Neuroinflammation & multiple sclerosis
1/91
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
92 Terms
How is the central nervous system protected from toxins and pathogens?
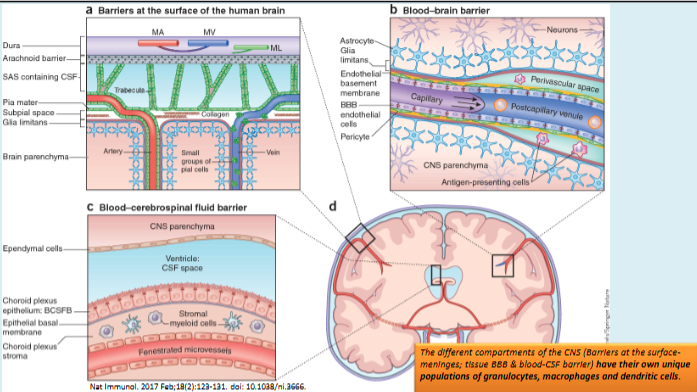
The brain and spinal cord are protected by the blood–brain barrier, a series of specialised barriers that maintain CNS homeostasis and prevent harmful substances from entering neural tissue; disruption of these barriers can contribute to neurological disease.
What is neuroinflammation and what are its common causes?
Neuroinflammation is inflammation of the central nervous system, involving reactive gliosis (activation of microglia and astrocytes). It can be caused by infections (bacterial, viral, or parasitic) or sterile triggers such as trauma, stroke, or neurotoxicity.
How does the immune response function during neuroinflammation?
It involves activation of CNS glial cells (microglia and astrocytes), which interact with immune cells entering from the blood to help restore normal tissue homeostasis.
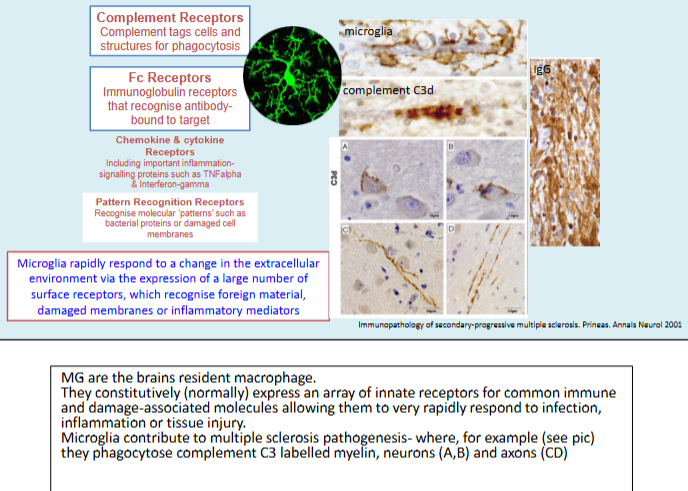
Why are microglia important for immune defence in the brain?
Because the blood–brain barrier limits entry of blood immune cells, microglia act as the brain’s resident immune cells, providing defence within the CNS.
What are the main functions of microglia in the central nervous system?
They remove pathogens and cellular debris, regulate brain development, maintain neuronal networks, and act as key mediators of neuroinflammation.
How are microglial alterations linked to disease?
Dysfunction in microglia is associated with brain development and ageing, as well as neuroinflammatory and neurodegenerative diseases such as Alzheimer's disease and Multiple sclerosis.
How do microglia sense infection or inflammation?
What is astrogliosis and what are its key features?
Astrogliosis is the activation and proliferation of astrocytes, characterised by increased GFAP expression. Activated astrocytes help repair tissue, secrete cytokines to recruit immune cells, and release growth factors (e.g. NGF, BDNF, GDNF) to support cell survival and remodelling.
What is microgliosis and how do microglia respond to CNS injury?
Microgliosis refers to the activation of microglia, which rapidly proliferate, change shape and function, and migrate to damaged areas where they phagocytose pathogens and debris to restore tissue health.
What evidence supports a role for neuroinflammation and reactive gliosis in Alzheimer's disease?
Evidence includes reduced risk with anti-inflammatory drugs, faster decline with systemic infections, experimental models showing inflammation-induced pathology, elevated immune markers in brain and CSF, PET imaging of inflammation, and genetic links (e.g. TREM2, CD33), all suggesting immune involvement in disease progression.
