Introduction to Lipid Catabolism
1/112
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
113 Terms
What represents one of the body’s most energy- rich biochemical systems centered around the breakdown and utilization of fats as fuel
Lipid metabolism
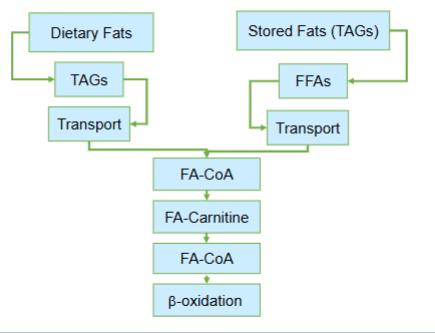
At its core Lipid Metabolism is divided into two fundamentally different physiological contexts—
What are they?
handling of dietary lipids immediately after a meal
mobilization of stored lipids duyring energy demand such as fasting

After eating, what mode is the body in
an energy storage
What happens during energy storage mode
incoming lipids from diet must be processed, packaged, and distributed safely

During fasting or low blood glucose , what mode does the body shift to
an energy mobilization mode
what happens during energy mobilization mode ( fatty acids needs to be mobilized to give energy)
stored fats are broken down to sustain vital processes
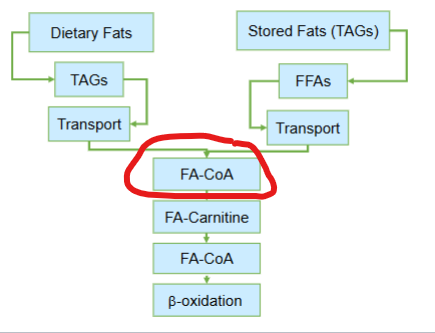
Despite the differences between dietary lipids and storage lipids , what central metabolic fate do both pathways ultimately converge at
the conversion of fatty acids in to fatty acyl-CoA
What does the conversion of fatty acids to fatty acyl-CoA do
it commits fatty acids to mitochondrial degredation through beta oxidation
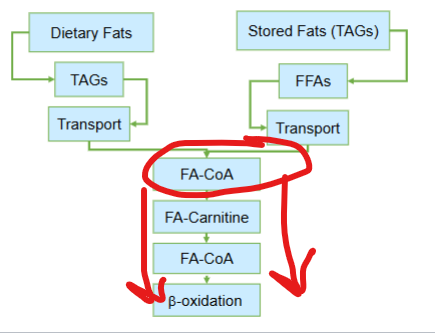
The convergence of fatty acids from dietary or stored lipids to fatty acyl-CoA to mitochondrial degredation is critical in what way?
it represents a point of metabolic integration— regardless of origin, fatty acids must pass through the same biochemical gateway before they can be used for energy production
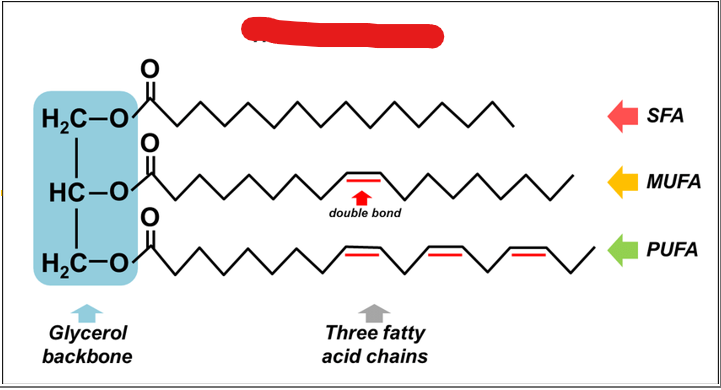
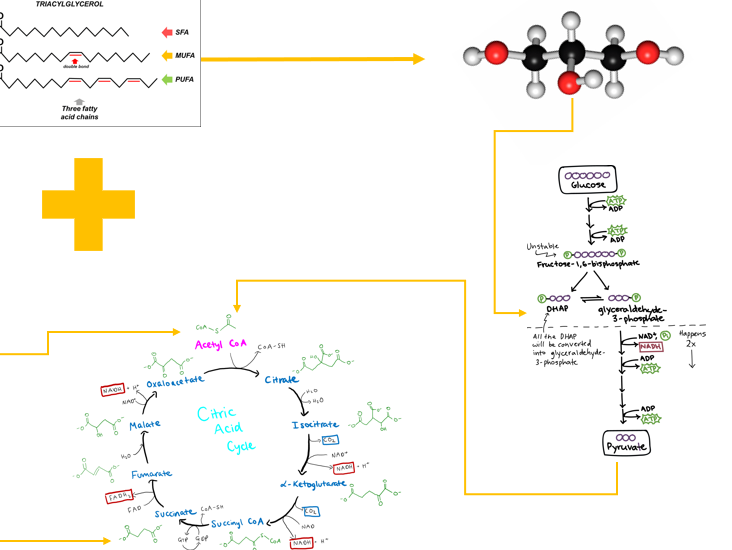
What are the primary storage form of lipids in the body and are uniquely suited for long term energy storage due to their highly reduced chemical structure
triacylglycerols
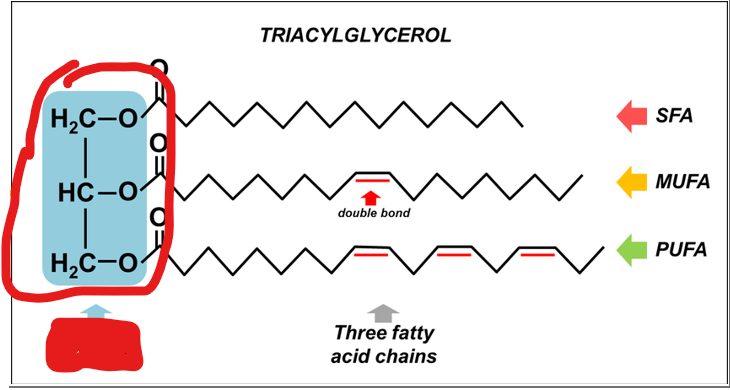
What must each traicyl glyceral molecule consist of
a glycerol backbone
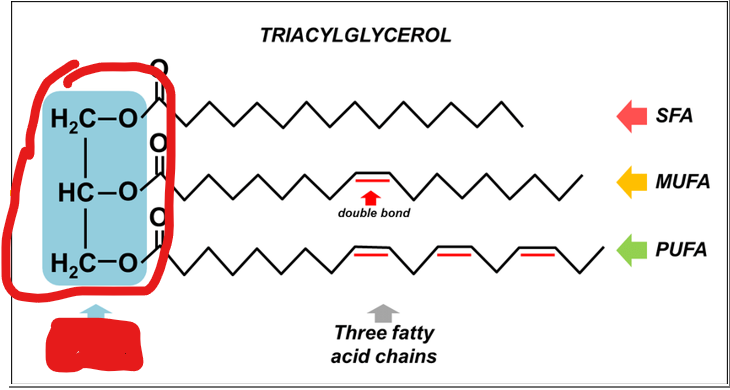
what makes up a glycerol backbone
a three-carbon alcohol, esterified to three fatty acid chains via ester bonds formed between the hydroxyl groups of glycerol and the carboxyl groups of fatty acids
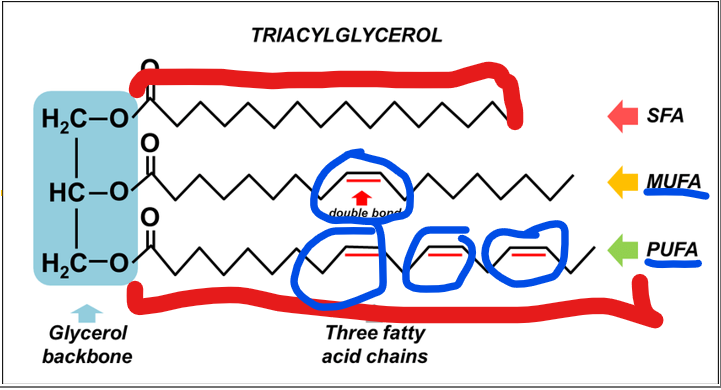
Fatty acids in a triacylglycerol can vary in length and saturation. What does this mean?
they may be saturated (no double bonds), monounsaturated, or polyunsaturated
Where does the key biochemical significance of triacylglycerol lie
in their high energy density
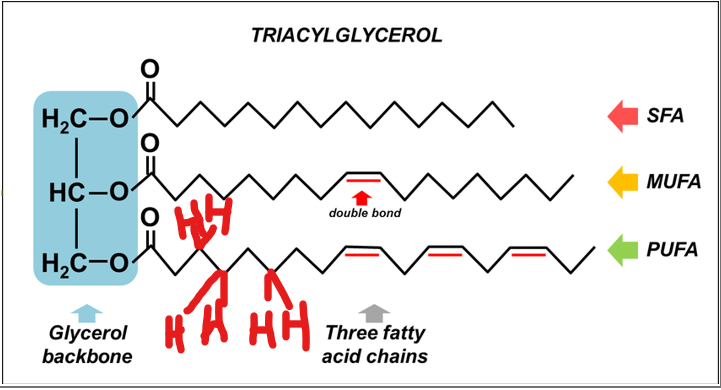
The key biochemical significance of triacylglycerols lies in their high energy density.
