Cell Bio test #3
1/66
Earn XP
Description and Tags
Pray for me its getting worser and worserp
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
67 Terms
What are some of the primary function of cells
A. to move- they have skeletal and mollecular nanoscale motors
B. generate and consume energy
C. reproduce and use genome
D. synthesise traffic
What are some of the functions and locations of organellses in the cell (their structure i mean) (nucleolus, mitochondria, cjloroplast, ribosomes, lysosomes and peroxisomes, vacuoles endoplasmic reticulum
Nucleolus
Structure and location:
largest organelle
Has a double layered membrane
Nucleolus are inside (produces the ribosomal DNA
Function:
Control center of the cell. Contains the DNA (builds ribosomes)
Q? How is the nucleus organized
Into nuclear bodies (areas with specific functions)
Mitochondria
STructure and location
Has two membranes. (inner membrane = cristae = increase surface area
Function: powerhouse of the cell (produces ATP
Chloroplasts
Structure and location: found in plant cells
Contains Thalakoids
Stroma surrounds the Thylakoids that carry their own DNA
Function
Photosynthesis
Riboomes
STructure
Small comple of ribosomal RNA and proteins
Functions
Covnerts mRNA into proteins
*** Lysosomes and perodiomes
Structure and locations
Samll membrane bound sacs filled with enzymes
Lysosomes: (works in acidic conditions = contains hydraulic enzymes)
Function: Breaks down old cellular components, pathogens, or large unneeded molecules
Uses a protein pump to keep high H+ conditions (if bursting will not activate the basic cell)
Peroxiomes: Iholds oxidative enzymes that contains catalysts
Function: helps detoxify harmful sbustances and metabolize fatty acids
Vacuoles
Structure and location:
Animals have many small ones, plants have one large one in each cell
Function:
Stores water and nutrients for the cell
Endoplasmic reticulum: series of interconnected membranes.. Sacs and tubules that modifies proteins and synthesises lipids
Smooth ER:
Structure- does not have surface ribosomes
Purpose
Synth
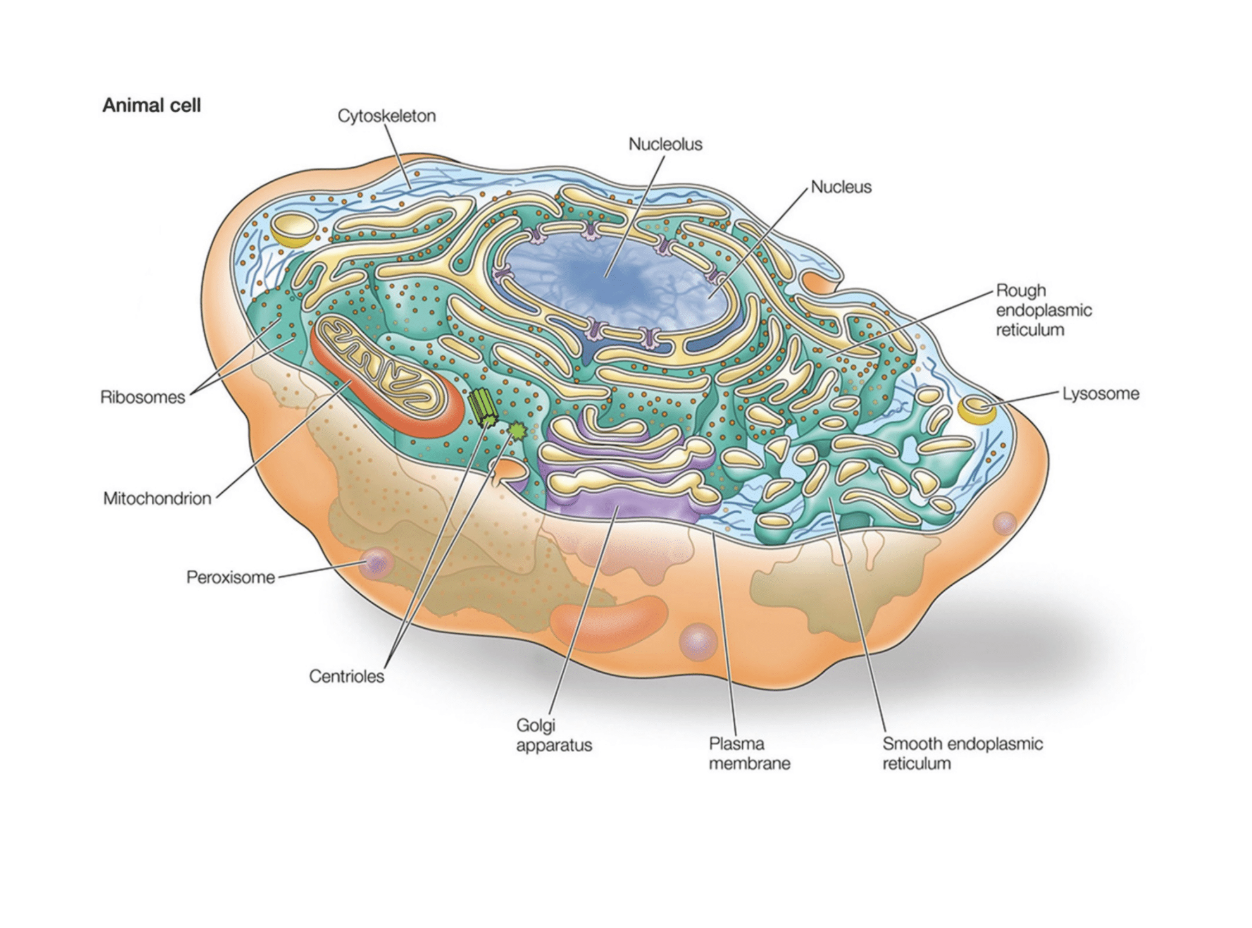
esis lipids
Detoxifies drugs and poisons
Stores caclium

What are the different structure sin the Endoplasmic reticulum and waht is it?
Endoplasmic reticulum: series of interconnected membranes.. Sacs and tubules that modifies proteins and synthesises lipids
Smooth ER:
Structure- does not have surface ribosomes
Purpose
Synthesis lipids
Detoxifies drugs and poisons
Stores caclium
Rough ER
Strucutre-
Network of membrane- bound sacks/tubles with ribosomes = looks rough
Purposes:
Syntesises proteins especially ones for secretion into membranes
Begins post-translational modification of proteins
What is the function of the golgi apparatus and what is its structure
Structure:
Stacks of flattened membrane bound stacs
Cis faced (receiving)- Closer to teh ER
, Trans face(shipping) - farther from the ER
Function:
Modifies, sorts, and packages proteins and lipid
Dispatch Products via vesicles for secretin within the cell
What ar esome of the cytoskeletal elements and what do they do?
Actin filaments
Consist of two coiled strand made of actin. They are found just beineth the plasma membrane=
Function: to stiffen the membrane and used for cell movements
Intermediate filaments
Structure: made up of a wide range of proteins (long string like)
Function: structural support. Found just inside the nuclear envelope to stiffen up its structure and provide attachemtn sites for chromosomes
Microtubles
Logn hollow calendars
Made up of protein combinations of alpha and beta tubulin
Functinon: helps transport cell cargo
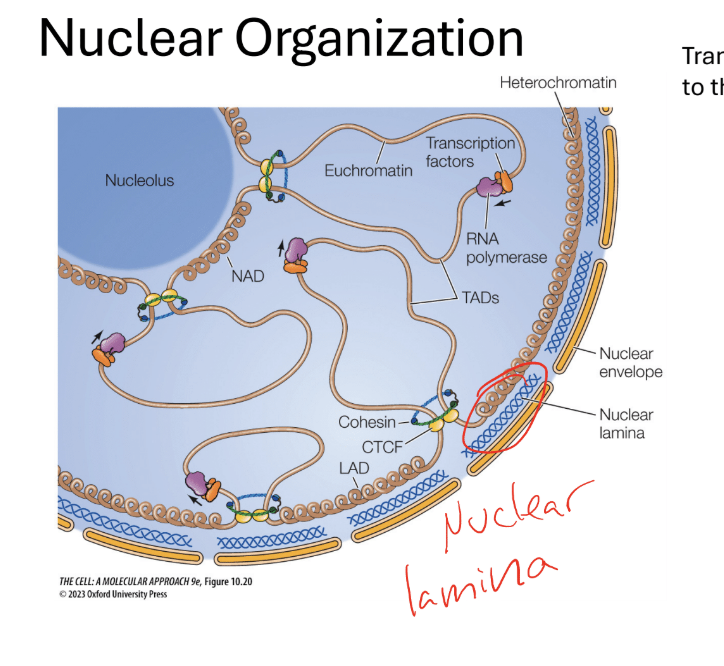
Descrie trends in nuclear organization? how do heterochromatin and euchromatin play into it?