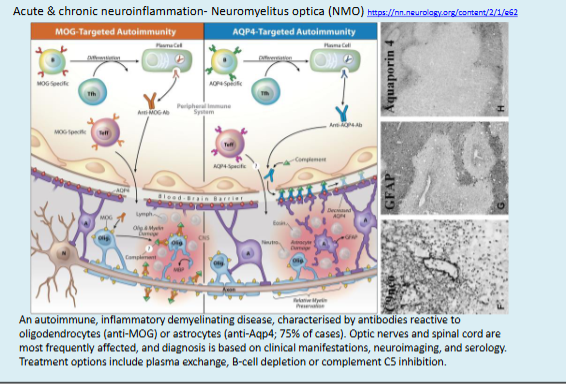
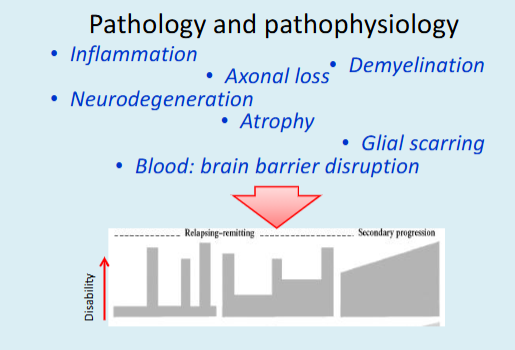
What characterises Multiple sclerosis as a neuroinflammatory disease?
It involves inflammation, demyelination, and neuronal/axonal damage in the brain and spinal cord, disrupting nerve signal transmission and leading to motor, sensory, visual, and autonomic dysfunction.
What are the key epidemiological features of Multiple sclerosis?
It is more common in women, usually begins between ages 20–50, has higher prevalence in Northern Europe and North America, and is a leading non-traumatic cause of disability in young adults, affecting ~130,000 people in the UK and ~2.8 million worldwide.
What do twin and family studies reveal about the genetic risk of Multiple sclerosis?
Concordance rates show higher risk in relatives: ~25–30% in monozygotic twins, 2.3% in dizygotic twins, 1.9% in siblings, compared with 0.3% in the general population, indicating a significant but not deterministic genetic component.
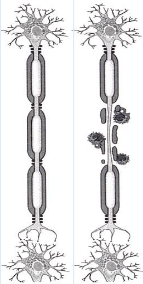
Which genes are linked to increased risk of Multiple sclerosis and how do they influence the disease?
Over 200 risk loci have been identified, including HLA-DR15/Dw2 (affecting antigen presentation and immune control) and IL-7R (involved in T-lymphocyte activation). MS results from a complex interplay of genetic, environmental, and epigenetic factors rather than a single-gene inheritance.
MS is an autoimmune demyelinating disease:genetic risk factors and effective treatments
What are the key epidemiological patterns of Multiple sclerosis?
• MS risk relates to latitude (risk increases with distance from the equator)
• More common in people of Northern European descent
• Rare in native Americans, Australian aboriginals and Japanese
• Consequence of genetic and environmental factors
• Evidence for environmental influence on MS risk- People who are younger than 15 years at the time of migration tend to adopt the MS risk of the country to which they migrate, whereas those who are older than 15 years have a risk of MS that is similar to their country of origin


How does gut health influence the risk and management of Multiple sclerosis?
Gut microorganisms, diet, and obesity affect MS risk; gut dysbiosis is implicated in disease development, and dietary interventions along with self-care are now important aspects of MS management.

What are short-chain fatty acids (SCFAs) and how are they produced?
SCFAs, such as butyric acid, acetic acid, and propionic acid, are bacterial fermentation products generated by the anaerobic breakdown of indigestible dietary fibres and starch in the gut.
What roles do SCFAs play in gut and brain health?
They provide energy for intestinal epithelial cells, help maintain gut-barrier function, and can cross the blood–brain barrier to support BBB integrity.
How do SCFAs influence the nervous and immune systems?
SCFAs activate free fatty acid receptors (FFAR2, FFAR3) on immune and nervous system cells, and stimulate hormone secretion (e.g., peptide YY, GLP-1, ghrelin) by enteroendocrine cells.
How does short-chain fatty acid (SCFA) supplementation affect immune function in MS?
SCFA supplementation reduces inflammation, promotes immune tolerance, and enhances the function of T-regulatory cells, which secrete IL-10 to help control autoimmunity.
What effects does propionic acid (PA) supplementation have in people with Multiple sclerosis?
PA increases T-regulatory cells, restores balance between Tregs and effector T cells (e.g., Th17), and is associated with a reduction in MS-specific symptoms.
What are the clinical signs of MS?
