Carboxylic Acids and their Derivatives: General Exam Questions
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
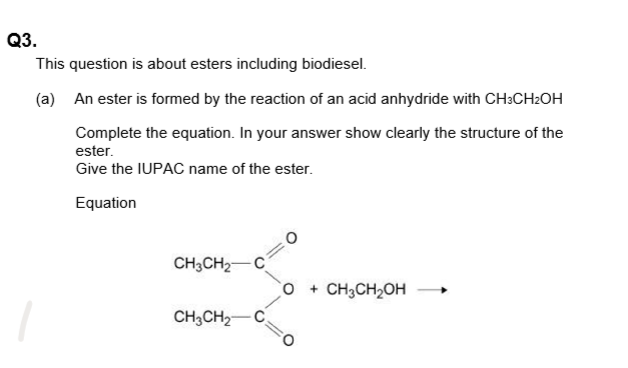
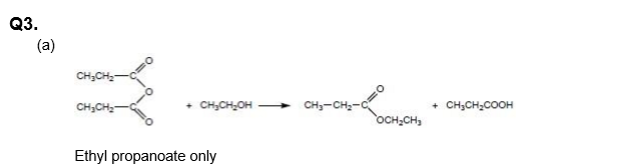
- Acid Anhydride + Alcohol → Ester + Carboxylic Acid

Triglyceride + 3NaOH → Glycerol + 3 Sodium Carboxylate
For saponification questions, remember it always reacts with 3NaOH,Glycerol stays the exact same, and the Carboxylate ions are of the longest of each chain (e.g.C17H33) but with an addition COONa
Biodiesels are the same but the Na switched for CH3
(methyl ester)
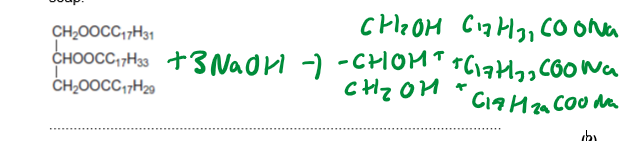
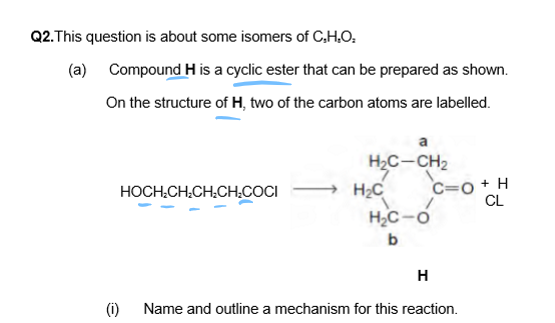
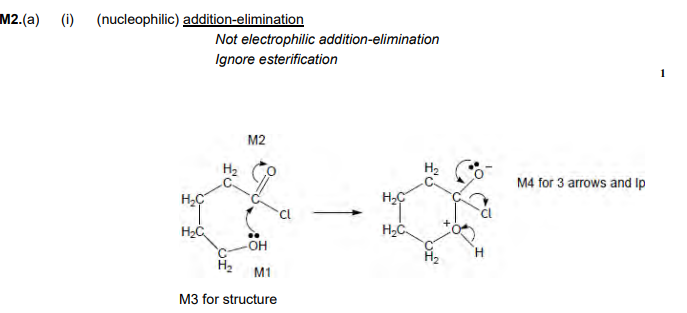
Nucleophilic Addition-Elimination
Cyclic compounds with OH react with themselves to form the compound, as shown here

Ester + H₂O → Fatty Acid(Carboxylic Acid,RCOOH) + Glycerol(CH₂OHCHOHCH₂OH)
b)Keeping the foodstuff dry


Anti-oxidants react with the free radicals, and are used up in the reaction, hence not being regenerated.

