SCIENCE - Periodic Table, Elements and Bonding
1/49
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
50 Terms
Atom
The smallest part of an element, which still has the properties of that element. Everything is made of atoms. No one has ever seen an atom.
Element
Contains one type of atom. It is the simplest form of a substance. Each element is given a unique symbol, e.g. O for Oxygen
Molecule
When 2 or more atoms join together chemically (bonded), e.g. O2,
Compound
It forms when two different elements chemically combine, e.g. H2O
What is air made of?
A mixture of molecules and compounds
What is the atom made of?
Protons (+)
Neutrons
Electrons (-)
Where are Protons and Neutrons found?
The nucleus
What do electrons do?
They spin around the nucleus
What are Protons, Neutrons and Electrons known as?
Subatomic particles
Proton Mass
1
Neutron Mass
1
Electron Mass
0 - It would take 2000 electrons to weigh the same as a proton
Atomic Number
The number of protons in the nucleus of that element
Number of Protons = Number of Electrons
True or False: Elements are completely neutral
True
Mass Number
The number of protons and neutrons in the nucleus of the atom
How do you find the amount of Neutrons?
Mass Number - Atomic Number = Number of Neutrons
What model is typically used to show an atom?
The Bohr Model
Electron Configuration
How you display electrons in an atom
Electron Configuration Rules for Bohr Model
Fill from inside to outside
First orbit - Up to 2 electrons
Second orbit - Up to 8 electrons
Third orbit - Up to 8 electrons
Periodic Table
A list of all known elements (118) created by Demitri Mendeleev
Vertical Columns
Called Groups
They have elements that have similar physical and chemical properties
Horizontal Rows
Called Periods
They are in order of increasing proton number
Group Names
Group 1 - The Alkali Metals
Group 2 - The Alkali Earth Metals
Group 7 - The Halogens
Group 8 - The Noble Gasses
Group 1
THE ALKALI METALS:
They have 1 electron in their outer shell - They want to get rid of this electron
This group is very reactive - Can even react with the air - Increasing reactivity as you move down the group
Very soft - can be cut with a knife
Form +1 ions
Group 2
THE ALKALINE EARTH METALS:
2 electrons in outer shell
Reactive - not as much as Group 1
Harder than Group 1
Form +2 ions
Group 7
THE HALOGENS:
7 electrons in outer shell - need to gain one to have a full shell
Highly reactive
Form -1 ions
Group 8
THE NOBLE GASSES:
All gasses
Full shell of outer electrons
Very unreactive - Stable
Do not form ions
Full Outer shell =
Stability
Group Number Electron Correlation
The group number correlates with the number of electrons in the atom’s outer shell
Chemical Bonding
Two types:
Ionic and Covalent
Ionic Bonding
When one atom loses electrons and another atom gains electrons.
An ionic bond is the force of attraction between oppositely charged ions in a compound
Where does Ionic bonding occur?
Between metals and non-metals
Examples of Ionic Bonding
NaCl (Sodium Chloride)
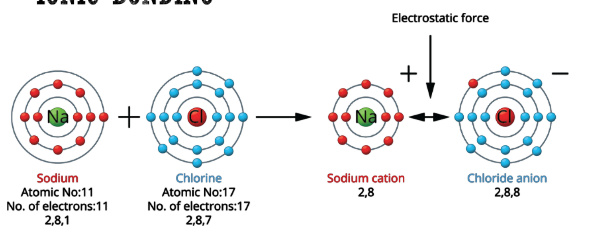
Sodium Ion
Sodium has only one electron in it’s outer shell
It needs to lose the electron
If it loses one electron, it will become positive as there are more protons than electrons
After losing an electron, sodium becomes an ion and is represented like [Na]+
Atom that has lost electrons
Cation (+)
Atom that has gained electrons
Anion (-)
Why do Ions attract each other?
They attract each other because the have charges. Opposite charges make this bond very strong.
Ionic Bonding Properties
Most ionically bonded substances are solids at room temp.
Most have high melting points because they are so strong
Because the ions have their own charge, they conduct electricity
They are soluble in water
Covalent Bonding
Covalent bonds allow atoms to share electrons so that they can get a full outer shell
It is a strong bond made up of a shared pair of electrons between two atoms
Where does Covalent Bonding occur
Between non-metal atoms
Simplest Covalent Bond
Hydrogen (H2)
Covalent Bonding examples
H2O Water
CH4 Methane
Cl2 Chlorine
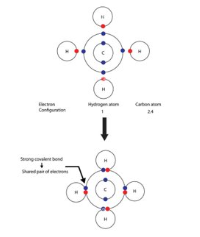
Methane
CH4
Carbon wants to gain 4 electrons
Hydrogen wants to gain 1 electron
One carbon can bond and share electrons with four hydrogens
Chemical Formulas
Every compound has a formula. It tells you the type and number of atoms present. Made up of letters which denote the type of atom and numbers which tell how much of that atom is there, e.g. CH4 = 1 carbon 4 hydrogen
Combining Metals and Non-Metals (naming)
When metals and non-metals are combined they are named as follows:
Metal + Non-metal + ide
Examples: Sodium + Chlorine = Sodium Chloride
Combining Metals, Non-metals and Oxygen (naming)
When metals, non-metals and oxygen are combined they are named as follows:
Metal + Non-metal + ate
Example: Potassium + Phosphorous + Oxygen = Potassium Phosphate
Combining Metals and Non-metals
We need to work out the combining power of an atom to combine it (This means how many electrons does the atom need to become stable and non-reactive)
Example:
Hydrogen - Group 1 - Needs 1 more electron to be stable - Combining power = 1
Carbon - Group 4 - Needs 4 more electrons to be stable - Combining power = 4
It would take 4 hydrogen atoms to every 1 carbon atom to keep both atoms stable
CH4 is formed
Which 3 elements is the Octet rule not true for
Hydrogen, Berylium and Boron
Outer shell also known as
Valence shell