THE NUCLEUS IS ORGANIZED INTO NUCLEAR BODIES WITH SPECIFIC FUNCTIONS
Ribosomes are assembled in the nucleolus
Transcriptionally active genes are localized to the interior of the nucleus
What are the different localizations of euchromatin and heterochromatin in the cell
Transcriptionally active genes are localized into the interior of the nucleus
Heterochromatin = associated with the nuclear envelope, periphery of the nucleolus (more transcriptionally active)
Q?What is the nuclear laminar
Made up of lamin protiins (blue windy structures in between the nuclear envelope and
membrane)- pushed against nuclear proteins
Q? What is the purposes of Heterochromatin protein 1 (HP1)
Binds to inactive chromatin- binds to Lamin B receptor (LBR), linking the heterochomatin to the nuclear envelope
What do you think might be somedisruptions/mutations that would affect nuclearorganization? What would the consequence be?
HP1, LBR could be mutated in ways that they are nonfunctional,
can’t bind to chromatin
•
LBR no longer inserts into nuclear membrane
•
This could affect location of chromatin in the nucleus, and
possibly disrupt levels of transcription
What is the endomembrane system and what is it used for? What is found in the system and discribe what they are
it is a grouop of organelles that helps modify, packag,e and transport lipids in the proteins?
nuclar envelope
lysosomes
vesicles
plasma membrane
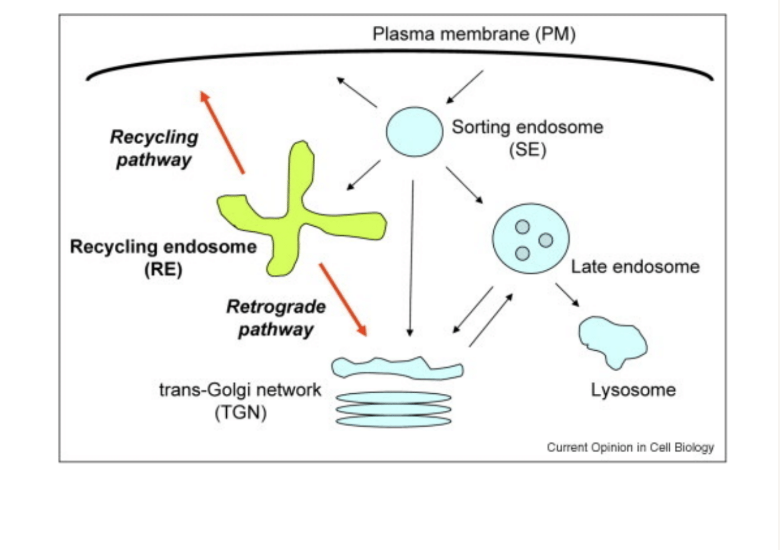
What are teh differnt types of endosomes and lysosomes and what do they do
Endosomes- used for sorting
endocytosis; when you take up molecule from otuside the cell in endocytic vesicle
Sorting endosome: sorts componnets to be recycled or degraded
Recycling endosome: brings membrane receptors back to the membrane
Late endosomes: fuse with vesicels carrying acid hydrolases
creates a lysosome
lysosomes- used to degrade intracellular materials
Can break down bological polymers
works at very high acidic conditions to ensure saftey just incase the structure bursts
uses proton pumps to keep concentrations high
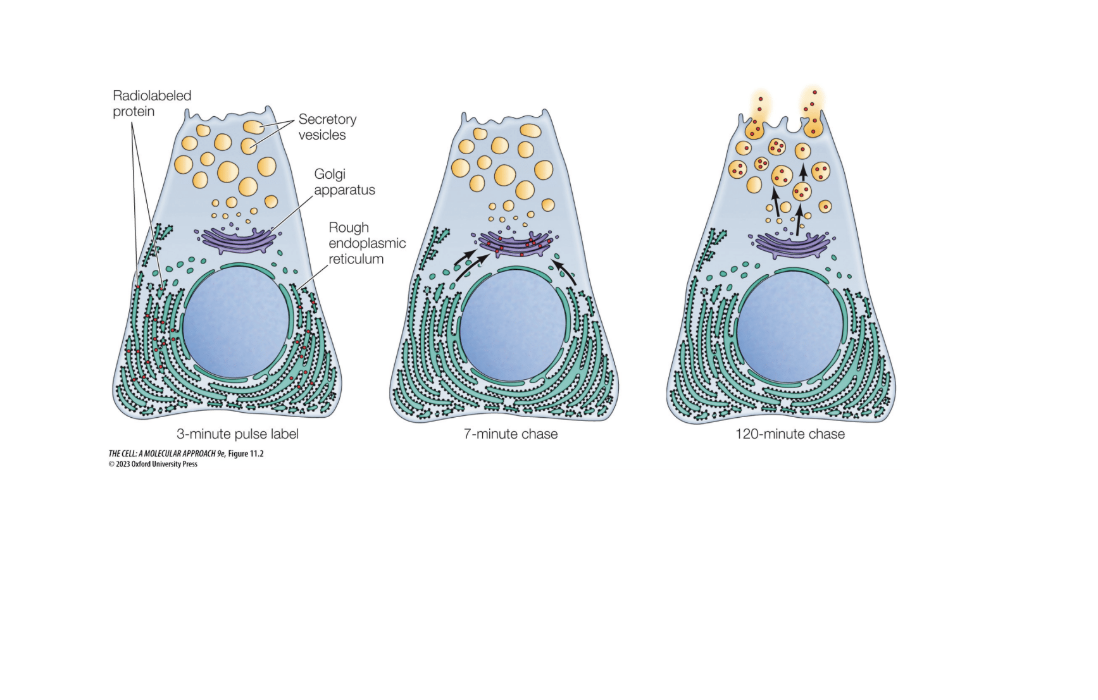
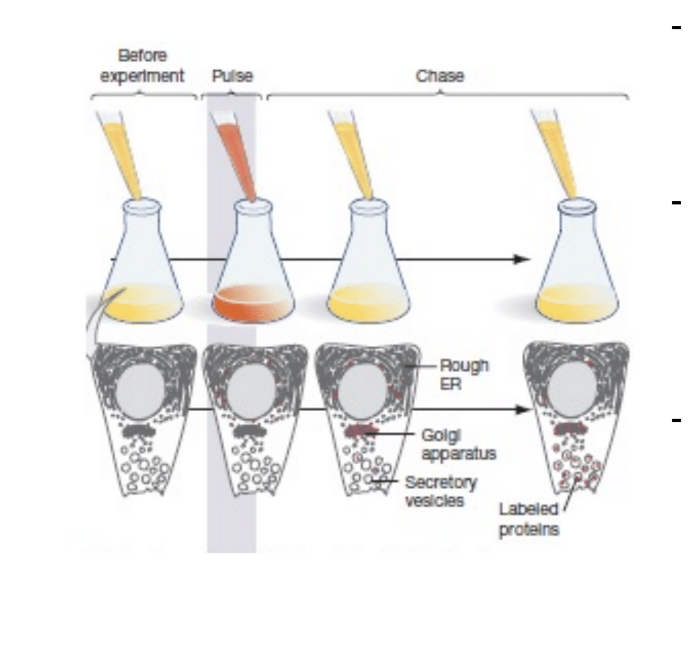
Describe the puls chase experiement and its effectiveness
When pancreatic cells make digestive enzymes into the small intestine
“Pulse” = short exposure to radioactive amino acids
Newly synthesized proteins were detectable since they were radioactive
Chase = incubated cells for various amounts o
f time then images (only lasts a short amount of time0
At around 120 minutes they leave the cell
How are proteins and lipids transported from the ER? descrube anteriograde and retro grade transport sequences and what receptors they use
Anterograde transport
Proteins and lipids transported out of the ER through (ER exit sites (ERES)
Retrograde transport