– Visual: Optic neuritis, Monocular vision loss, double vision, defects in visual field
– Motor: Weakness of limbs with spasticity and hyper-reflexia, hemiplegia, poor coordination, ataxia
– Sensory: pain or sensory deficits, Lhermitte’s sign (electric shock radiating down back and triggered by neck flexion), paraesthesia (tingling, pins & needles sensation),
– Cognitive and psychiatric: Dysarthria (problems with speech), Impairment of concentration or memory, depression, anxiety
– Bowel, bladder: Urinary urgency and incontinence
– Sexual: sex drive and dysfunction
– Severe fatigue
• Onset: hours to days
• Recovery: days to months
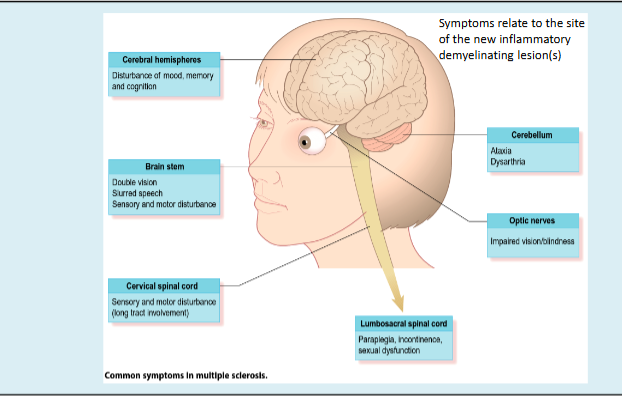
Why are the symptoms of MS so varied ?
Because the location, number, and extent of demyelinated plaques differ in each person, not all lesions cause symptoms, and the overall burden of CNS damage determines the disease experience.
What are the main clinical subtypes of Multiple sclerosis?
Clinically isolated syndrome (CIS), Relapsing Remitting MS (RRMS), Secondary Progressive MS (SPMS), and Primary Progressive MS (PPMS).
How does RRMS typically progress over time?
Most people (>75%) are initially diagnosed with RRMS, which often transitions to SPMS, characterised by fewer relapses but greater cumulative disability.
How are SPMS and PPMS further classified for treatment purposes?
They are described as ACTIVE or INACTIVE (inflammation) and STABLE or WORSENING (disability), which helps determine eligibility for disease-modifying therapies.
What are the key requirements for diagnosing Multiple sclerosis?
Diagnosis requires evidence of CNS lesion dissemination in space and time, and exclusion of other likely causes.
What tests are used to support an MS diagnosis?
Clinical relapses, MRI for new lesions, cerebrospinal fluid analysis (immunoglobulin production, oligoclonal bands), and electrophysiology (e.g., visual evoked potentials), currently guided by the revised McDonald criteria.
How variable is the clinical course of Multiple sclerosis?
The course is highly variable and often unpredictable; about 10% have a benign course with minimal disability after 15 years, while 50% of RRMS cases progress to secondary progressive MS within 10 years.
What is the typical long-term outlook for people with MS?
Around 50% will need walking assistance within 15 years, median survival is ~30 years from diagnosis, and outcomes are improving due to earlier diagnosis, better care, and effective disease-modifying treatments.
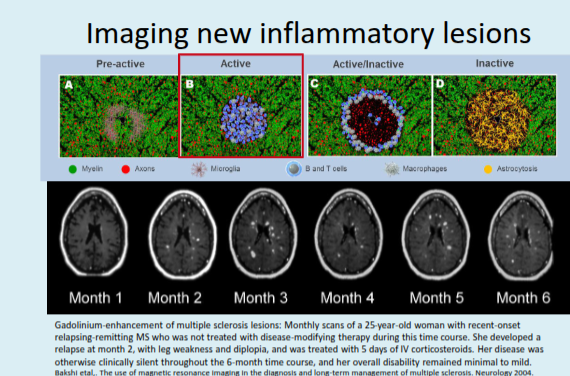
What is the purpose of gadolinium-enhanced T1-weighted MRI scans in MS?
They detect new, active lesions by showing areas where gadolinium crosses the blood–brain barrier, indicating current inflammation.
What do T2-weighted MRI scans reveal in people with MS?
T2 scans show the number and size of lesions, including older, inactive lesions, and are useful for tracking long-term lesion evolution.
What does ring or peripheral enhancement of lesions on a gadolinium T1 MRI indicate?
It indicates breakdown of the blood–brain barrier and active inflammation within those lesions.
What are "black holes" on a T1-weighted MRI scan in MS?
Regions of diminished signal representing chronic, long-standing lesions in the periventricular white matter and corpus callosum.
What changes in white cell counts are seen in the cerebrospinal fluid (CSF) of people with Multiple sclerosis?
CSF white cell counts are usually normal or mildly elevated, reflecting a modest increase in leukocytes.
What is the significance of CSF oligoclonal IgG bands in MS?
The presence of IgG bands in CSF, not seen in blood, indicates CNS inflammation; this sensitive test is positive in over 90% of MS patients but is not specific to MS.