What is the explanation behind this?
Because their carbons are in a highly reduced state—meaning they contain many C-H bonds and few C-O bonds—they yield significantly more energy upon oxidation compared to carbohydrates like glucose
The key biochemical significance of triacylglycerols lies in their high energy density. Because their carbons are in a highly reduced state—meaning they contain many C-H bonds and few C-O bonds—they yield significantly more energy upon oxidation compared to carbohydrates like glucose.
What does this clarify in terms of fats?
this is why fats provide roughly twice as much energy per gram as carbohydrates
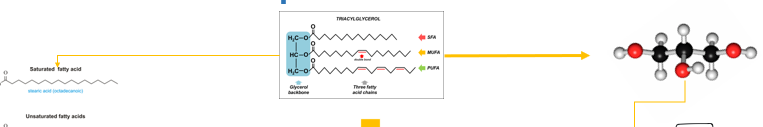
What do triacylglycerols yield when they are broken down
free fatty acids and glycerol
When triacylglycerols are broken down, they yield free fatty acids and glycerol
What happens to each?
they each enter different metabolic pathways
When triacylglycerols are broken down, they yield free fatty acids and glycerol, each of which enters different metabolic pathways.
Where are the fatty acids directed?
towards oxidation for energy
When triacylglycerols are broken down, they yield free fatty acids and glycerol, each of which enters different metabolic pathways.
Where is the glycerol directed
it serves as the bridge between lipid and carbohydrate metabolism
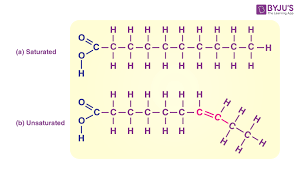
Why are lipids such a powerful energy source ?
In fatty acids, carbons are highly reduced, meaning they are bonded primarily to hydrogen rather than oxygen. In contrast, carbons in glucose are partially oxidized due to their association with hydroxyl groups
Whhat happens when fatty acids undergo oxidation?
electrons are stripped from these reduced carbons and transferred to electron carriers such as NAD⁺ and FAD, forming NADH and FADH₂
When fatty acids undergo oxidation, electrons are stripped from these reduced carbons and transferred to electron carriers such as NAD⁺ and FAD, forming NADH and FADH₂.
What do these carriers do?
then deliever high-energy electrons to the ETC, ultimately driving ATP production
Because fatty acids contain more reducible electrons per carbon than glucose, what does that mean for the result of their oxidation in comparison to carbohydrates
fatty acid oxidation produces significantly more ATP
Because fatty acids contain more reducible electrons per carbon than glucose, their oxidation produces significantly more ATP
What does this emphasize the importance of
why lipid metabolism is especially important during prolonged fasting, endurance exercise, or starvation—situations where efficient energy extraction is critical.
What is the process reponsible for lipid breakdown
beta oxidation
what is beta oxidation
it systematically removes 2 carbon units from fatty acids in the form of acetyl-CoA, feeding directly into the citric acid cycle
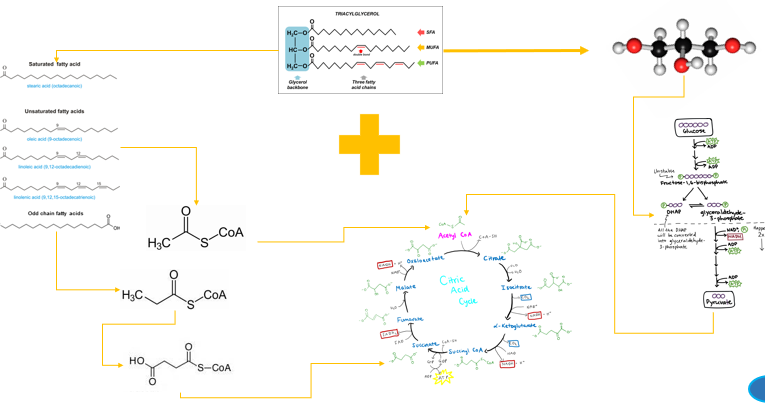
An important biochemical distinction in lipid metabolism is whether a fatty acid has an even or odd number of carbons
What does this determine?
the final products of beta-oxidation.
Most naturally occur have how many carbons
an even number of carbons
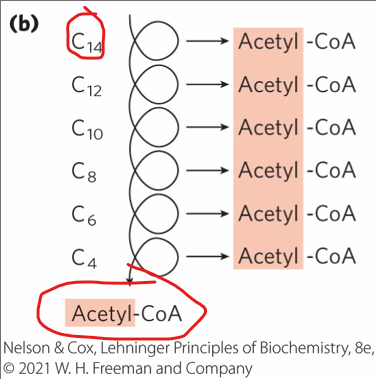
Most naturally occurring fatty acids have an even number of carbons.
what does their complete breakdown yield?
ONLY acetyl-CoA - a two carbon molecule that enters the TCA cycle
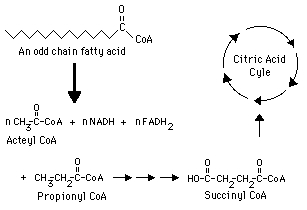
How do odd chain fatty acids behave differently in lipid metabolism
While they also undergo repeated cycles of beta-oxidation, their final breakdown step produces one molecule of propionyl-CoA, a three-carbon compound, in addition to acetyl-CoA units
What is important to know about propionyl-CoA in terms of continuing fatty acid metabolism in which the acetyl-CoA can enter the TCA cycle
Propionyl-CoA cannot directly enter the citric acid cycle
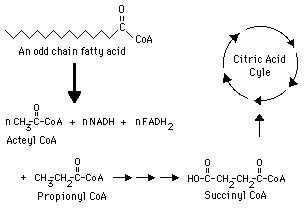
Propionyl-CoA cannot directly enter the citric acid cycle, what must occur for it to enter the TCA cycle
it must first be converted into succinyl-CoA, an intermediate of the cycle
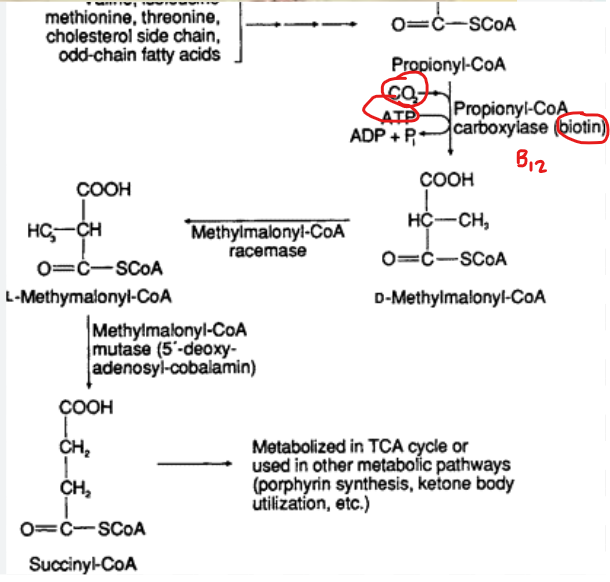
What does the conversion of propionyl-CoA to succinyl-Coa require
several enzymatic steps and cofactors- mainly biotin and B12
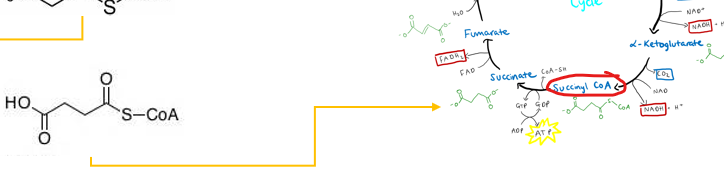
What impact does the ability of succinyl-CoA entering the TCA cycle have inregards to odd chain fatty acids
it makes odd chain fatty acids more metabolically versatile as they can contribute to gluconeogenesis, unlike acetyl-CoA, which cannot be used to generate glucose-
this is done by Succinyl-CoA contributes to gluconeogenesis by acting as an anaplerotic substrate that replenishes TCA cycle intermediates, specifically increasing the pool of oxaloacetate
The distinction of odd chain fatty acids providing succnyl-CoA to contribute to gluconeogensis compared to acetyl-CoA which cant be used to generate glucose becomes especially impotant during: ________
prolonged fasting where maintaing blood glucose levels is critical
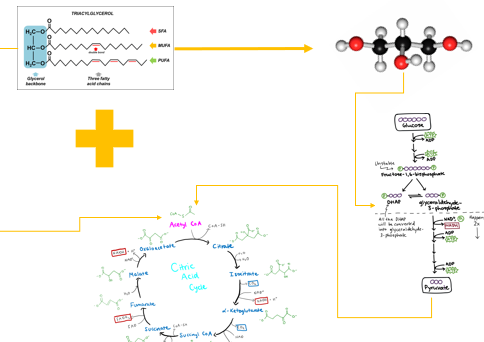
When triacylglycerols are broken down, what happens to the glycerol backbone
it is released alongside fatty acids and serves as a crucial connection point between lipid and carbohydrate metabolism
Glycerol itself is not directly useful for energy production. When does it become useful?