Uses the KDEL sequence
Receptors recognises the sequences and transports back to ER
Usually stuff that is important to keep in the ER
what wold be the results of the pulse chase expeiremnt look like if you 1. label a protein with a KDEL sequence and 2. lbe a protein that gets secreted from the cell
1a. you would go from the ER to golgi but back to the ER
2a. you would go from the er to secratory vesicles to be released ro the cell
how would you run a neceesary vs suffiencent expeirement for the sequence
deletions… just think about it
What are some of the mechanisms of protein transport? What are the 3 families of vesical coated proteins
COPI-coated vesicles
Bud form the ERGIC or golgi and returs proteisn to ealerer compartments (retrograde transport)
COPII- coated vesicels
carries proteins from the ERGIC to the golgi (cis fac
Clatherin-coated vesicles
transports in both directions (beteen the trans golig, endosomes, lyssomes, and plasma membrane) (works aroudn the cell
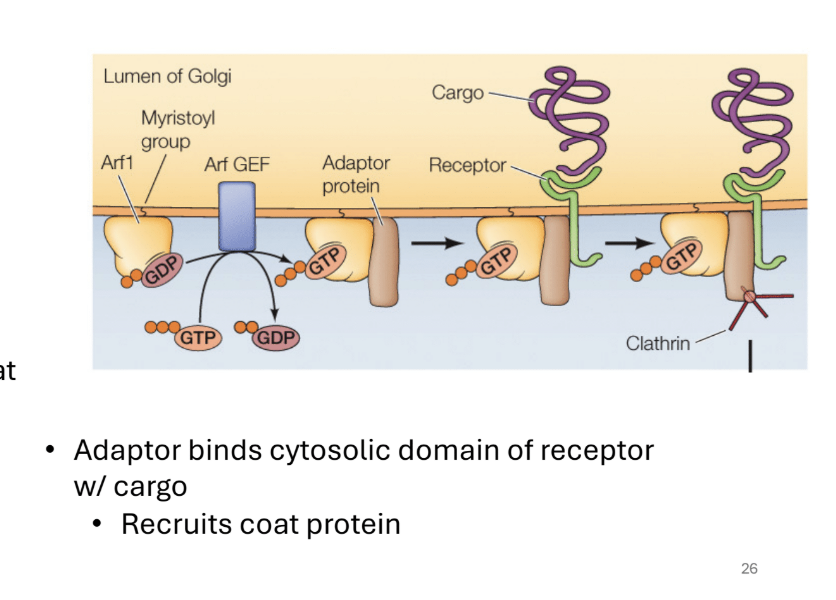
What are the GTP binding proteins and their functions
They help regulate the formation of coated vesicles
A. How>
gtp-proteins recruit adaptor proteins = interacts with the receptors bound to cargo proteins
the adaptor proteins bind = recruits the coat proteins
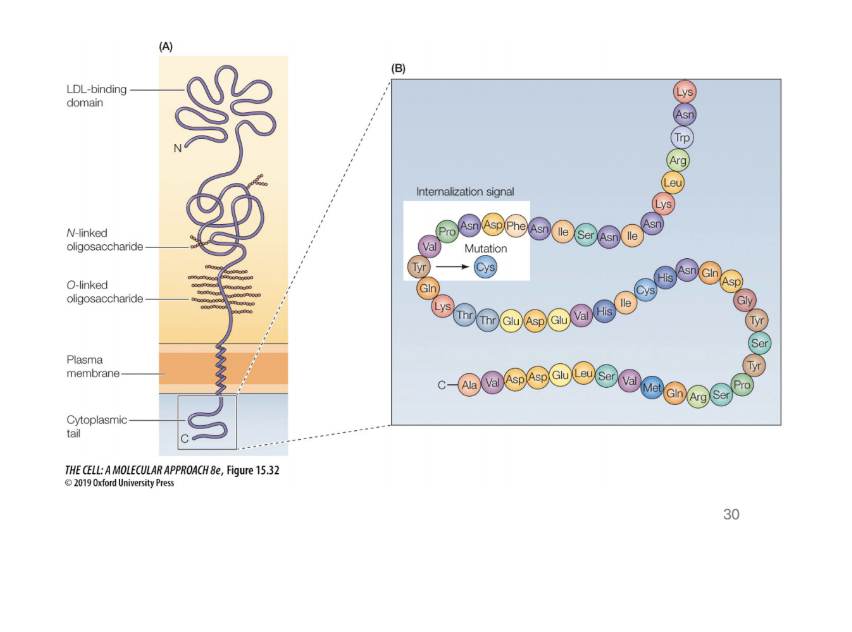
Describe the LDL example of protein transport
LDL receptor on cell membrane binds to LDL
Endocytose the LDL (brings it into the cell)
LDL is released in the EArly endosomes
The receptor is recycled
Lysosomes break down the LDL and releases its components
Where can you find mutations in the LDL receptors
Cyytoplasmic tails = used to associate with coat proteins. Mutations are tytpically found here
How does protein sorting work? (describe it and what can be found through it)? how does a protein/ibosome know if it should go to the ER or stay in the cytosol
How does it work? ther are ribosomes on the rough ER and in the cytoplasm
free ribosomes: proteins go to the nucleisu, mitochondria, and chloroplast
membrane boudn: golgi, plasma membrane, endoo and lysosomes, nucelar membrane, secratory vesicles
knows wehre to go based on signals made on the proteins
how do proteins from the ribosome know where to go based on where they were made
Immunoglobulin light chain proteins
Secreted from cells (found in the ER of membrane bound proteins)
Larger proteins are unable to pass through the ER since tey are signal sequences that are used to direct it to the ER
The signal is eventually cut out
How do you know the orientation of systolic free ribosomes
When translation first takes place, the fist proteins formed = (ER signal sequence- is recognized and bound to ER (HYDROPHOBIC) (N SEQUENCE)
How does protein sorting work>
Describe the steps for this mechanism