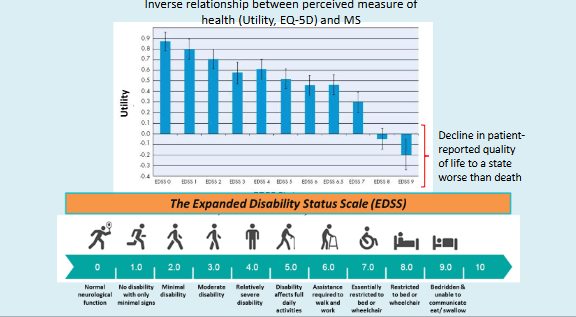
Measuring MS severity and quality of life
The EDSS is still the most commonly used method of assessing disability but can be
combined with other tests that assess mood, memory and upper limb function. The
MSIS-29 is an important questionnaire-based test that someone can complete in their
own time
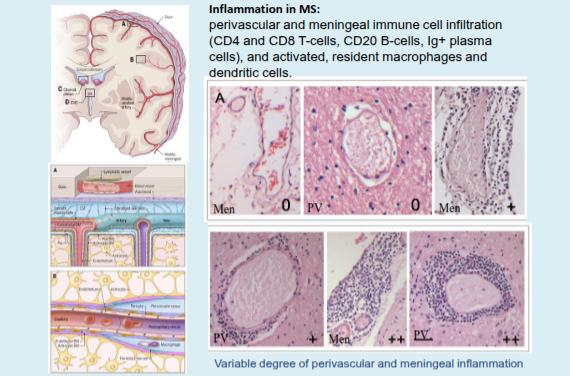
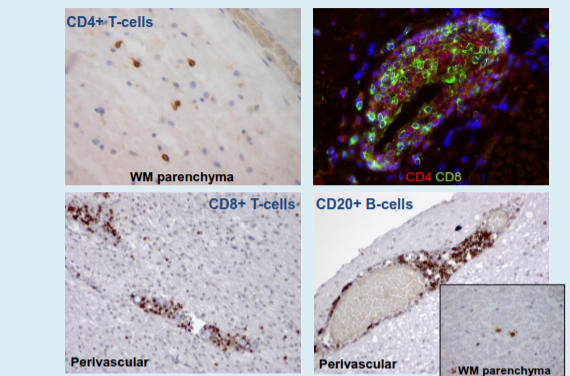
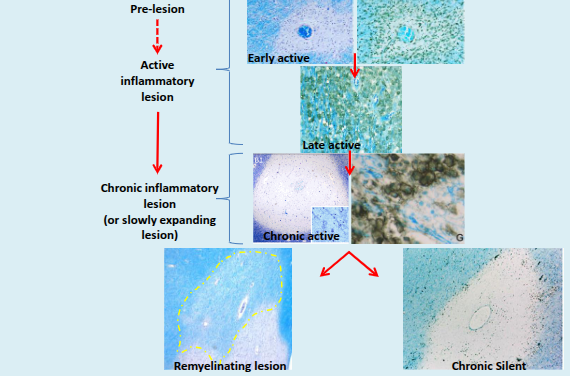
What characterises initial (pre-active and active) MS lesions?
They contain apoptotic oligodendrocytes and activated microglia, occurring before any observable inflammatory lymphocyte response.
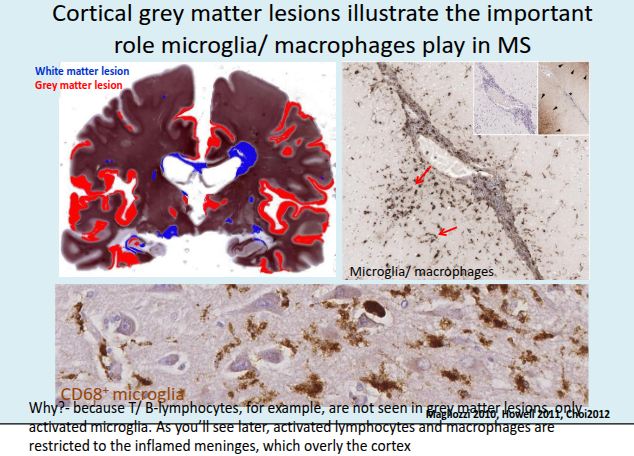
What defines chronic active MS lesions in progressive multiple sclerosis?
They are dominated by activated microglia and macrophages, with T and B cells largely restricted to perivascular and meningeal spaces.
What response does demyelination trigger in MS lesions?
Demyelination triggers an astroglial defence response known as astrogliosis.
What is the role of astrogliosis in MS?
Astrogliosis helps limit damage and supports compensatory remodelling and recovery of neural function, but it can also contribute to tissue damage.
What do BA-1+ microglia/macrophages indicate in MS lesions?
They contain myelin proteins, indicating recent or ongoing myelin phagocytosis; early myelin damage involves myelinosome formation, which is surrounded by microglial/macrophage processes and promoted by anti-myelin antibodies and complement.
