Chapter 6 Lecture Flashcards
1/46
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
47 Terms
Cells require:
•a membrane separating the inside of the cell from the outside
•a way to encode/transmit information
•energy to do the work of the cell: move, grow, pump ions, and perform reactions necessary for cellular function, among other things
Cells require a _________ separating the inside of the cell from the outside
membrane
Cells require a way to ______________ information
encode/transmit
Cells require ______ to do the work of the cell: move, grow, pump ions, and perform reactions necessary for _____ ______, among other things
energy, cellular function
______ is the capacity to cause change
Energy
Energy exists in various ______, some of which can perform work
forms
___ is the movement of matter against opposing forces, such as gravity and friction
Work
_____ ______ is energy associated with motion
Kinetic energy
_________ _______ is kinetic energy associated with random movement of atoms or molecules
Thermal energy
___ is thermal energy in transfer from one object to another
Heat
_____ is another type of energy that can be harnessed to perform work
Light
________ _______ is energy that matter possesses because of its location or structure
Potential energy
______ ______ is potential energy available for release in a chemical reaction
Chemical energy
Energy can be __________ from one form to another
converted
What is an example of potential energy?
Energy within chemical bonds of a molecule
Catabolism
breakdown of molecules into smaller units, producing ATP
Anabolism
building of molecules from smaller units, requiring an input of energy, ATP
Gibbs Free Energy
ΔG = ENERGY IN PRODUCT – ENERGY IN REACTANTS (ΔG = Gproducts – Greactants)
Endergonic Reaction have a
positive ΔG
Exergonic reactions have a
negative -ΔG
ΔG = ΔH - TΔS
S = entropy
H = enthalpy
T = temperature (Kelvin)
G = Gibbs free energy
Δ = “change in”
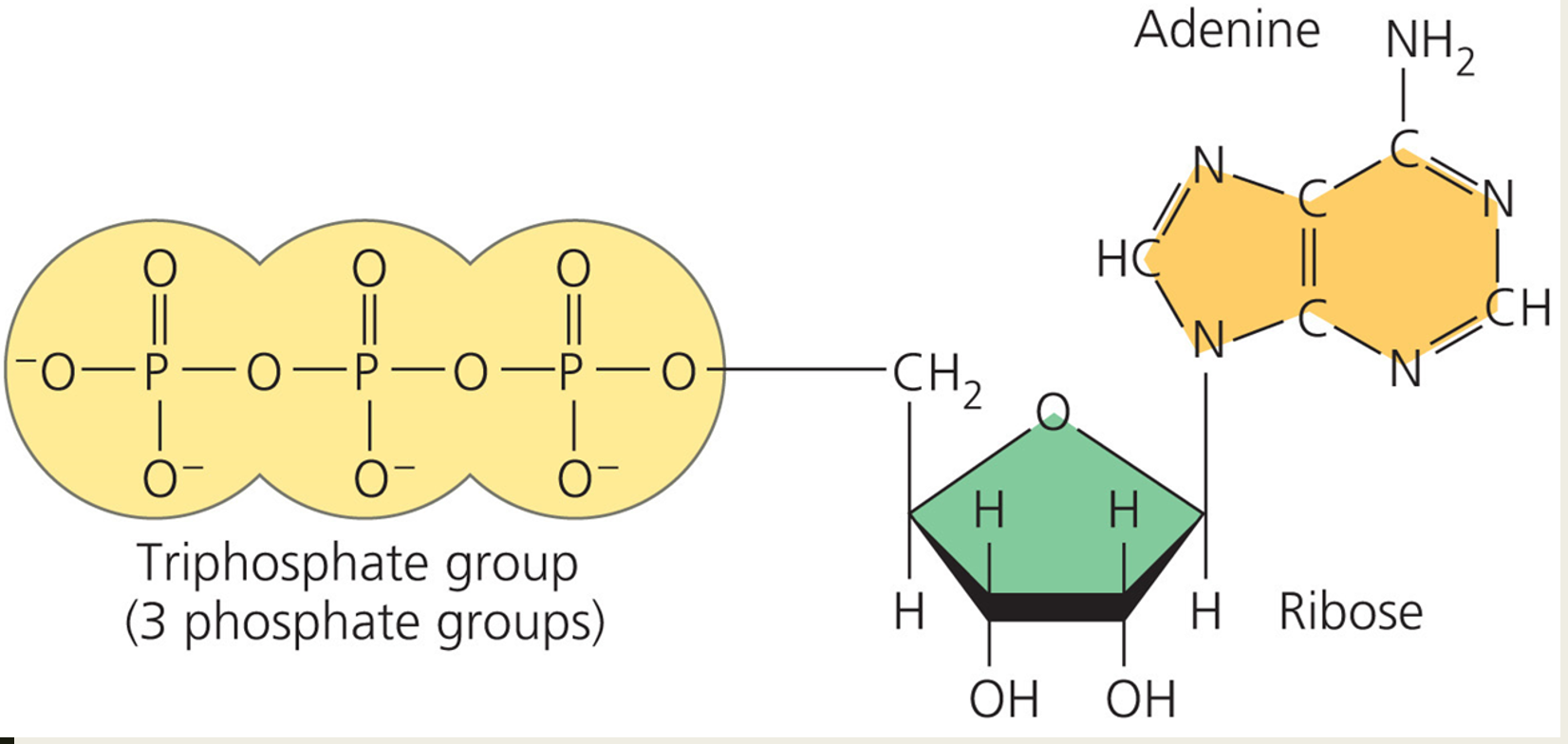
___ – the energy currency of the cell
ATP
Enzymes work by __________ the energy required to have a reaction
lowering
A reaction with a positive ΔG value is ______
Endergonic
_______ ___ _______ depends on energy needed to activate reaction
Rate of a Reaction
_______ are substances that increase the rate of a chemical reaction without themselves being consumed.
Catalysts
Catalysts are usually proteins called ______
ENZYMES
Enzymes are _______ ________, therefore they play a crucial role in determining which chemical reactions take place.
HIGHLY SPECIFIC
Hydrolysis of sucrose by the enzyme sucrase is an example of an ________ ______ reaction.
enzyme-catalyzed
What is the specific region on the enzyme where the substrate binds?
The active site
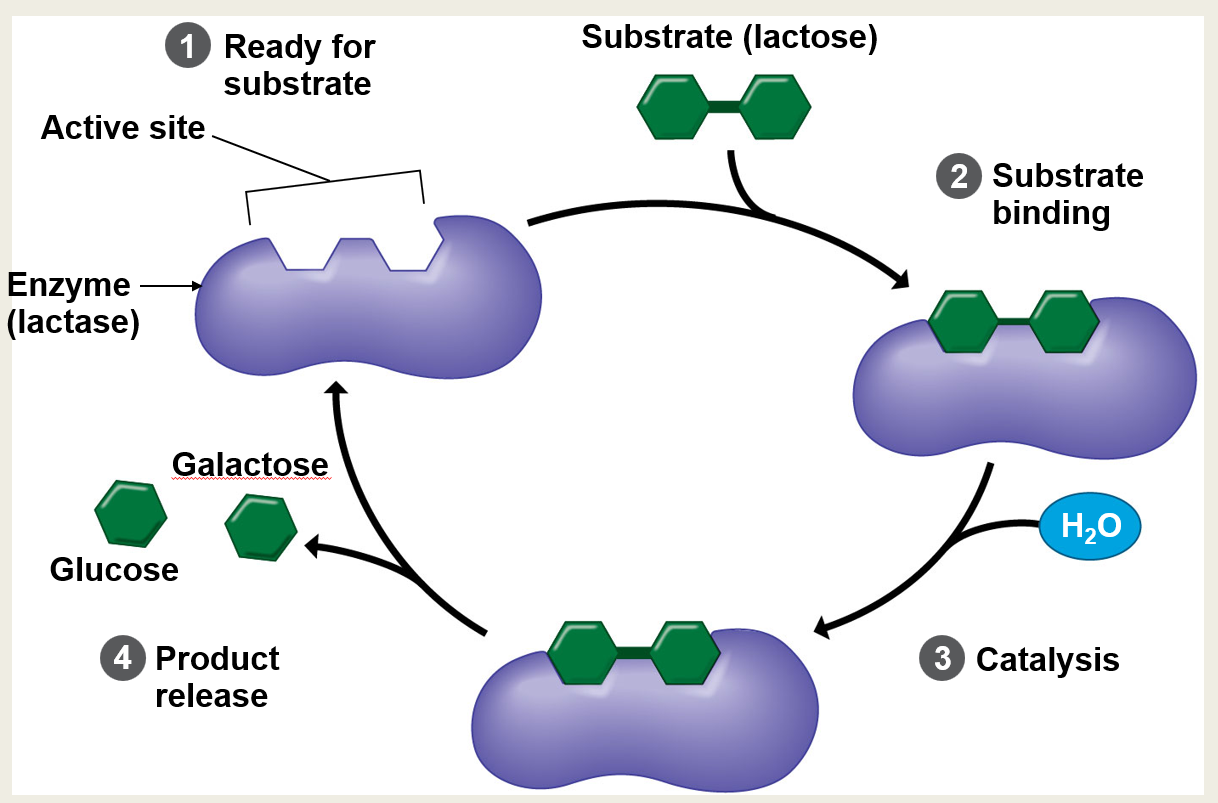
In this specific example, identify the enzyme and its target substrate.