Signal is recognized By SRP receptor (FROM N TERMINUS FROM FIRST TRANSLATION)
What is there
Srp receptor is right next to the translocon
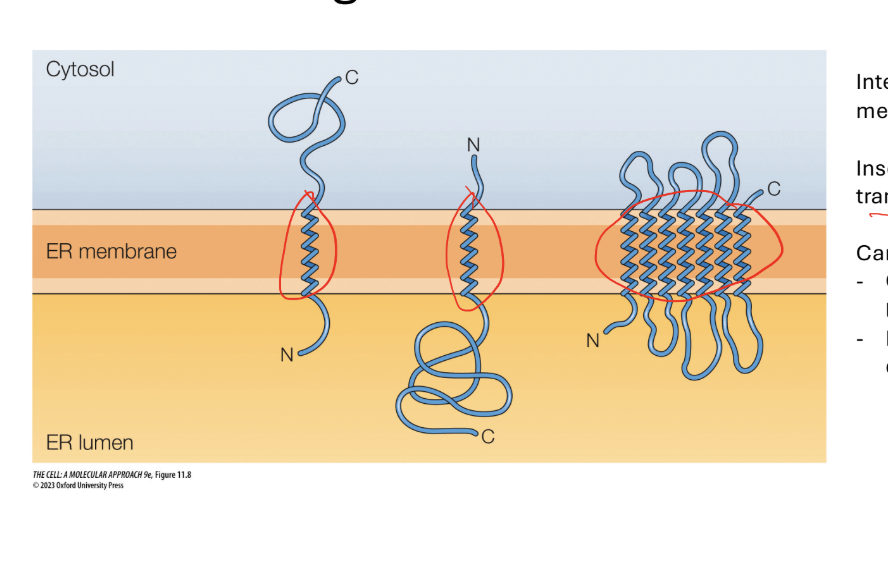
Integral membrane proteins are on the membrane via alpha helices (the spindley part in the membrane)
They are inserted into the membrane during the translational process ans the protein is being translate
Can have many different orientations
How do you remember the orientation of proteins during protien sorting
Steps and rules
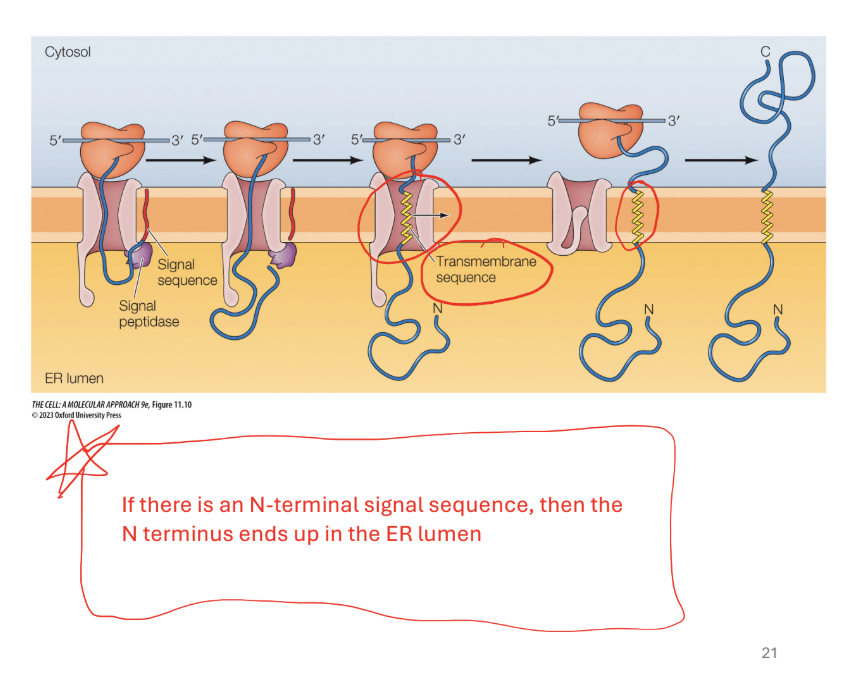
Step 1. Signal sequence is cleaved as poly peptide crosees the membrane N Terminus to the ER
Rule 1: N-terminal signal sequence (first protein form) = N terminus in the lumen
Step 2: if there is a transmembrane sequence (yellow alpha helix with a bunch of hydrophobic amino acids
Translocation halted when translocon recognizes trans membrane sequence
Results… shifts protein over and moves it to the plasma membrane (shifted to the right)
Step 3. Continue translation off of the membrane and forms the C terminus outside in the cytosol
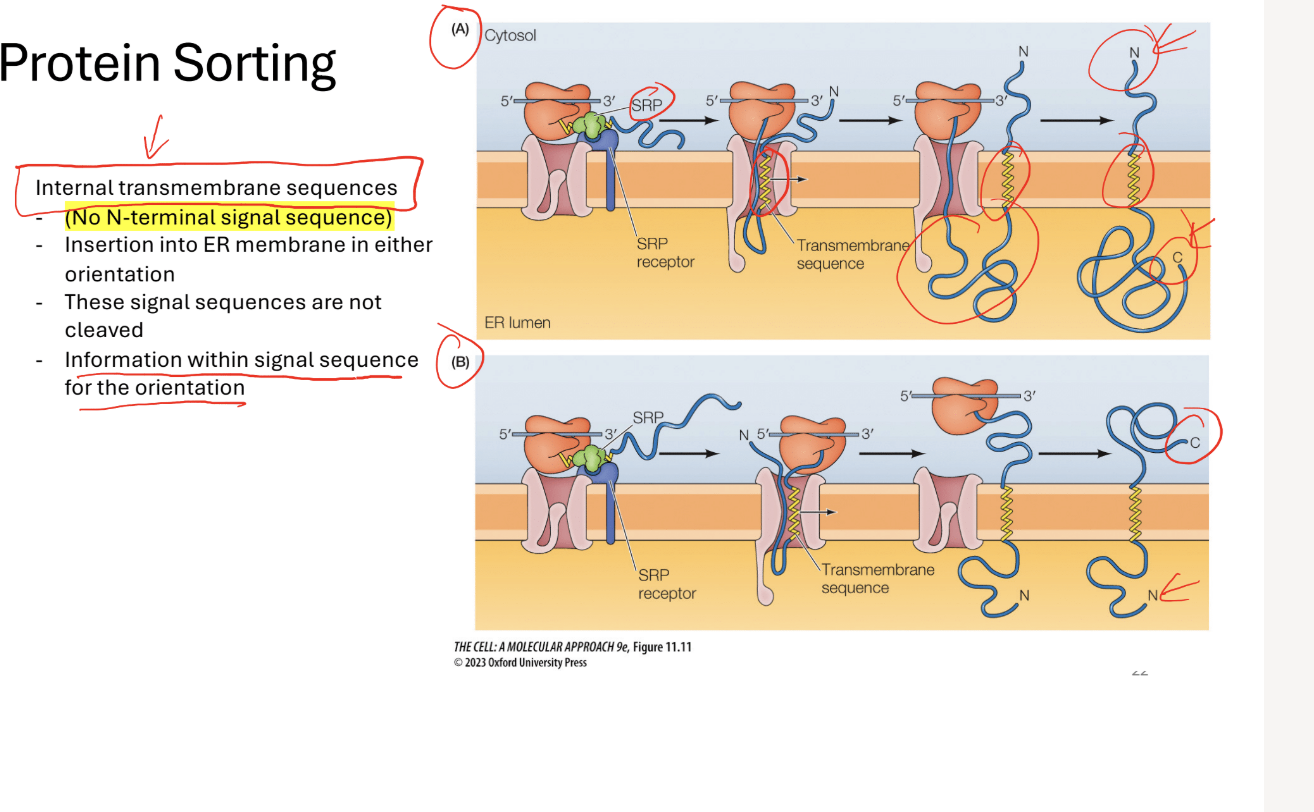
Q? What would you do with no N-Terminal Siganl sequence
Step 1: *uses internal trasnmembrane sequences)
Step 1: SRP recognizes transmembrane sequence alpha helix
Step 2: puts the alpha helix into the membrane
Translocon will move the alpha helix outt (n terminus on the outside in the cytosol)
Rest of the protein is made in the lumen
(can be oriented diffferenty with depending on the instructions of the alpha helix)
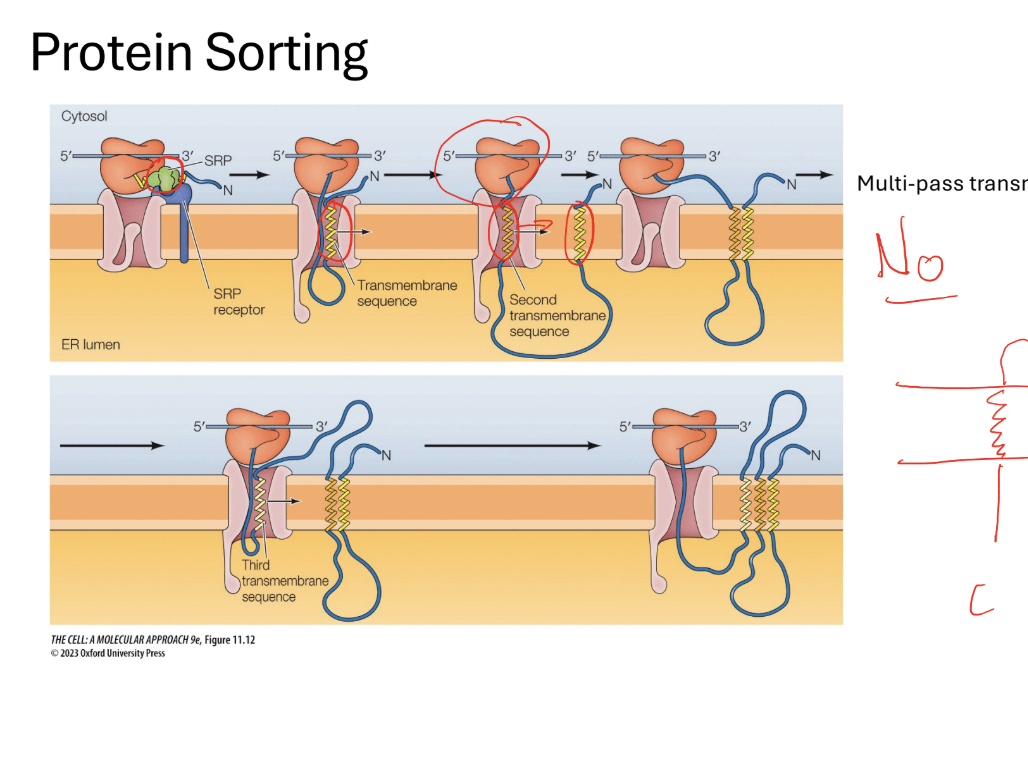
Q? What would you do with multi alpha helix proteins: (multi pass)
Steps 1: the same things happens again but restarts the sequence a bunch of times.
Note: to pass through a membrane you need an alpha helix (it will direct it in the opposite direction… like threading a needle
Protein sorting conceptual questions 1.
Consider a protein that is normally cytoplasmic. What
would happen if you fuse an N terminal signal sequence to it?
consider a transmembrane protein with the structure..
a: the protein would end up in the lumen of th ER
Consider a protein that is normally cytoplasmic What would happen if you fuse an N terminal signal sequence to it?
The protein would end up in the Lumen of the ER
Why?
Be careful of wording: if it is cytoplasmic it would be listed as such
. NOt transmembrane because it has no mention of an alpha helixIf it is cytoplasmic… (
HAS AN Er signal sequence complex
Consider a transmembrane protein with the structure. You fuse the ER signal sequence at the n terminus protein. Which diagram shows the position
Know that n terminus has to be in the ER lumen. Be careful with the transmembrane. Cannot cross membrane without the alpha helices
Try to maintain the same number of alpha helix before and after.