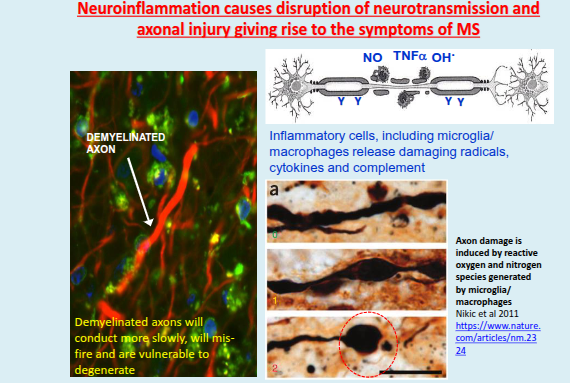
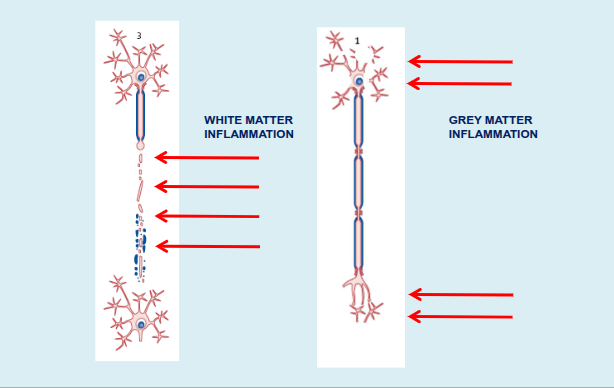
How does inflammation affect nerve function in MS?
Inflammation can cause conduction block, alterations at the node of Ranvier, and mitochondrial disruption, leading to impaired nerve signalling.
What are the effects of demyelination on nerve conduction in MS?
Demyelination can lead to conduction block, slowed conduction, an increased refractory period, axonal cross talk, and eventual axonal and neuronal degeneration.
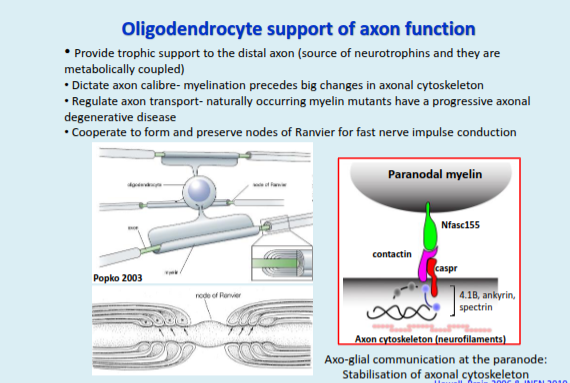
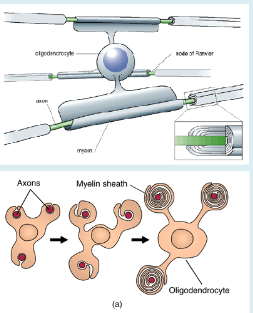
How do oligodendrocytes support axonal function?
They provide trophic and metabolic support to axons, regulate axon calibre and transport, and work with axons to form and maintain nodes of Ranvier; axo-glial communication at the paranode also stabilises the axonal cytoskeleton.
The take home message here is that myelin is more than an inert insulating membrane.
Myelination stabilises axonal proteins and elements of the axon cytoskeleton (without
which, axons can degenerate- next slide).
What does the accumulation of organelles in axons of PLP-null mice indicate?
It reflects impaired (retarded) axonal transport, demonstrating that oligodendrocytes and myelin are essential for normal axon transport.
How do PLP-null mice show the role of oligodendrocytes in ion channel organisation?
They exhibit disrupted distribution of Na⁺ channels at the node and K⁺ channels at the juxtaparanode, showing oligodendrocytes are required for proper ion channel maintenance.
What do naturally occurring myelin mutants reveal about oligodendrocyte function?
They develop progressive neurological disease with axonal damage and loss, mimicking human leukodystrophies and highlighting the importance of myelin in axon survival.
What is the significance of these studies for multiple sclerosis (MS)?
They show that oligodendrocyte dysfunction can drive neurodegeneration, suggesting that promoting myelin repair may help slow or prevent disease progression in MS.
What is myelin?
➢ Multiple layers of oligodendrocyte (or Schwann cell) membrane that is tightly packed around the axon,
whereby the cytoplasm is squeezed out by the fusion of the inner leaflets of the plasma membrane.