when it is converted into intermediates of glycolysis or gluconeogensis
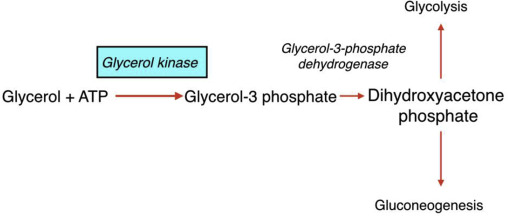
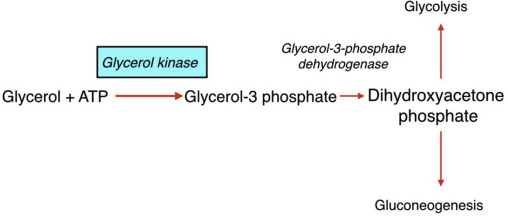
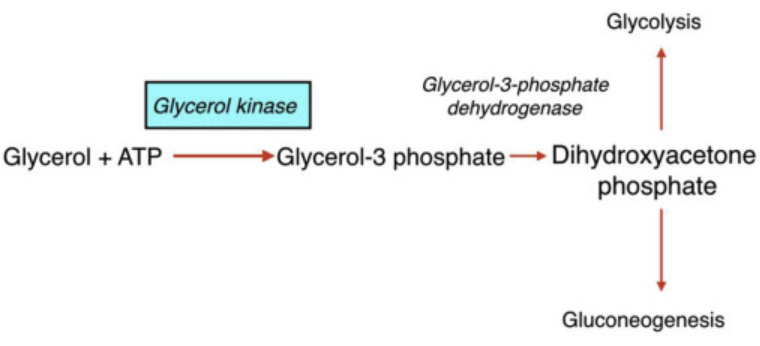
How does the conversion of glycerol begin
when glycerol kinase phosphorylates glycerol to form glycerol-3-phosphate
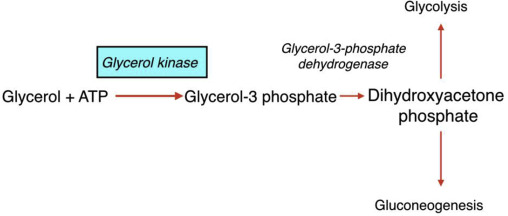
What happens to glycerol 3 phosphate
it is then oxidzed to dihydroxyacetone phosphate (DHAP)
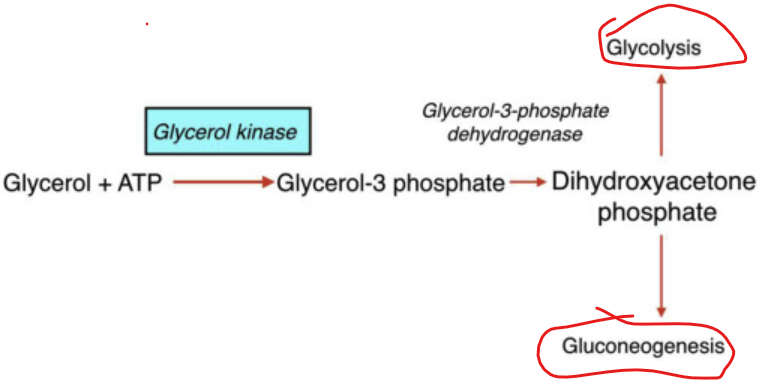
What is significant about glycerol 3 phosphate oxidized to dihydroxyacetone phosphate (DHAP)
DHAP is a key intermediate in glycolysis - from DHAP, the metabolic fate depends heavily on the tissue and physiological state
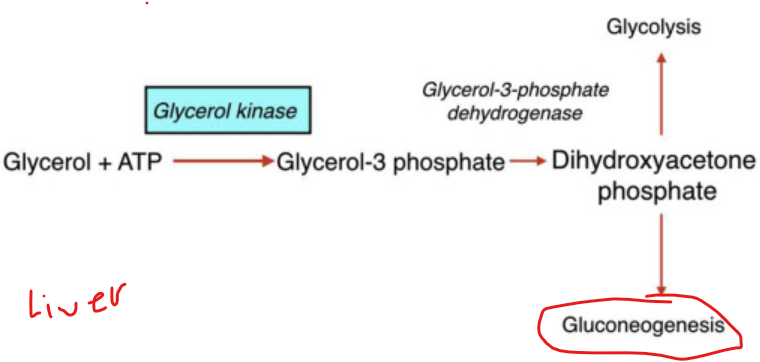
In the liver, what is dihydroxyacetone phosphate (DHAP) typically directed toward
toward gluconeogensis - especially during fasting to help maintain blood glucose levels
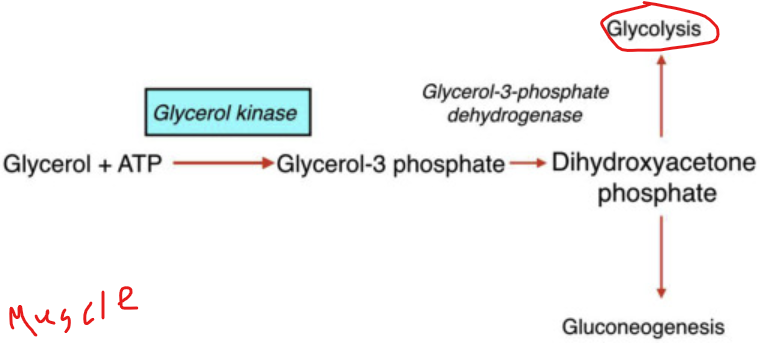
In the muscle tissue, what happens to DHAP
It i converted into glyceraldehyde-3-phosphate and enters glycolysis to produce ATP
Whay does the dual role of glycerol highlight
glycerol’s importance as a flexible metabolic intermediate that helps integrate lipid and carbohydrate pathways, ensuring that energy production is adapted to the body’s needs
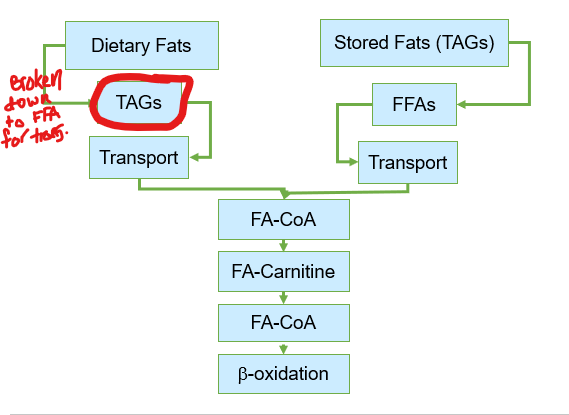
What must dietary lipids go through before they can be utlized by tissues?