Keep on putting alpha helices from where the came from
What is the yeast UPR pathway? how would you diagram it? what triggers it?
it is triggered by a large sum of unfolded proteins in the ER
E stress signaling is charachtersed in the Saccharomues cerevisaie
goverened by the stress receptor (IRE1)
downstream transcrption factor HAC1 used
it helps upregulat ehte proteins to help deminish sthress to help proteins fold correctly
How does the UPR pathway play into necessary and sufficient shit
Do you think IRE1p is necessary
Yes? Why? It is the censor.. Without it you cant sense anythign
Do you think HAC1 is necessary
Yes. It would be necessary because it is a straight pathway
How would you design an experiment
You could create an experiment to get rid of those transcription factors. (deltion)
What about sufficiency in these components? Would you be able to test that
Would not be able to test since they are all necessary
What are teh steps to vertebrate UPR pathways
Unfolded proteins vind to the receptors (receptors bind to unfoled proteins)
pathways activated= transcription factors that increase the UPR target genes
first wave (very quick) second wave(activates tehe transcription factors)
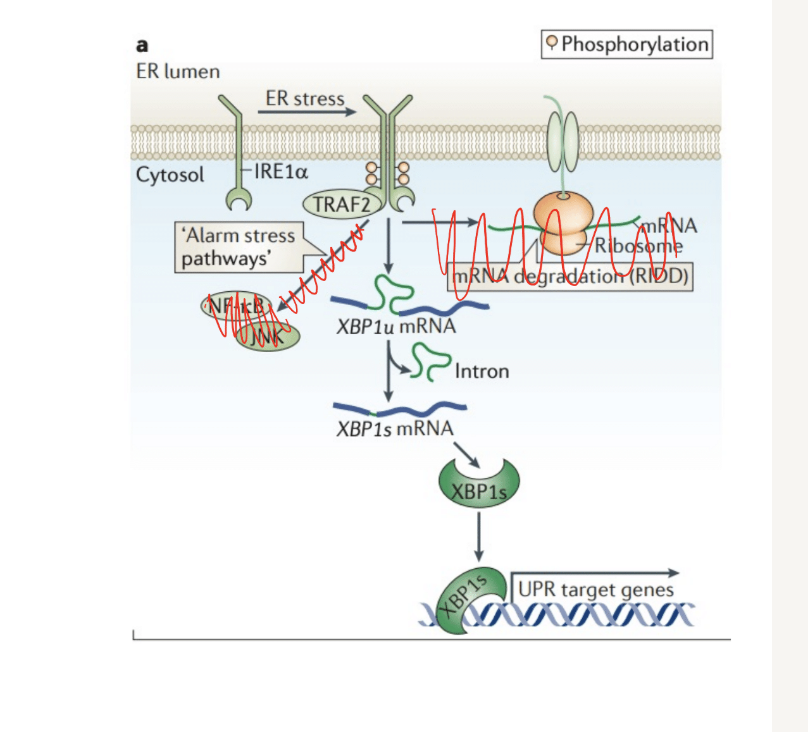
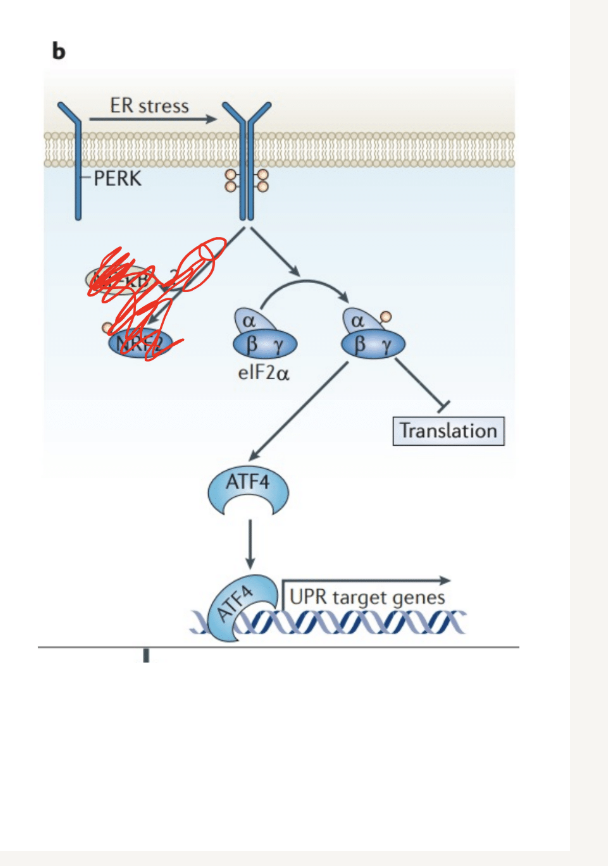
What happens during the first wave of vertibrate UPR pathways
Activation of PERK
Inhibits protein translation through the phosphorylation of eIF2a (be able to read off figure)
Alpha components gets phosphoylated and transcription is blocked
(PERK –> eIf2a)
Selective degradation of MRNA
Is initiated through regulated IRE-1 dependant decay (RIDD)
Q? What does this mean and where is it shown on the figure
ER-located proteins = proteins that are made into the er
mRNA deredation (RIDD)
IRE1a - triggers the rIDD process
What nhappens during the second wave of resposnes during the UPR pathway? what are the 3 pathways to the second wave of UPR
All components of the first wave of responses go into the second wave = UPR target genes (proteins- helps decrease the number of unfolded proteins)
Q? What are the 3 pathways to the second wave of UPR (figures will be shown. Know how to interpret)
IRE1a
Ire1A (receptor). Dimerizes (brings two receptors together) and autotransphosphorliate to activate
Processes the mRNA for XBP1 allowing it to be translated into protein
Protein is a transcription factor- goes to the nucleus to upregulate UPR genes
PERK (protein kinase RNA-like ER Kinase)
Perkk Phsophorylates the eiF2a activated
Allows for selective translation of ATF4 ( eIF2a is an initiation factor involved in translation)
ATF4 is a transcription factor- increases expression of UPR tearet genes
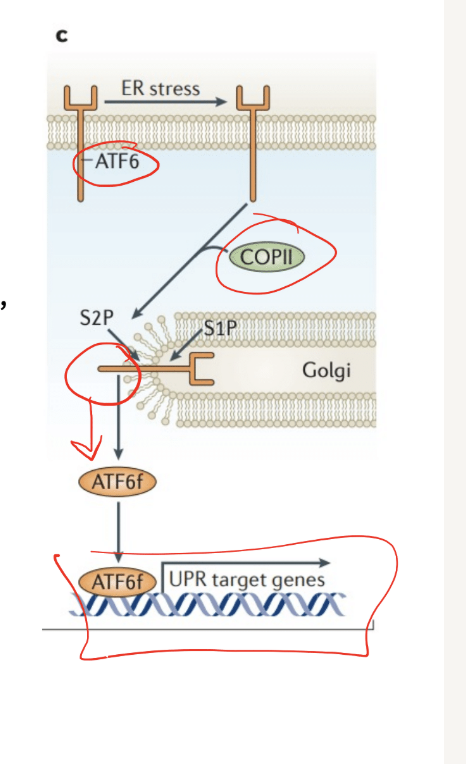
ATF6
ATF6 translocates to the golig after detecting unfolded proteins
Processed by proteases (break down proteins), = releases ATF6f
ATF6f= transcription factor= upregualtes UPR target genes
What would be expected outcome if a mutation prevented the phosphrylation of eIF2a in a vertebrate cell
reduced ability to regulate protein load in the ER, leading ot increased sress
What is and what are teh components of the nucelar envelope? what are the components and discription of the nuclear pore complexes
Nuclear envelope consists of
2 nuclear membranes (inner and outer)
Is continuous with the ER
Underlying nuclear lamina (mesh network on the edge of the inner membrane)
Provides structural support
Made up of proteins called lamins (associate with eachother
Nuclear pore complexes (description and componnetns)
Made up of many nucleoporins (proteins
Spans both the inner and outer membrane (nucleus to cytoplasm)
Rings on the cytoplasmic and nuclear sides
Protein filaments extend from either side
Central channel lined by proteins
Varrier to permeability, regulate transport
How are proteins targeted to the nucleus called and singled?
