containing anions derived from weak acids are generally more soluble in acidic solutions.
Insoluble base in acid:
Mg(OH)2 (s)⇌ Mg2+ (aq) + 2OH−
Adding acid (↑ H₃O⁺) → H₃O⁺ + OH⁻ → 2H₂O
Removes OH⁻ → shifts equilibrium right → more dissolves.
Insoluble acid in base:
Al(OH)3 (s) + OH−(aq) ⇌ [Al(OH)4]−(aq)
Adding base (↑ OH⁻) forms complex ion → increases solubility
ΔH (enthalpy), ΔS (entropy), Temperature (T)
energy change→ Negative = favorable
disorder→ Positive = favorable
temperature → Controls importance of entropy
Equation of free energy change + spontaneity
ΔG=ΔH−TΔS
ΔG < 0 → spontaneous
ΔG > 0 → non-spontaneous
ΔG = 0 → equilibrium
Enthalpy + Disorder Results at different temps.

More moles of gas
↑ entropy
Gas > Liquid > Solid
Smixture>Spure substance
· Shigh temperature>Slow temperature
· Sincreasing mass>Sdecreasing mass
· · More complex structures → greater entropy
· · For allotropes: more ordered forms → lower entropy
Temperature Dependence of ΔG
Determines spontaneity at different temperatures
Threshold temperature (when ΔG = 0):
T=ΔH/ΔS
ΔS universe
ΔS system+ΔS surroundings
Phase Change Formula
ΔS=ΔH/T
T must be in Kelvin
Units: J/mol·K
ΔS∘
∑S∘products−∑S∘reactants
First Law of Thermodynamics
Second Law of Thermodynamics
Third Law of Thermodynamics
energy of the universe is constant, but the various forms of energy can be interchanged in physical and chemical processes.
in any spontaneous process, there is always an increase in entropy.
a perfect crystal at absolute zero (0 Kelvin) has an entropy value of zero.
Gibbs Free Energy
measures the maximum usable energy in a system to perform work at constant temperature and pressure, and it predicts whether a reaction is spontaneous.
ΔG rxn∘
∑nΔGf∘(products)−∑mΔGf∘(reactants)
Standard conditions: 298 K, 1 atm, 1 M
ΔGf∘ of pure elements = 0
ΔG (not at equilibrium)
ΔG = ΔG∘ +RTln(Q)
Q=[products]/[reactants]
Same form as equilibrium constant K
Large K
more negative ΔG° (product-favored)