➢ Oligodendrocyte membranes are enriched with lipids, inc. galactolipids, phospholipids and cholesterol
➢ Acts as an electrical insulator to enable fast and efficient transduction of action potentials
➢ Cytoplasm filled channels exist within the myelin sheath to allow movement of essential
macromolecules to distal parts of oligodendrocyte AND axon
➢ Disruption of myelin results in damage to the oligodendrocyte AND to the axon
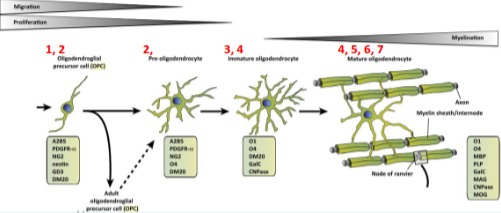
Recall the stages of myelination
1. Proliferation and migration of oligodendrocyte precursor cells (OPCs)
2. Recognition of axons
3. Differentiation of OPCs to myelin forming oligodendrocytes
4. Myelin membrane outgrowth and wrapping of axons
5. Trafficking of myelin membrane proteins
6. Compaction of the myelin sheath
7. Formation of the node of Ranvier
Formation of myelin
-Oligodendrocyte process contacts axon
-Leading process tucks under and extends around axon forming multiple wraps
-Myelin eventually becomes compacted and cytoplasm is extruded (squeezed out)
What is the role and prevalence of oligodendrocyte precursor cells (OPCs) in the adult CNS?
OPCs make up around 5% of adult brain cells, continue to divide and die, and can differentiate into myelinating oligodendrocytes; existing oligodendrocytes also maintain and replenish myelin throughout life.
What is the significance of OPCs for multiple sclerosis (MS) treatment?
OPCs are present in and around demyelinated lesions and, alongside surviving oligodendrocytes, offer potential for remyelination; however, current therapies mainly reduce inflammation and do not directly promote repair, raising interest in enhancing natural remyelination.
What are micropillar arrays used for in multiple sclerosis research?
They are a high-throughput screening platform used to identify compounds that promote OPC differentiation and myelin membrane wrapping, highlighting potential remyelinating therapies.
What key findings came from micropillar array drug screening studies?
A cluster of antimuscarinic compounds was identified and validated in both in vitro and in vivo models of demyelination and remyelination.
What evidence supports the use of clemastine fumarate in MS?What evidence supports the use of clemastine fumarate in MS?
Clinical studies showed electrophysiological evidence of remyelination, including improved nerve conduction (via VEP) and preliminary improvements in vision in patients with optic neuritis.

What is the general target of disease-modifying treatments (DMTs) in MS?
All DMTs primarily target the peripheral immune response to reduce inflammation and disease activity.
What treatments are used for acute relapses and relapsing-remitting MS (RRMS)?
Acute relapses are treated with high-dose steroids, while RRMS is managed with immunomodulatory therapies such as interferon-β, glatiramer acetate, teriflunomide, natalizumab, fingolimod, and ocrelizumab.
What options exist for progressive MS and what is their limitation?
Treatments like ocrelizumab and siponimod can help slow disability progression, but there are fewer effective therapies for SPMS/PPMS and they benefit only a minority of patients.
What is the mechanism and clinical use of Natalizumab in MS?
Natalizumab is a humanised monoclonal antibody targeting the α4 integrin subunit, blocking lymphocyte migration across the vessel wall into the CNS; it is used in active RRMS or when first-line treatments fail.
What are the key risks and side effects of Natalizumab?
It is associated with rare cases of progressive multifocal leukoencephalopathy (PML), as well as side effects such as joint pain, fever, and allergic reactions.
What is the “imaging paradox” in multiple sclerosis?
It refers to the poor correlation between MRI-detected white matter inflammation/demyelination and clinical disability, meaning lesion load does not reliably reflect disease worsening.
Why might MRI findings not fully reflect MS progression?
MRI often misses grey matter lesions, and while immunomodulatory therapies reduce white matter lesions and relapses, disease progression correlates more strongly with grey matter atrophy.