undergo a complex process of digestion, absorption, and transport
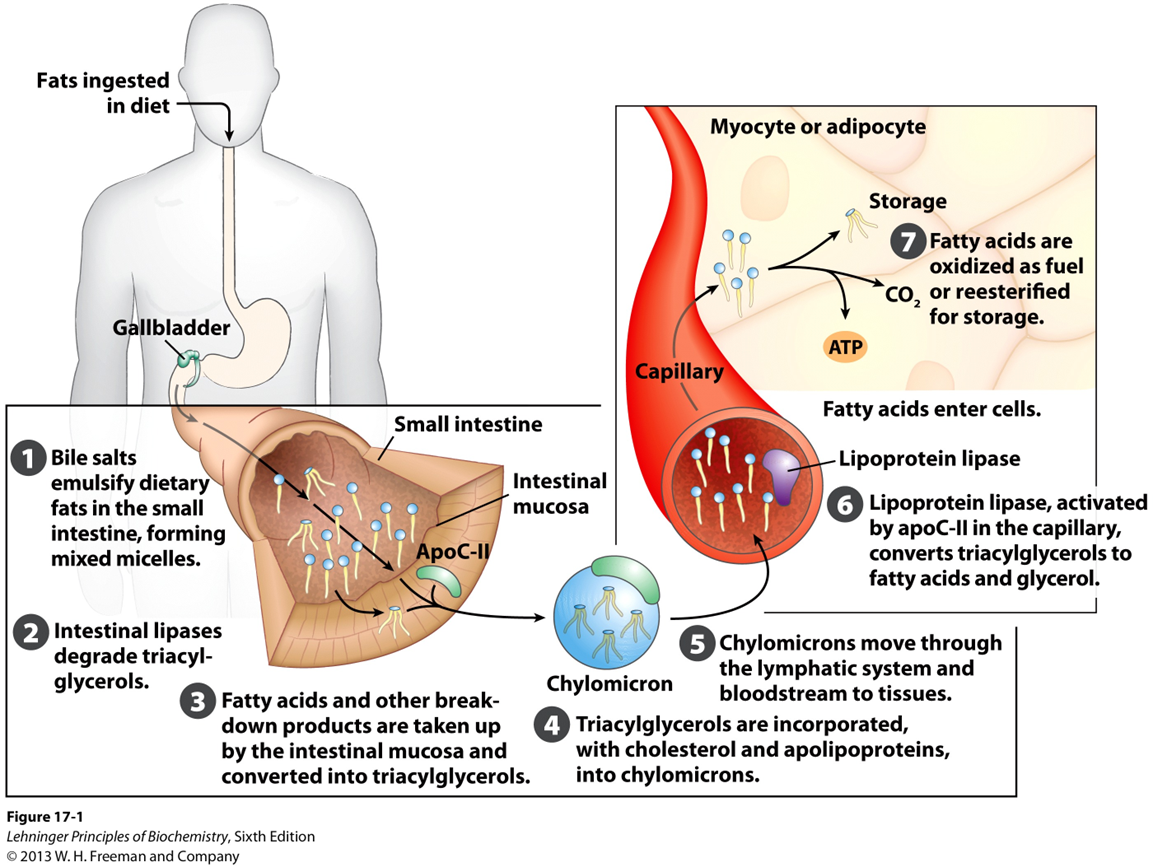
What happens first to dietary lipids
they must be emulsified and enzymatically digested
After emulsification and enzymatic digestion in the intestine, what happens to free fatty acids and monoacylglycerols
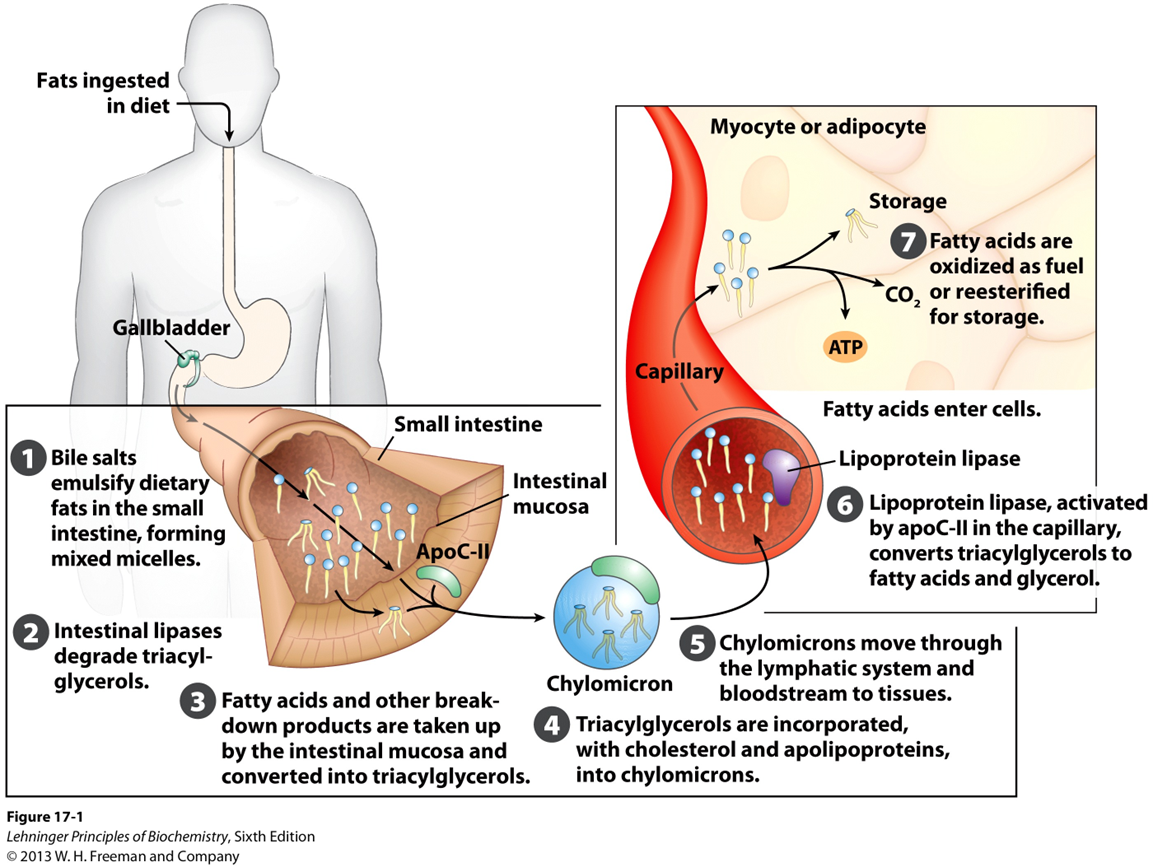
free fatty acids and monoacylglycerols are absorbed by intestinal cells (enterocytes)
After emulsification and enzymatic digestion in the intestine, free fatty acids and monoacylglycerols are absorbed by intestinal cells (enterocytes)
What is the problem with this?
These components cannot be released directly into the bloosdtream due to their hydrophobic nature
After emulsification and enzymatic digestion in the intestine, free fatty acids and monoacylglycerols are absorbed by intestinal cells (enterocytes). However, these components cannot be released directly into the bloodstream due to their hydrophobic nature.
What happens to solve this issue
they are re-esterified back into triacylglycerols within the enterocytes
Once free fatty acids and monoacylglycerold are re-esterified within the enterocytes, what happens next?
These newly synthesized triacylglycerols are then packaged into specialized transport particles called chylomicrons

What class of molecules do chylomicrons belong to
lipoprotiens
What makes up chylomicrons
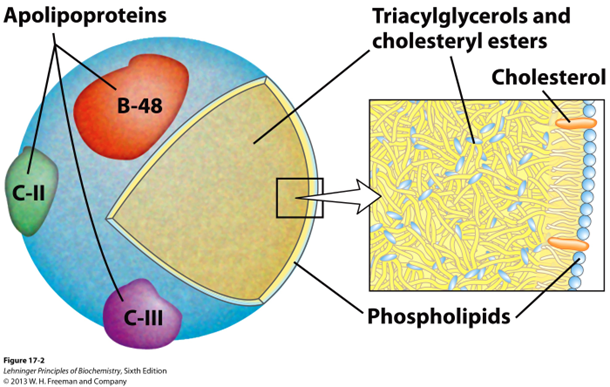
a unique structure consisting of a lipid core surrounded by a monolayer of phospholipids and embedded proteins called apolipoproteins.