They are called nuclear localization signals (NLS) targeted by specific amino acid sequences)
What are the steps to nucelar import and export
Nuclear Import (part 1: to the left)
Need to get cargo protein into the nucleus
Cargo protein has (NLS)
NLS on protein recognized by importin
Cargo protein/importin complex bind to and pass through nuclear pore complexes
Ran-GTP binds to importan, relases protien in the nuclius
NI part 2- Importan back to cytop
Need to get important back to the cytoplasm
Importin/Ran-GTP complex binds and passes through nuclear pore complex
RAN GAP (attached to the nuclear pore complex) (GTPphase-activating protein)- hydrolyzes GTP to GDP
Importin is released into the cytoplasm
Now we have to bring the ran into the nucleus (start of nuclear import)
Nuclear Import (part 3)- RAN-GTP back to the nucleus
Ran-GDP binds to NTF2 (import receptor)
Need to make it RAN GTP
RAN-GEF (bound to chromatin) exchanges GDP to GTP
Nuclear export (uses NES- nuclear export singnal)
Ran-GTP, exportin, and cargo protein forms a complex
Bind and pass through the nuclear pore complexes
Ran GAP hydrolyzes GTP into GDP and causes release of the cargo (outside in the cytoplasm)
RAN GDP and exportin returned to the nucleus
ame as ran GDP for nuclear import (look at part 3 of nuclar import)
Exportin passes through on its own
RAN GEF exchagnges GDP for GTP
HOw does the transport of MRNAs work
mRNAs are transported by RAN-independent mechanism (no ram)
Associated wit about 20 proteins in the nucleus
Some of the proteins are part of an exporter complex, which exports the RNA out of the nucleus
Helicases on the cytoplasmic side remodels mRNA and removes exporter proteins
What are redox reactions and what are tehir trends
A. Reduxion is gaining. Transfer of electrons in association with the transfer of hydrogen ions
Oxidation is losing the electrons
You can always follow the path of the hydrogens
NAD+ to NADH
Oxidation= NAD+ to NADH
Reduction= NADH to NAD+
ATP and phosphrylation. What happens to the reaction energy when you phosphrylate a reactant
a. there is an increase in the free enrergy of the reaction
endergonic to exergonic
non-spontaneous to spontaneous (from needeing an addition of energy to not needing an addition to energY0
chasnges the conformation to make it more energetically favorable
What are all the componenets of the erngy cicycle/cellular respiration in the cell
Aerobic cellular respiration
glycolisis
citric acid/krebs cycle
oxidative phoshprilation
Aerioic cellular respiration
Glycolysis - Glucose (6c) → pyruvate (3c) - produces 2 net ATP (cytosol), NADH
pyruvate processing after
Goes into the cytric acid cycle in the mitochondria - creates 6CO2 and 2 atp, NADH and FADH2
NADH an dFADH2 go to oxidative phsopohrilation to create 34 atp (and h20)
What are the components of glycolysis
A. one glucose creates 2 pyruvate
B. 2 total atp is created (for created -2 that is consumed)
phorporilation of the glucose makes things more energyetically favorable Aerioic cellular respiration
Glycolysis - Glucose (6c) → pyruvate (3c) - produces 2 net ATP (cytosol), NADH
Goes into the cytric acid cycle in the mitochondria - creates 6CO2 and 2 atp, NADH and FADH2
NADH an dFADH2 go to oxidative phsopohrilation to create 34 atp (and h20)
What are the components of glycolyisi
One glucose creates 2 pyruvate
Two total atp is created (2atp in 4 atp out)
Phosphorylation making things energetically favorable
What is pyruvate processing + the citirc acid (krebs cycle) and what do they do
Pyruvate is oxidised and CO2 is formed
Acytl COa is created
Rest of the oxiation of carbon molecules
3nadh and 1 fadh formed
adp is turned into atp
Describe the steps and fuction of oxidative phosphorilation
Electron carriers will give up their electrons (NADH and FADH2). (oxidised)
Electrons will go through structure sin the proteins
Hydrogen pumps from the mitochondrial matrix to the intermembrane space
Matrex = low hydrogen
Intermembrane spacehas high concentration
ATP formation
Hdyrogens from the outer membrane goes through ATP syntahse and ADP is phoshorilated
What part. ofthe mitochondira is used for cellular repsiration
There is the outer membrane and the inner membrane.
Inner Membrane: where electron transport proteins go. Electron carrieres are attached ehr
Describe ATP synthase and how it forms ATP? What is it? What is the F1 complex? The F0 complex
ATP synthase = molecular machine in the inner mitochondrial membrane
F0 complex (membrane bound)
Works like proton channel = allows H+ ions to flow back into the mitochondrial matrix down their concentration gradient
Movement drives rotation of the enzyme
F1 complex (catalytic portion in the matrix)
Rotational movement in F0 is transmitted to conformational changes in F1 = atp production
Enzyme works in 3 steps (driven by mechanical movement)
Binding of ADP and Pi
Catalyzing the reaction to form ATP
Releasing ATP
Major difference between mitochondrial and chloro plast structures
ery sisilar but chloroplasts have thylakoids
Write out and describe the pathway of a protein designed for the mitochondrial matrix or the inner mitochondrial membrane
Proteins are fully made in the cytosol
Positive presequence recognized and brought to: the tom complex.
Repelled quickly through intermembreane space (lots of positive charges reel the presequence)
WHY? - because its positively charged and is repelled from the intermembran space
Through TIM complex
Into the matrix or inner membrane
If destined for the matrix = goes through instantly
Destined for the inner membrane: translocase recognizses if it is supposed to be in the inner membrane
MPP cleaves the presequence
Cuts the sequence (not fully formed protein met)
Write out the pathway of a protein Write out the pathway of a protein made from the mitochondrial genome and destined for the inner mitochondrial membraneand destined for the inner mitochondrial membrane
These are mainly made from the mitochondrial dna
Protein made in the matrix
Inserted into the membrane through Oxa 1 translocase (no positive charges0
Helper proteins help carry it through yellow
Write out the pathway of a protein destined for the outer membrane that has alpha helix or beta barrels
Peorwina with alpha helix go throug MIm1
Proteins with beta barrel first come through tom complex then through sam complex
Note” proteins that are made for outer mitochondrial membrane are made up of nuclear genome*
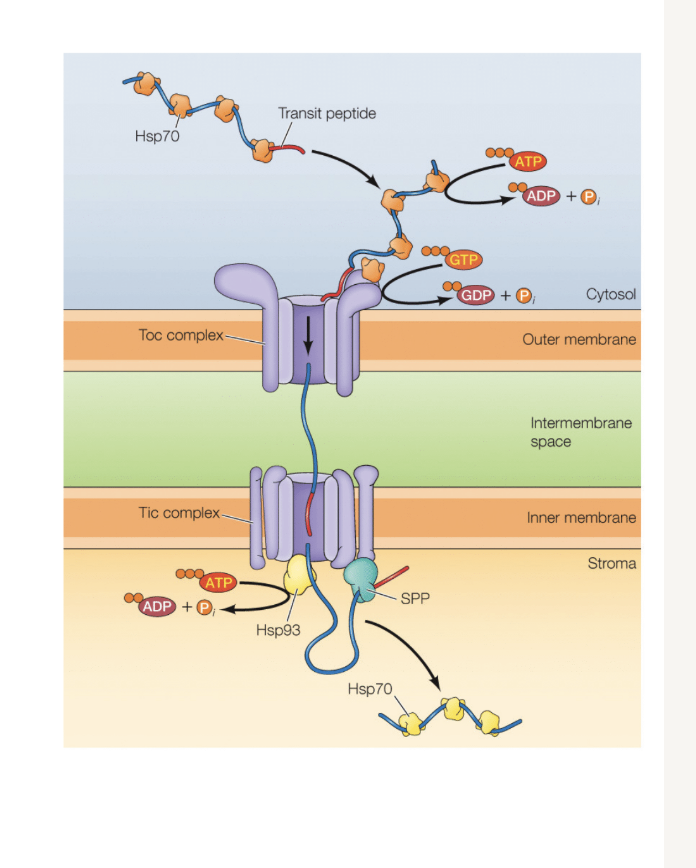
Write out the pathway for a protein destined for chloroplast stroma (chloroplast protein import)
more simple pathway to be completed
N-terminal signal sequence
Go through TOC complex
Then through TIC complex (requires energy from atp)
Sequence signal cleaved by spp
HSP are chaperones (do not go through the complexes)
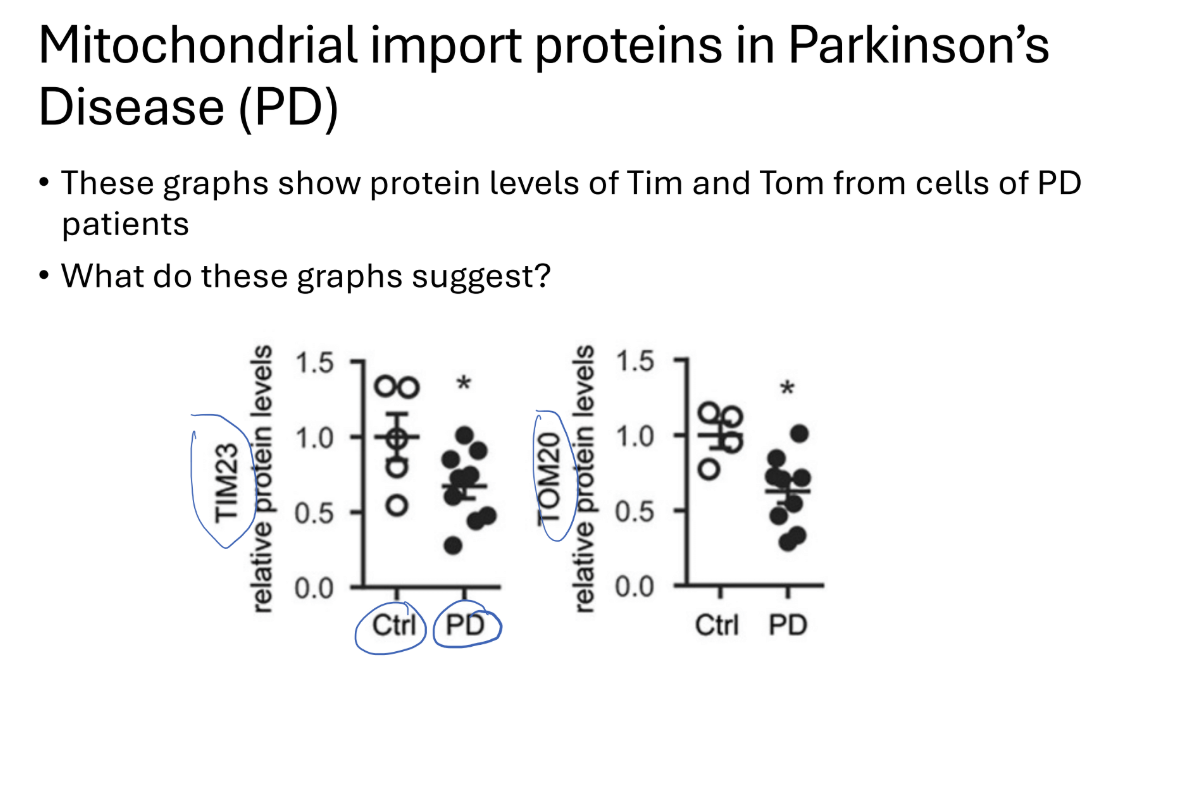
How does mitochondrial import protieins in parkisons diease
Know how to look at the graph
Tim and tom complex proteins levels are decreased in PD
Leads to cell dysfunction and cell death
Overexpresison of TOM in PD patient cells
As ros levels indicate cell stress increased ROS with increased cell stress)
Researchers overexpressed TOm in pd cells (in vitro)
REsult?