The enzyme is lactase and the substrate is lactose.
What are the four main steps of the enzyme cycle shown in the diagram?
1. Ready for substrate 2. Substrate binding 3. Catalysis 4. Product release
What molecule is introduced during the catalysis step to help break down the substrate?
Water (H₂O). Note: This indicates it's a hydrolysis reaction.
What are the specific products released after lactase breaks down lactose?
Glucose and galactose.
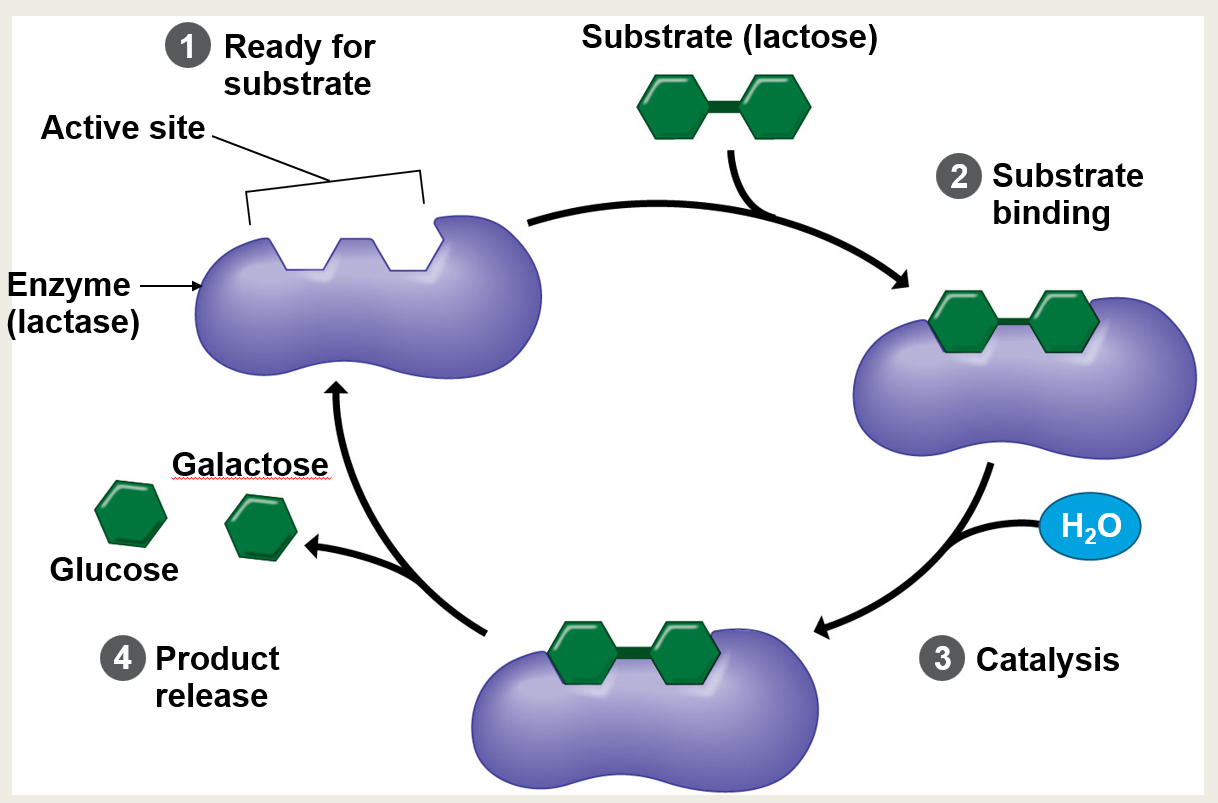
Is the process shown in the slide an example of anabolism or catabolism?