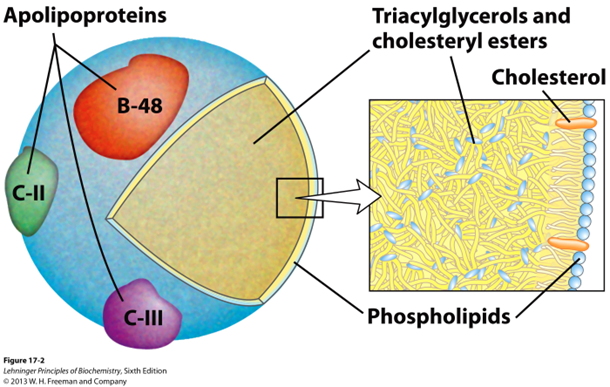
Why are apolipoproteins essential for chylomicrons
they are essential for directing chylomicrons to target tissue
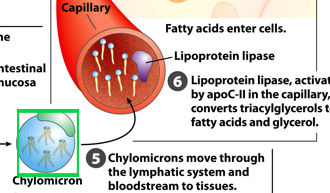
Once in circulation, what do chylomicrons interact with
lipoprotein lipase
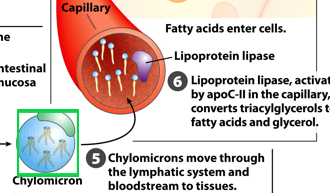
where is the enzyme lipoprotein lipase located
on capillary walls
What do lipoprotein lipases do
hydrolyzes the triacylglycerols into free fatty acids for uptake by tissues
In contrast to dietary lipid processing, the mobilization of stored fats occurs under what conditions?
energy demand, particularly during fasting
In contrast to dietary lipid processing, the mobilization of stored fats occurs under conditions of energy demand, particularly during fasting.
What is this process tightly regulated by
hromones
which hormone plays a central role in the mobilization of stored fats
glucagon
Study the stored hormone cycle:
When blood glucose levels drop, glucagon is released and binds to receptors on adipocytes (fat cells), initiating a signaling cascade involving G proteins, adenylate cyclase, and cyclic AMP (cAMP). This cascade activates protein kinase A (PKA), which phosphorylates key proteins involved in lipid mobilization. One of these proteins is perilipin, which normally acts as a protective barrier around lipid droplets. Upon phosphorylation, perilipin undergoes a conformational change that exposes the stored triacylglycerols to lipases. At the same time, PKA activates hormone-sensitive lipase (HSL) and facilitates the activity of adipose triglyceride lipase (ATGL). Together, these enzymes sequentially break down triacylglycerols into free fatty acids and glycerol
Okay!
Enzymes sequentially breaking down triacylglycerols into free fatty acids and glycerol is a process known as ____ and is essential for releasing stored energy in a controlled and regulated manner
lipolysis
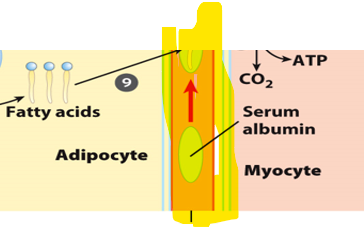
Once free fatty acids are released from adipocytes, they must be transported to target tissues.
Where do they get transported through
the bloodstream
Once free fatty acids are released from adipocytes, they must be transported through the bloodstream to target tissues
However, due to a particular reason , they cannot travel freely in the aqueous environment of blood.
What os this reason?
they are hydrophobic in nature
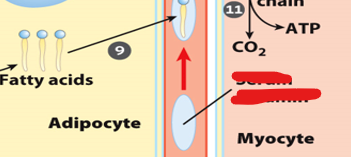
Once free fatty acids are released from adipocytes, they must be transported through the bloodstream to target tissues. However, due to their hydrophobic nature, they cannot travel freely in the aqueous environment of blood.
What do they do to mitigate this problem
they bind to carrier called serum albumin
what can serum albumin do
it can carry multiple fatty acids simultaneously
What does the binding of fatty acids to serum albumin allow for
it allows fatty acids to remain soluble and prevent them from forming harmful aggregates
How does albumin act as a shuttle