Decreased cell stress
Possible target for further research on PD
Q? what would happen if there is no proton gradeient
protiens would get stuck in the intermembrane space- no drive to go through the Tim complex
Where would proteins be if their presequence was cleaved off prematurely
the cytosol
What are some similarietes and differences between mitochodnrial and chloroplast transport

What are the components of the mitochondria (plymer and monomer forms) (tructure and use)
Actin
“Monomer” form
G-actin (globular)
Single protein
Attached to ATP (or ADP)
“Polymer” form
Polarity
F-actin (filament)
Monomers attach together
Has polarity (ends are different )(plus and minus end)
Function(s)
Cell movement, cell structure microvilli
Microtubules
Monomer” form
Tubulin dimer: alpha and beta subunits
Attached to GTP (or GDP)
“Polymer” form
Cylender shape, dimers strung togehter
Function(s)
Mitotic/meiotic spindle, Cell shape, movement, organelle transportli
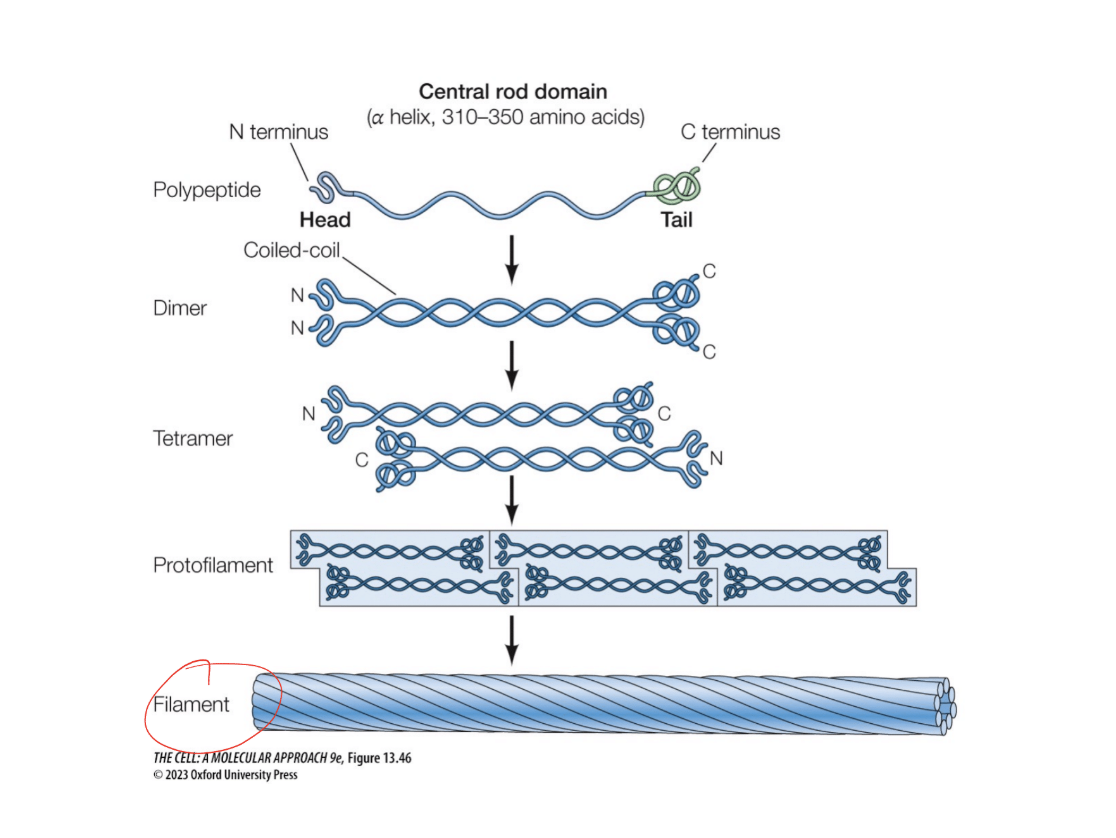
Intermediate filaments
Monomer form
Two polypetiedes come together in a coiled-coil structure
Polymer form
A tetramer forms. Tetramers come together as protofilaments. Multiple protofilaments come to form a filament
Function
Keeps cells together, especially as we think about tissues
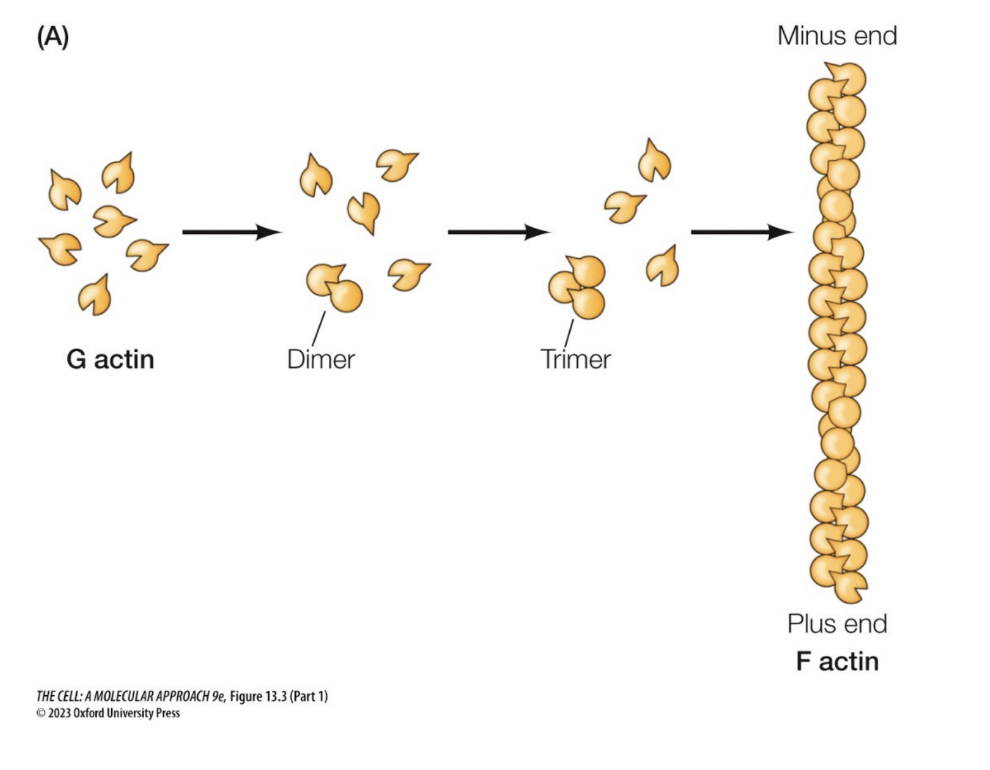
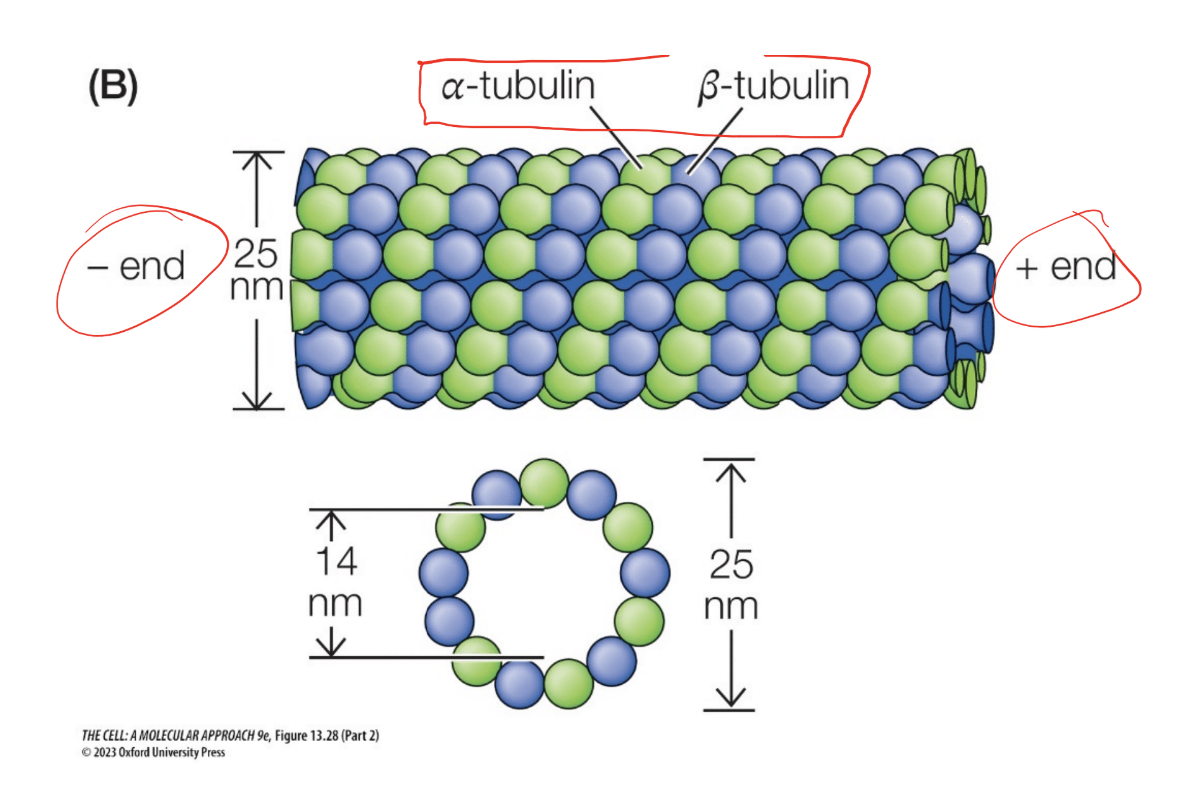
How does the actin polymer form
A. g actin globulars have different ends( they have polarity due to differences in natural shape)
actin molecules are able to attach to become a polymer

Describe the tubulin structure
tubulin structures have alpha tubulin and beta tubulin. Has a plus and minus end for polarity.

Do intermediate fillaments have polarity???
No lol
What is treadilling? Which filamments undergo this mechanisms? how is it similar between those molecules? what would happen if you cut in between the fillaments
Treadmilling
Microtubule and actin
“ monomers” with ATP/GTP attached are added to the plus end (growing end) of the filament
ATP for actin, GTP for tubulin of microtubules
ATP/GTP hydrolyzed at some point after addition
Monomers with ADP/GDP attached are not as tightly bound and are lost off the minus end of the filament
Disassembly of the filaments
How do cells move and what is used in the fuction (which proteins and components
RHO
Receives the signal to migrate. Activates RHO on actin
Proflin
Exchange the Adp to atp on the monerm (forms ATP-actin)
Formin
Forms. Forms actin filaments in the forward direction
ArP2/3
Building the actin branches off of the actin filament
How are microtubles oriented in the cell?
Minus end at the centrosome in the center of the cell
Positive ends on the microtubles
Closer to the outside of the cell (cell membrane)
What happens when the cell is dividing
The minus sides remain cetered at the centrisomes
