5. Carbon capture and storage
1/10
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
What leads to the production of CO2
Combustion of fossil fuels leads to the production of CO2, which is one of a suite of greenhouse gases, attributed to anthropogenic driven global climate change
Why carbon capture and storage (CSS)
many industrial processes CO2
hydrogen required for non CO2 emitting heating will likely be derived from fossil fuels
renewable energy needs to be buffered by coal and gas fired power stations
emissions to atmosphere can be prevented by CSS
What is CCS & what can it do?
capturing CO2 at source, transporting it to a suitable storage location and then injecting it deep underground

CO2 separation process
absorption, adsorption and membrane separation
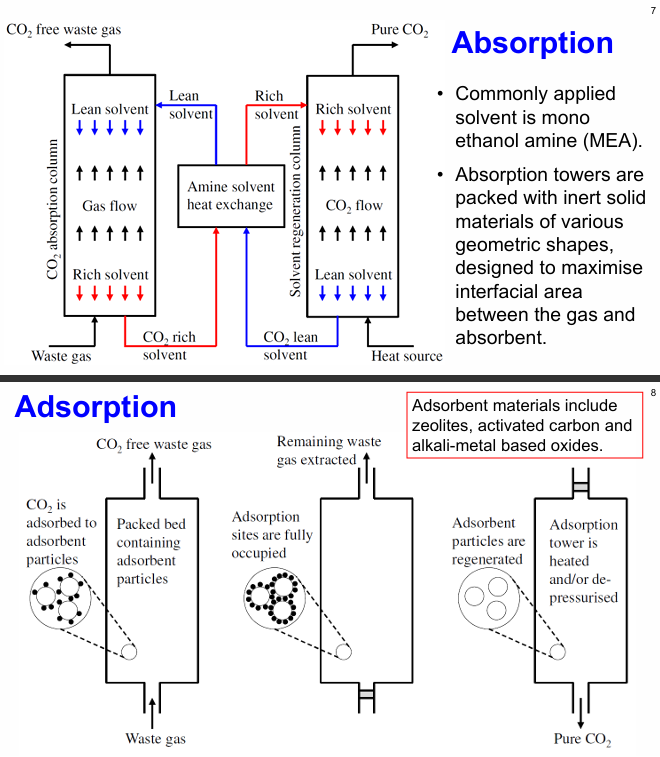
absorption
commonly applied solvent is mono ethanol amine (MEA)
absorption towers are packed with inert solid materials of various geometric shapes, designed to maximise interfacial area between the gas and absorbent
membrane separation materials
membrane materials include organic polymer options, inorganic options and mixed matrix options where inorganic particles are combined within a polymer matrix
The cost of CO2 separation, on a USD per tonne of CO2 basis, increases with
decreasing concentration in the gas stream
why does the cost of CO2 separation increases with decreasing concentration in the gas stream
reduced concs imply that a larger volume of waste gas must be handled for a given mass of CO2, which leads to an increase in capital cost for the separation equipment
the more dilute the source, the more energy is required to overcome the entropy of mixing, to separate out a pure stream of CO2
cement production
cement production involves sintering (fusing without melting) calcium carbonate with silica, alumina and iron oxide. This requires the CaCO3 to go through an intermediate process of calcination, whereby calcium oxide and CO2 are formed (CaCO3+heat→ CaO+CO2)
Iron and steel production
Iron and steel production involves mixing iron ore with coke (a high carbon content fuel) in the presence of preheated air within a blast furnace. The coke reacts with O2 in the air to form carbon monoxide. The CO is then used to reduce the iron ore to form elemental iron and CO2

Hydrogen production
Hydrogen is commonly formed from methane using a process known as steam methane reforming. This involves mixing preheated steam with CH4 to form CO and H2. The CO is then mixed with water, in a process known as a water gas shift reaction to produce additional H2 and CO2