Catabolism. (The enzyme is breaking down a larger molecule into smaller, simpler ones).
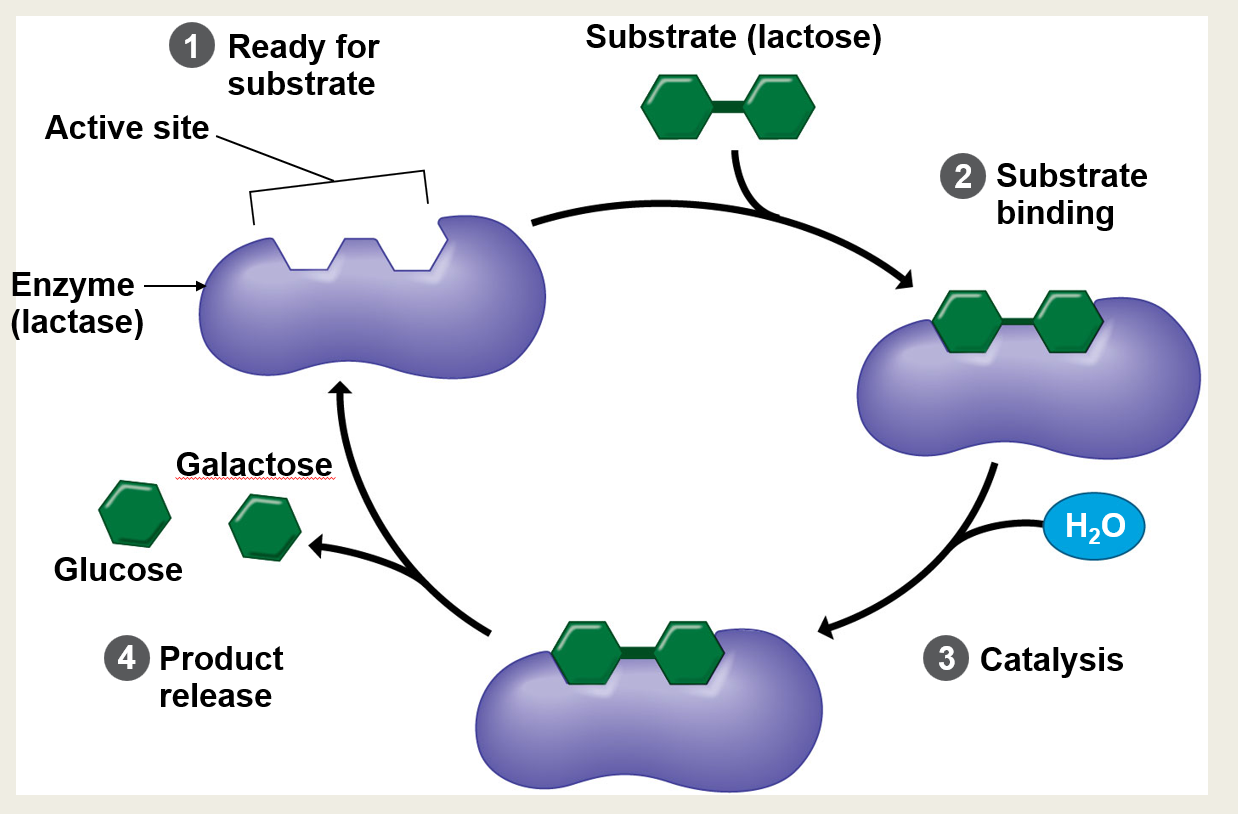
Is the reaction shown in the slide exergonic or endergonic?
Exergonic. (Catabolic reactions that break chemical bonds typically release energy).
What can affect enzyme activity?
Environmental/local factors like Temperature and pH
Regulation of reaction components
–Switching on or off the genes that encode specific enzymes
–Changing enzyme or substrate concentrations
–Regulating the activity of enzymes
Each enzyme functions best at an optimal:
–Temperature
–pH
Enzymes can be _________ outside of these optimal conditions
denatured
Denatured means:
Unfolded
Regulation of enzyme activity within the cell:
enzyme concentration
What can affect enzyme activity?
■Regulation of reaction components
–Switching on or off the genes that encode specific enzymes
–Changing enzyme or substrate concentrations
–Regulating the activity of enzymes
Inhibitors
decrease the activity of enzymes.
a.Competitive
b.Noncompetitive
c.Negative feed-back
Activators
increase the activity of enzymes.