Pluss signs overlap and attach to the chromosomes themselves
What ar ethe different types of microtubules and what are their funcitons
Kinetochore microtubles
proteins are in the chromoses
they try to bring the chromosome to one sid eof the cell
Kenisins (attach to the chromosome ) (microtuble dissasebly
Kensin will walk to the minus end of the microtubule
the microtuble is activley dissasembled
Interpollar microtubules
motor protiens (kinesin) walks to the plus end an dpushes the microtubles apart
Slides apart to pull the chromosomes to opposite ends
Astral microtubles
motor proteisn dynein walsk to inus end and pulls apart the microtubles to edges
What gies the ecm these charachteristics :STrong network, gel like consistancy, connection of ECM to cell
What would happen if interins are over or underactive
Integrins link cell to extracellular matrix
Focal adhesion?
Cell is grabbing onto the floor for cell movement (floor is ecm) (helps move forward)
Sends signal to move cell forward
What are the cell juncions
!. Adherins junctions and Desmosomes
Cadherins mediate adheision between fillaments
Adherein junctions link actin fillaments of adjacesnt cells
Desmosomes use desmosomal cadherins to connect intermeiate fillamets, provding structural support
Tight junctions
forms barriers between compartemnts and regulate molecular passage
prevents diffusion of membrane proteins
not linked to cytoskelital elements
Gap junctions
allwos direct cytoplasmic communication between adjacent cells through connections
enables the passage of ions and small molecules for cooridnated resposes (ex heart muscel)
What are some of the key components of the EC
Collagin:
most abundant ECM protein, composed of three polypetide chains wound tighly togehter. Glycine occurs at every third amino acid (helps keep in close packing of the chains)
Matrix polysacharides
Proteglycans: proteins with carbohydrait chains attached, on the outside of the cell
Glycosaminoglans (GAGS): long branched polycaccharides tha attach to proteins to form proteglycans
provide a gel- like consisency to the ECM by attracting water molecules due to negatively charged sulfate groups
Adhesion proteins
Fibronectin: connects collagin and proteglycanc to cell surfaces via integrins
Laminin: binds to eachother, integrins, and proteglycans promoting cell adhesion and communication.
describe some of the Cell-ecm interactions
cells can interact with the ECM through adhesion proteins (intigrins)
intigrins: transmembrane proteins that link the ECM to the cytoskeletin
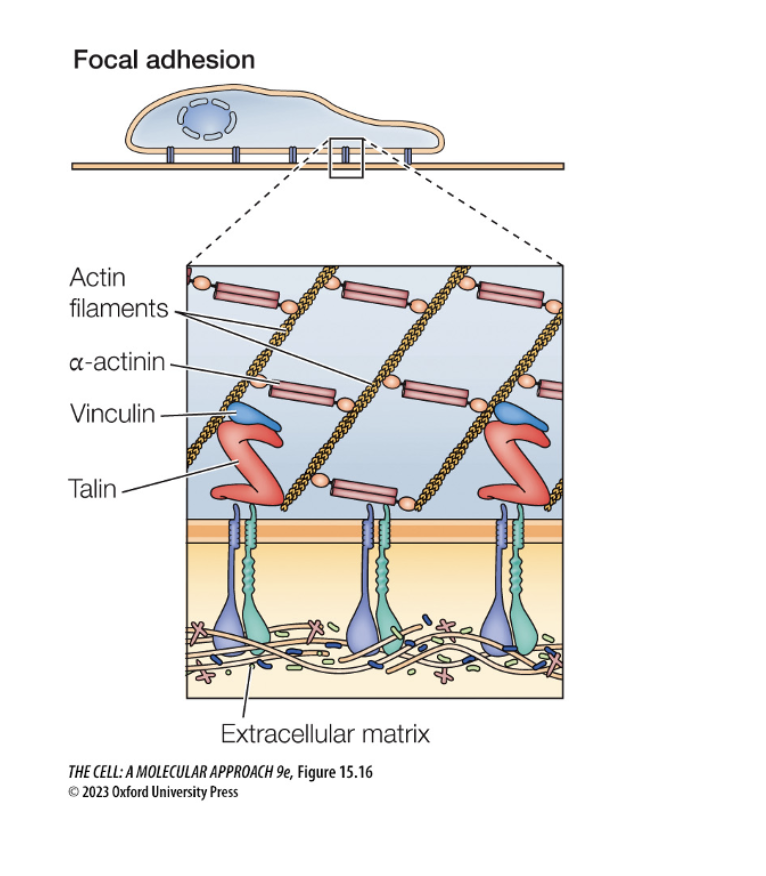
Which of the following would happen if clathrin-mediated vesicle formation is inhibited?
Endocytosis from the plasma membrane would be disrupted
Proteins destined to be secreted from the cell will be made by what
the rough er!!!