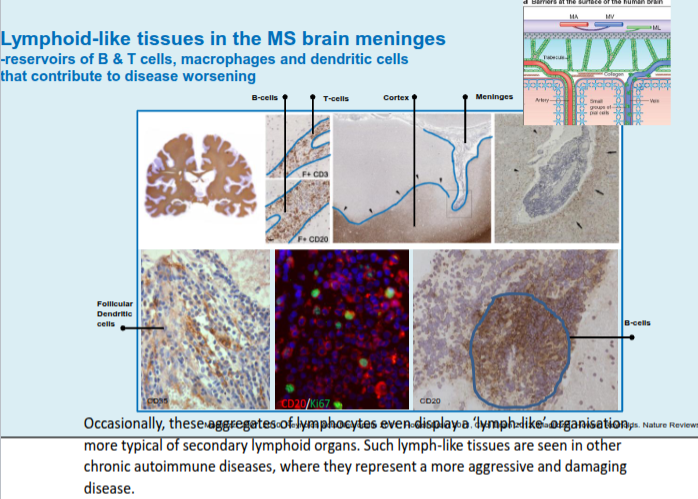
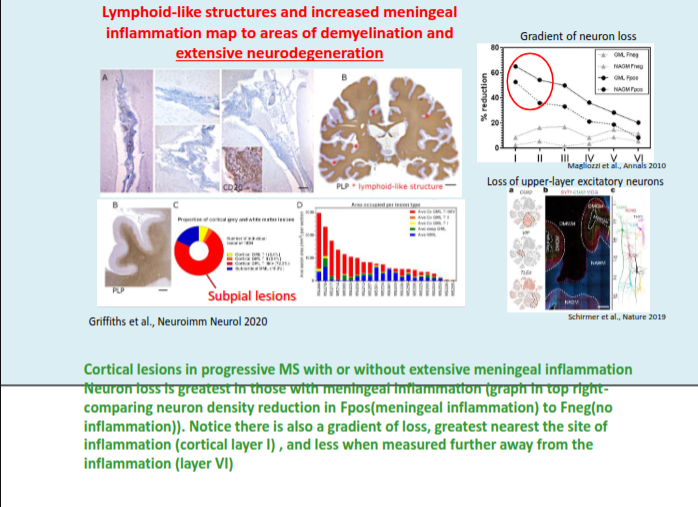
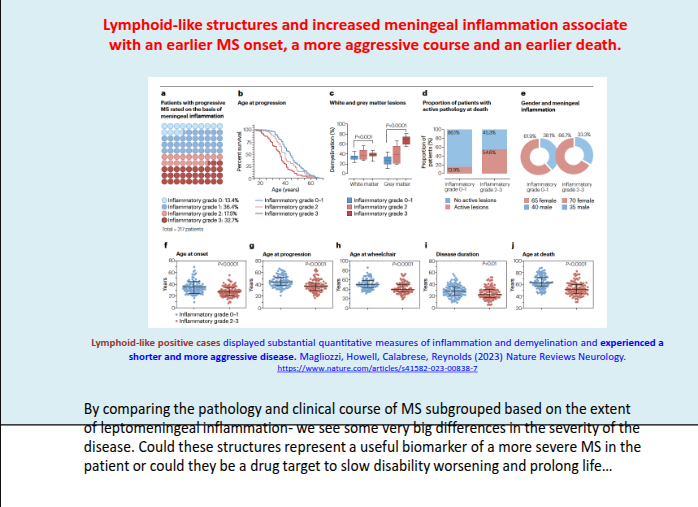
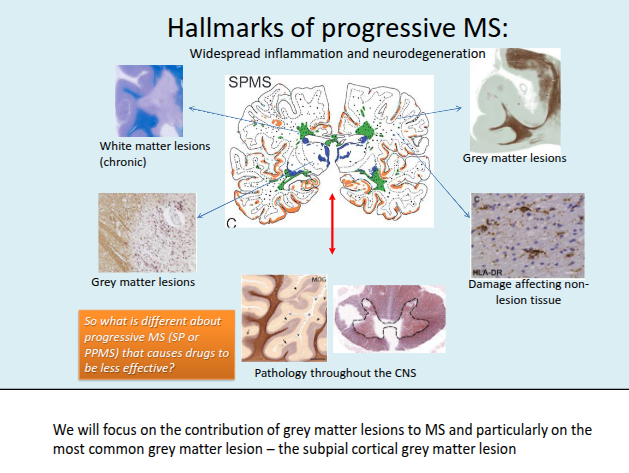
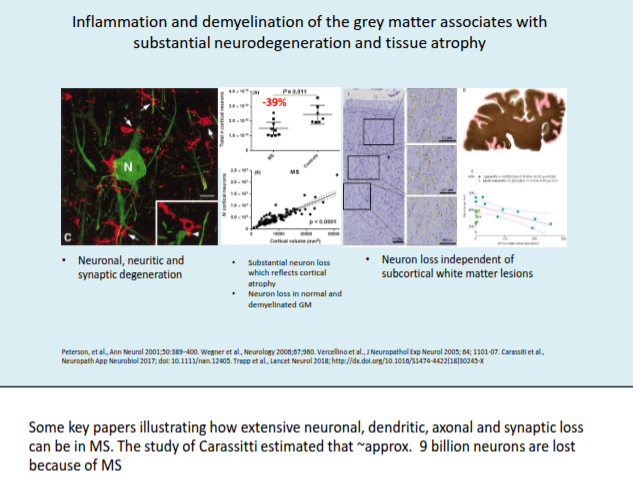
What is the role of grey matter pathology in progressive MS?
Cortical and deep grey matter lesions are a hallmark of the progressive phase, contributing to neuronal loss and overall brain atrophy.