it delivers fatty acids to tissues such as muscle and liver, where they can be taken up ans used for energy production
Why is the serum albumin transport mechanism crucial
because it ensures that fatty acids are distributed efficiently throughout the body without disrupting the integrity of the bloodstream.
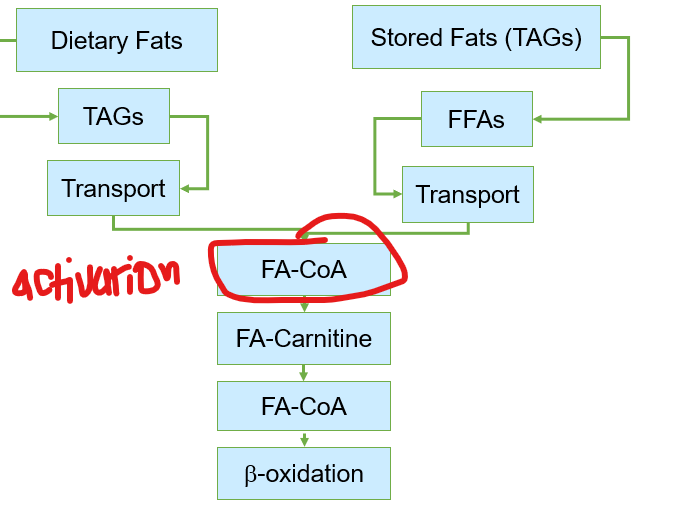
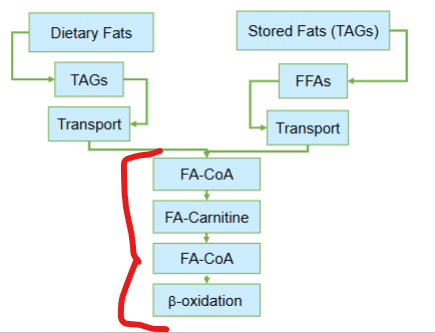
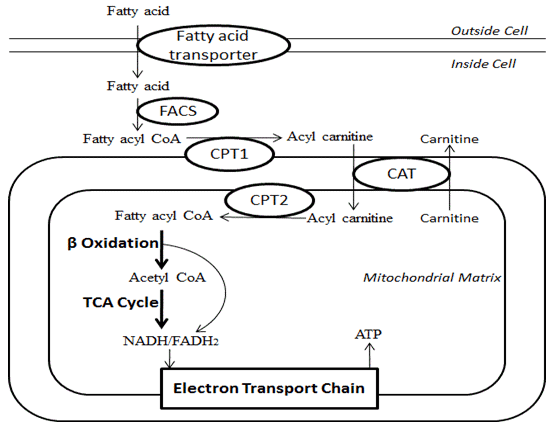
Before fatty acids can be oxidized for energy after transport, what must first occur
they must be activated
where are fatty acids activated before beta oxidation after being transported in the cell via serum albumin
the cytosol
what enzyme carries out fatty acid activation
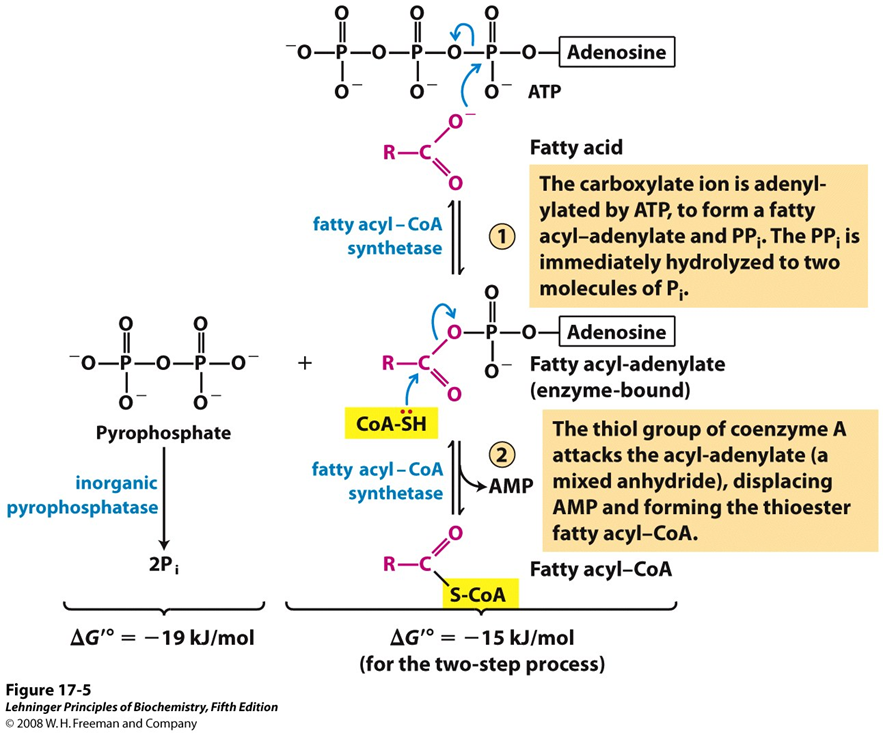
fatty acyl-CoA synthetase
what does fatty acyl-CoA synthetase do regarding fatty acid activation
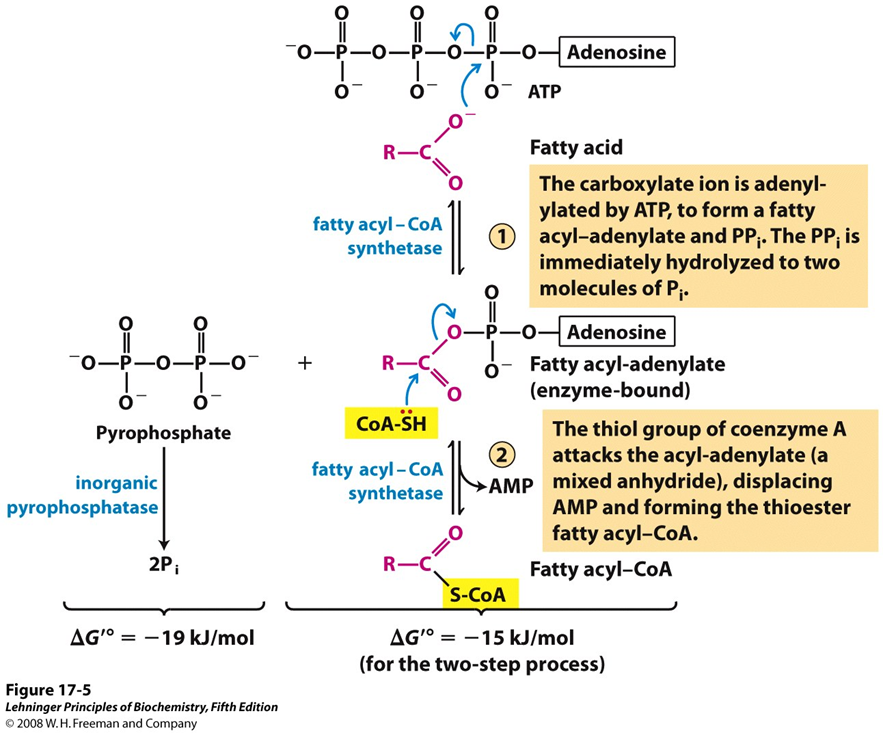
attaches coenzyme A (CoA) to the fatty acid via ATP , forming fatty acyl-CoA in the cytosol before getting transferred to mitochondria
What are the 2 purposes of activation of fatty acids
traps the fatty acid in the cell- prventing it from diffusing out
commits the fatty acid to degredation- signaling that it is destined for beta oxidation rather than storage
what is the activation of fatty acids analogous to
the phosphorylation of glucose to glucose-6-phosphate in carbohydrate metabolism—it represents a point of no return that directs the molecule into a specific metabolic pathway.
Although fatty acids are activated in the cytosol, where does beta-oxidation occur
the mitochondria
Although fatty acids are activated in the cytosol, beta-oxidation occurs in the mitochondria, creating a logistical challenge:
Fatty acyl-CoA cannot cross the mitochondrial membrane directly
What does the cell do to mitigate this?
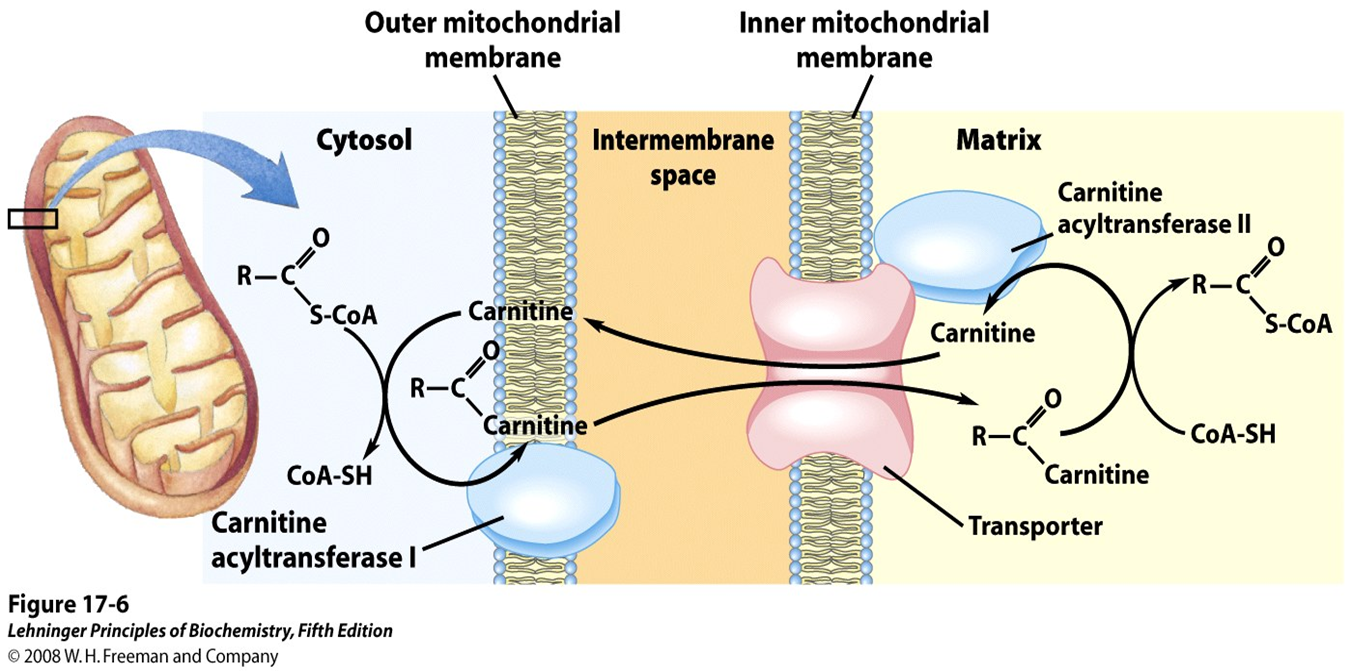
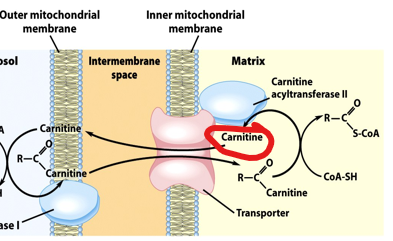
the cell uses a specialized transport system known as the carnitine shuttle
How does the carnitine shuttle work
the fatty acyl group is transferred from CoA to carnitine by the enzyme carnitine acyltransferase I (CPT1), forming fatty acyl-carnitine.
what enzyme transfers the fatty acyl group from CoA to carnitine to form acyl-carnitine
carnitine acyltransferase I (CPT1)
Fatty acyl-CoA cannot cross the mitochondrial membrane directly, so the cell uses a specialized transport system known as the carnitine shuttle.
In this system, the fatty acyl group is transferred from CoA to carnitine by the enzyme carnitine acyltransferase I (CPT1), forming ___________
This molecule can cross the mitochondrial membrane via a transporter.
What molecule can cross the mitochondrial membrane?
fatty acyl-carnitine
Fatty acyl-CoA cannot cross the mitochondrial membrane directly, so the cell uses a specialized transport system known as the carnitine shuttle
In this system, the fatty acyl group is transferred from CoA to carnitine by the enzyme carnitine acyltransferase I (CPT1), forming fatty acyl-carnitine.
This molecule can cross the mitochondrial membrane via a transporter.