Titration Question
-Write out Equation in regards to NaOH, if not, usually a 1:1 MR
-Work backwards, always use ALL data provided

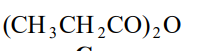
Name this compound.
Propanoic Anhydride
When (CH3CH2CO)2O is heated in water, it is hydrolysed to a carboxylic acid. Write the equation for the reaction that occurs
(CH3CH2CO)2O + H2O → 2CH3CH2COOH
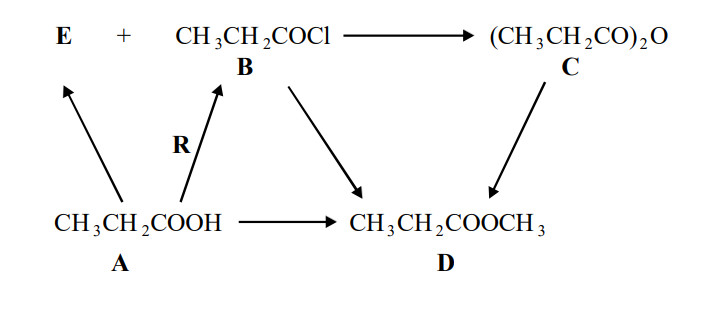
State the appropriate reaction conditions for the formation of compound D from each of the compounds A, B and C.
A: In the presence of strong acid catalyst
B: Room temperature
C: Heat/reflux, not acid
What are the uses of esters?
Solvents
Flavourings
Fragrances
Plasticizers

1. Equipment for Reagents
Propan-2-ol (10 cm³) & Ethanoic acid (10 cm³): Use a measuring cylinder. A pipette is too slow and unnecessary for this precision.
Concentrated Sulfuric acid (5 drops): Use a dropping/teat pipette.
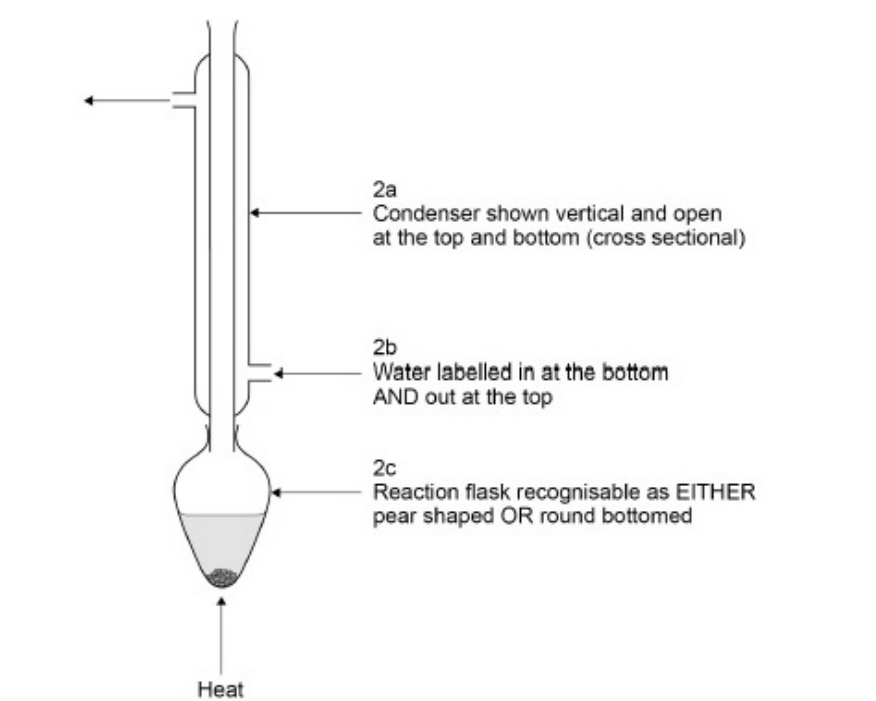
2. Reflux Diagram Checklist
To get full marks, your diagram must show:
Round-bottom or Pear-shaped flask containing the reaction mixture and anti-bumping granules.
Vertical condenser fitted into the flask (open at the top to prevent pressure build-up).
Water jacket with water entering at the bottom and exiting at the top.
Heat source: Use an electric heating mantle or water bath (propan-2-ol and ethanoic acid are flammable).
3. Safety Precautions (Non-eye)
Flammability: Use an electric heater/heating mantle instead of a Bunsen burner because the organic reagents are flammable.
Corrosivity: Wear gloves when handling concentrated $H_{2}SO_{4}$ as it is highly corrosive.

a) 1. Purpose of Sodium Carbonate
The Mark: To neutralise any unreacted/remaining acid (specifically the ethanoic acid and the sulfuric acid catalyst).
b) 2. Precaution
The Mark: Periodically open the tap/invert the funnel and vent/release the pressure while shaking.