How does grey matter atrophy relate to MS progression and disability?
Grey matter atrophy correlates more strongly with disease subtype and disability than white matter atrophy, and increasing cortical and cerebellar atrophy predicts conversion to secondary progressive MS (SPMS).
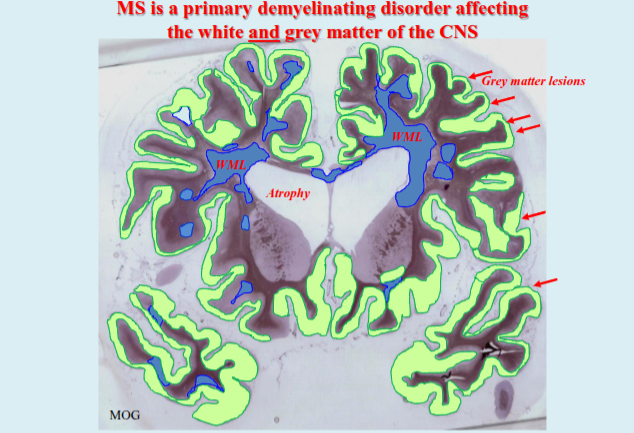
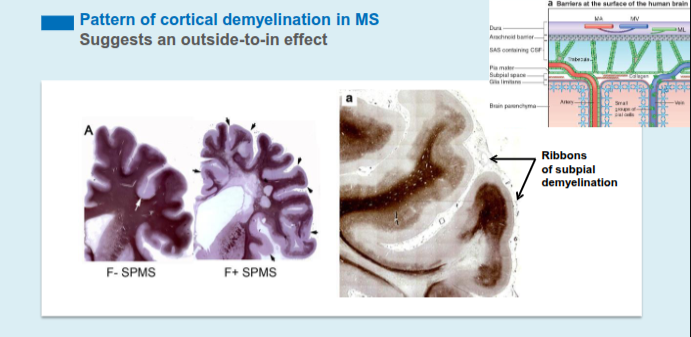
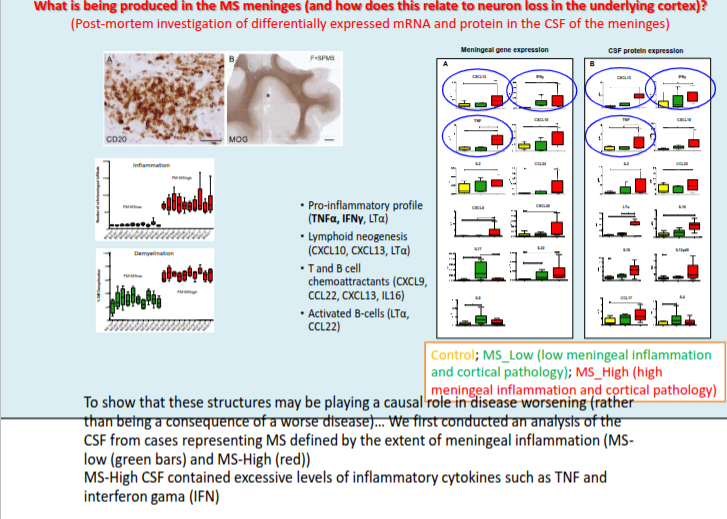
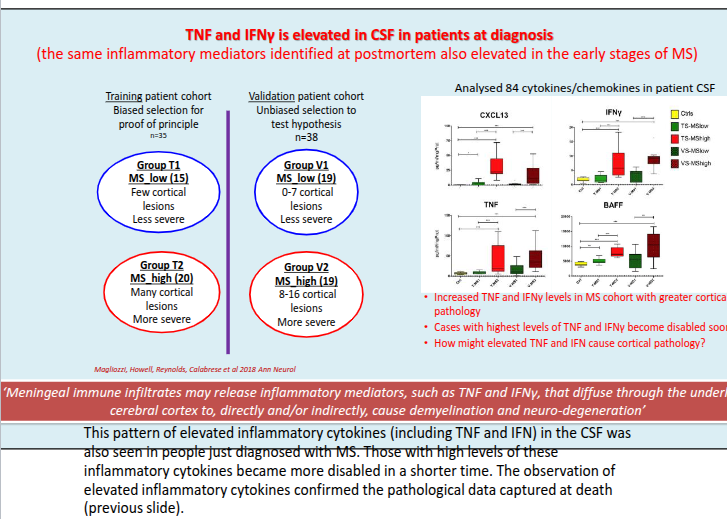
What is the typical pattern of cortical demyelination in MS and what does it suggest about disease origin?
Cortical demyelination commonly presents as ribbon-like subpial lesions in the outer cortical layers, closely associated with inflamed meninges and following gyri and sulci, suggesting an “outside-to-in” disease process.