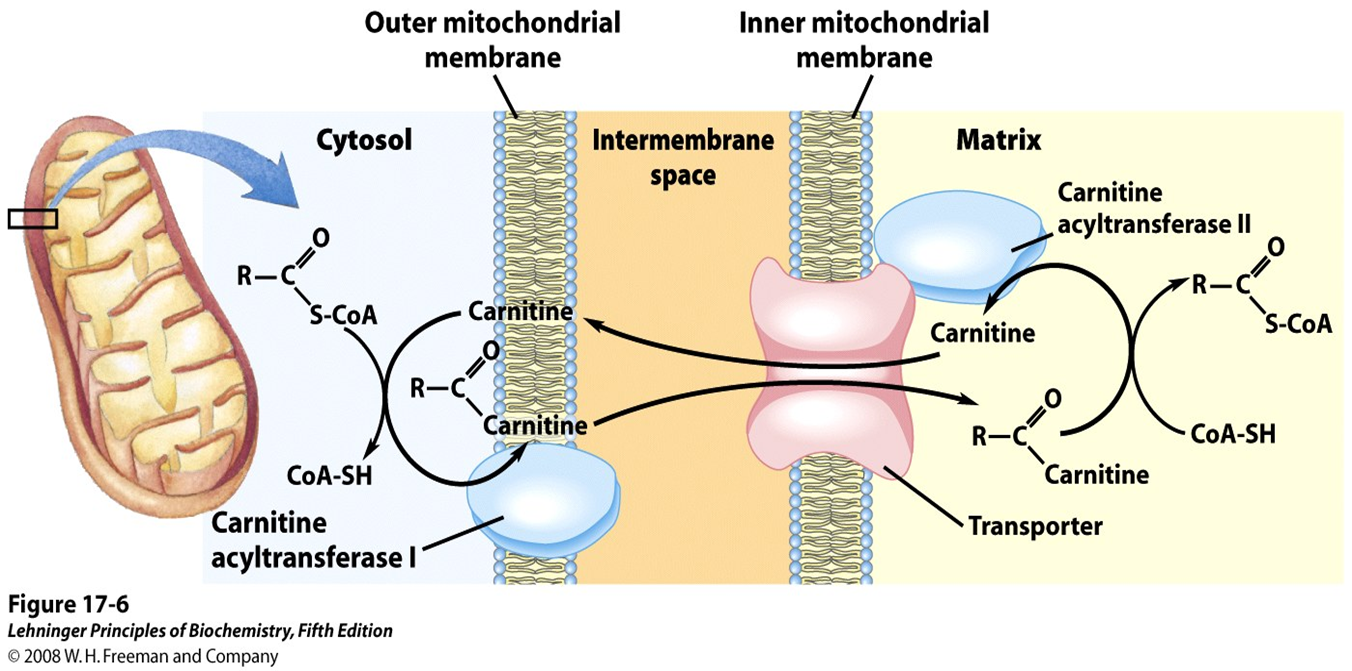
What happens to the acyl group once it gets inside the mitochondria
the acyl group is transferred back to CoA by CPT2,
what does CPT2 mean
carnitine acyltransferase II
What does carnitine acyltransferase II do?
takes the acyl group and transfers it back to CoA and regenerating fatty acyl-CoA in the mitochondrial matrix
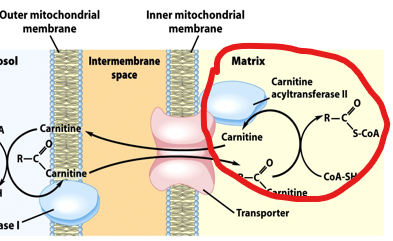
Fatty acyl-CoA cannot cross the mitochondrial membrane directly, so the cell uses a specialized transport system known as the carnitine shuttle
In this system, the fatty acyl group is transferred from CoA to carnitine by the enzyme carnitine acyltransferase I (CPT1), forming fatty acyl-carnitine.
This molecule can cross the mitochondrial membrane via a transporter.
Once inside, the acyl group is transferred back to CoA by CPT2, regenerating fatty acyl-CoA in the mitochondrial matrix.
What happens to carnitine?
it is recycled back into the cytosol
what is the carnitine shuttle system essential for
regulating fatty acid oxidation and ensuring that only properly activated fatty acids enter the mitochondria.
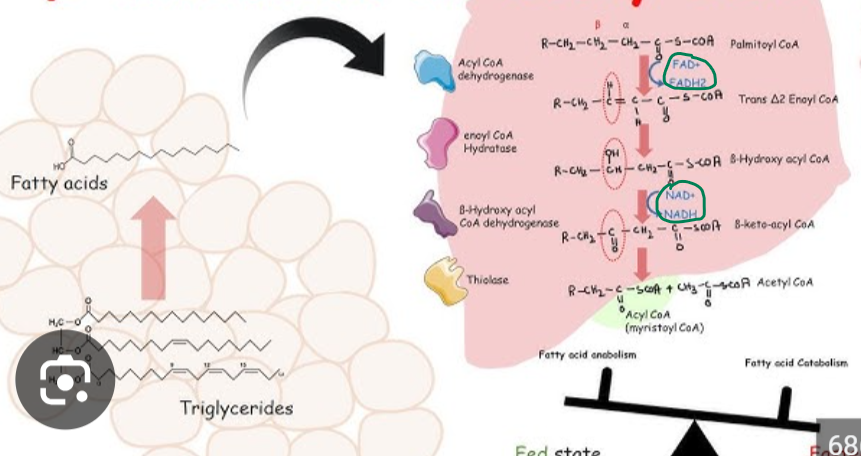
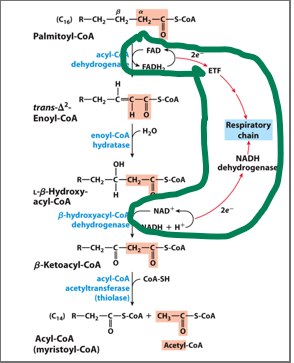
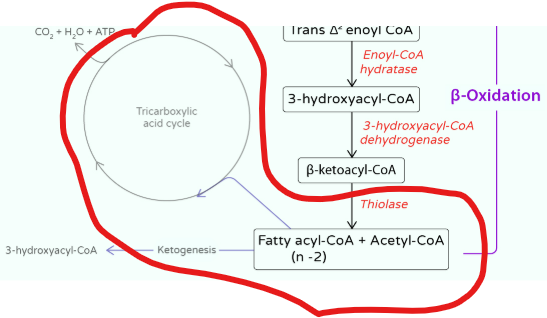
Beta-oxidation is the central pathway for fatty acid degradation and consists of a repeating cycle of four reactions.
What are the reactions in order?
oxidation
hydration
oxidation
clevage
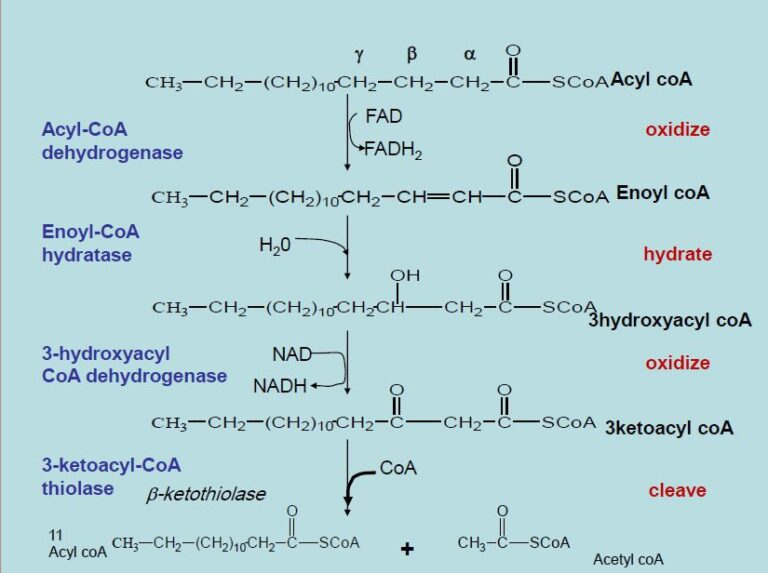
Beta-oxidation is the central pathway for fatty acid degradation and consists of a repeating cycle of four reactions: oxidation, hydration, oxidation, and cleavage.
What happens with each cycle
it shortens the fatty acid by 2 carbons, releasing one molecule of acetyl-CoA
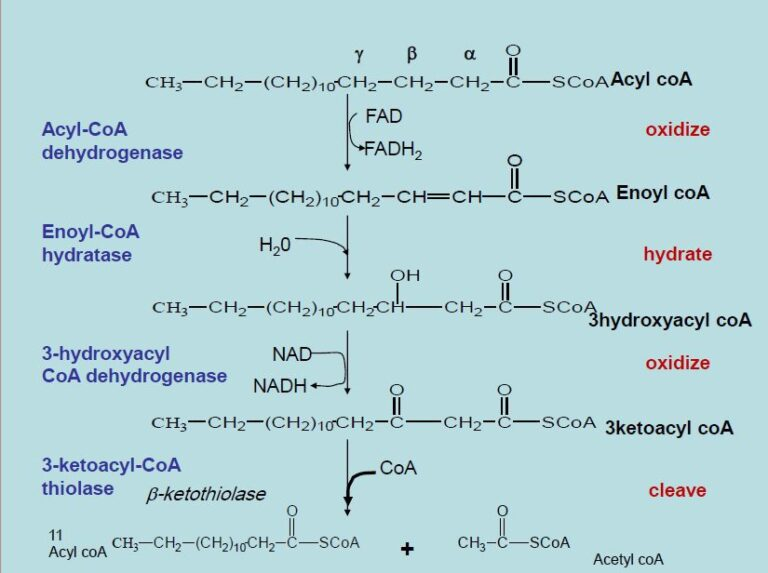
What happens during the first oxidation step of beta oxidation and what does it produce
forms a double bond and produces FADH₂
What does the second oxidation step produce
NADH
What do the electrson carried from the oxidation steps of beta oxidation have in relevence of generating ATP
These electron carriers feed into the electron transport chain, generating ATP.
What happens during the clevage step of beta oxidation
step releases acetyl-CoA, which enters the citric acid cycle for further energy production
How long does the beta oxidationrepeat
until the entire fatty acid is converted into acetyl-CoA units
True or False: Beta oxidation is energy efficient
True- it yields large amounts of ATP and makes fatty acids one of the most important long-term energy sources in the bod
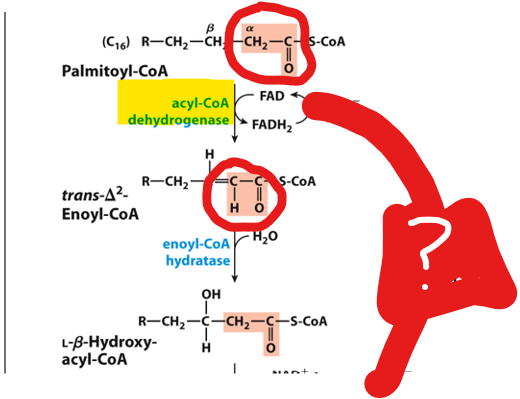
In the first oxidation step, acyl-CoA dehydrogenase, a flavoprotein that contains FAD, catalyzes the formation of a double bond between the alpha and beta carbons. This reaction reduces FAD to FADH₂,
What follows this?
FADH2 then donates electrons to the ETC via ubiquinone turning Q into QH2
In the first oxidation step, acyl-CoA dehydrogenase, a flavoprotein that contains FAD, catalyzes the formation of a double bond between the alpha and beta carbons. This reaction reduces FAD to FADH₂, which then donates electrons to the electron transport chain via ubiquinone (Q → QH₂)
What is this role analogous to?
glycerol 3-phosphate shuttle
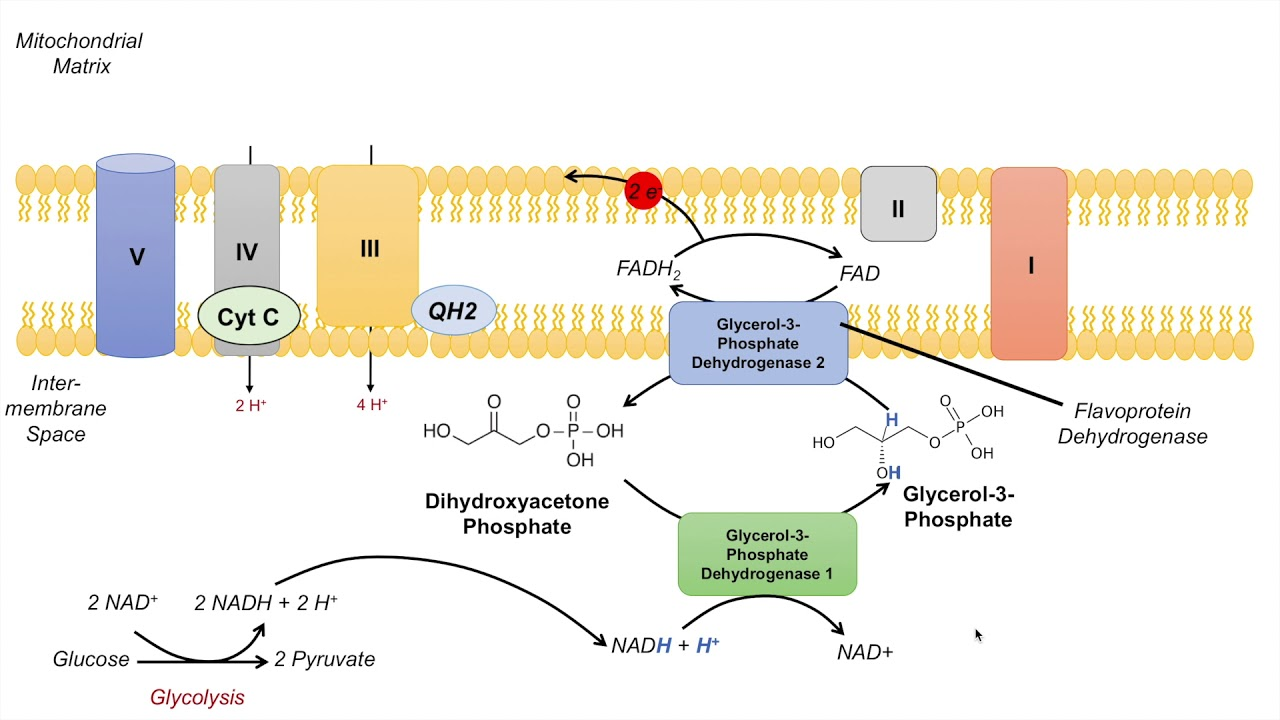
In the first oxidation step, acyl-CoA dehydrogenase, a flavoprotein that contains FAD, catalyzes the formation of a double bond between the alpha and beta carbons. This reaction reduces FAD to FADH₂, which then donates electrons to the electron transport chain via ubiquinone (Q → QH₂)
How is this analogous to the glycerol 3-phosphate shuttle
both processes use flavin adenine dinucleotide (FAD) to oxidize a substrate and directly transfer electrons to the mitochondrial ubiquinone (coenzyme Q) pool, bypassing Complex I of the electron transport chain
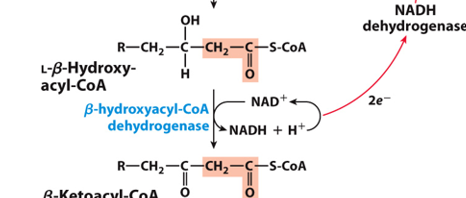
What happens to NAD+ in the secon oxidation step
NAD⁺ is reduced to NADH, generating another high-energy electron carrier
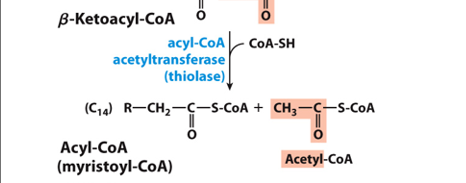
What doe sthe clevage step yield after the first , complete round of beta oxidation
releases an acetyl-CoA molecule and a shortened fatty acyl-CoA, ready to undergo additional cycles.
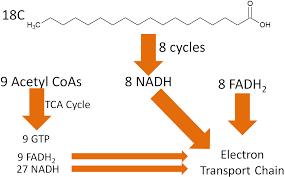
The cleavage step releases an acetyl-CoA molecule and a shortened fatty acyl-CoA, ready to undergo additional cycles. For example, a 16-carbon fatty acid undergoes 7 rounds of beta-oxidation, producing 8 acetyl-CoA, 7 NADH, and 7 FADH₂
Where can the acetyl-CoA molecules go?
they feed into the TCA cycle, generating further ATP
The cleavage step releases an acetyl-CoA molecule and a shortened fatty acyl-CoA, ready to undergo additional cycles. For example, a 16-carbon fatty acid undergoes 7 rounds of beta-oxidation, producing 8 acetyl-CoA, 7 NADH, and 7 FADH₂
Where can the NADH and FADH2 go?
drive oxidative phosphorylation.
the cleavage step releases an acetyl-CoA molecule and a shortened fatty acyl-CoA, ready to undergo additional cycles.
For example, a 16-carbon fatty acid undergoes 7 rounds of beta-oxidation, producing 8 acetyl-CoA, 7 NADH, and 7 FADH₂.
These acetyl-CoA molecules can feed into the TCA cycle, generating further ATP, while the NADH and FADH₂ drive oxidative phosphorylation.
What does this explain
why fats are such an energy- dense fuel: each carbon is ultimately fully oxidized to CO2, generating a large amount of ATP