Organic Chemistry 2026 Exam
1/66
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
67 Terms
Organic Molecules
Hydrocarbon skeleton that may contain functional groups including carboxylic acids, amides, amines, alcohols, esters, ketones and aldehydes
Functional Groups
Groups of atoms or bonds within organic molecules that are responsible for that molecule’s characteristic chemical properties
IUPAC Nomenclature
1. Priority order- carboxylic acids, amides, aldehydes, ketones, alcohols, amines etc.
2. The numbering system exception is applied to carboxylic acids, aldehydes and amides that all contain double bonds- ketones do not apply due to having two free bonds available
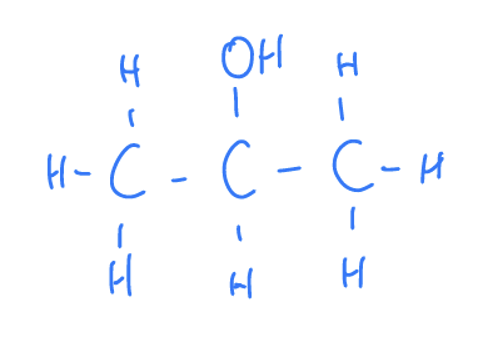
What Are Alcohols
Where a carbon is attatched to a hydroxyl group (-OH)
Properties of Alcohols
Priority functional group- the organic molecule ends with suffix ‘ol’ otherwise prefix ‘hydroxy’. They are not basic since the molecules do not ionise to create hydroxide ions (charged -OH group)
The Three Types of Alcohols
Primary, secondary, tertiary
Primary (1°) Alcohol
–OH carbon attached to one other carbon
Secondary (2°) Alcohol
–OH carbon attached to two other carbons
Tertiary (3°) alcohol
–OH carbon attached to three other carbons
What Are Aldehydes
They have carbonyl groups on the carbon at the end of the chain
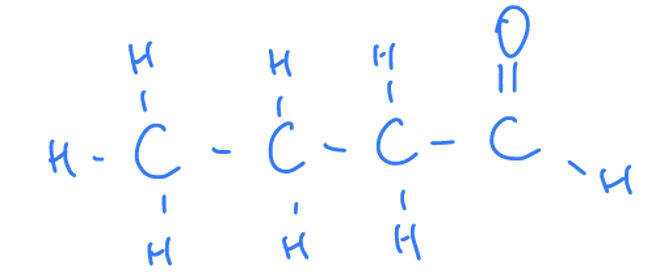
Properties Of Aldehydes
Aldehydes are named by placing the suffix “-al” at the end of the name of the longest carbon chain.
What Are Ketones
Have a carbonyl group attached to a carbon that is not at the end of the chain, ie, somewhere in the MIDDLE of the chain.
Properties of Ketones
The functional group is abbreviated as R-CO-R’. Ketones are named by placing the suffix “-one” at the end of the name of the longest carbon chain.
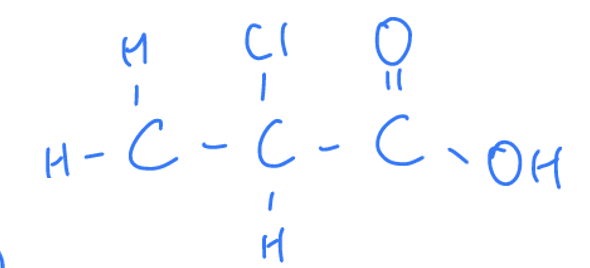
What Are Carboxylic Acids
Compounds that contain a carbonyl group and a hydroxyl group.
Properties of Carboxylic Acids
End in the suffix ‘oic acid’. They react with bases to form negatively charged carboxylate ions as acids lose hydrogen ions. React in the same way as weak inorganic acids HCl and HNO3 by partially ionising to form hydronium ions in water.
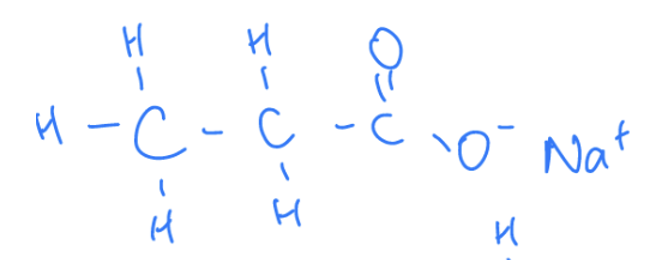
What Are Carboxylate Ions
When carboxylic acids react with bases or lose a hydrogen ion to form negatively charged carboxylate ions.
Properties of carboxylate ions
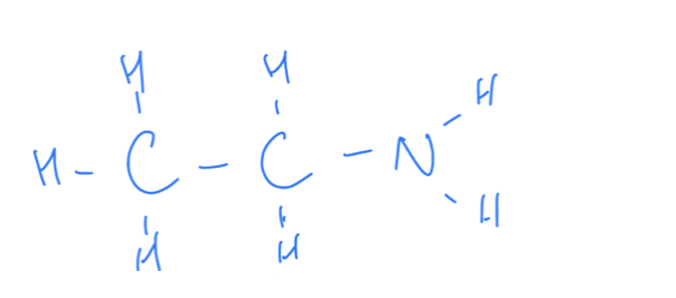
What Are Amines
Amine groups contain an amino functiona group (NH2)
Amine Properties
When considered the priority functional group the suffix ‘amine’ is used however more commonly the prefix ‘amino’ is used. Amines are basic functional groups as they can react with acids.
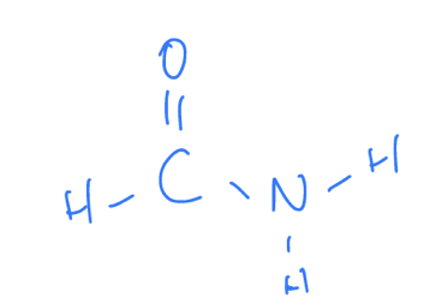
What Are Amides
Amides contain a carbonly functional group (C=O) attatched to an amine functional group.
Properties of Amides
Ends with the suffix ‘amide’ or in the rare case starts with prefix ‘amide’.
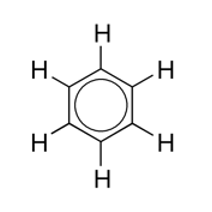
What Is Benzene (C6H6)
An aromatic compound that contains a benzene ring with a MF of C6H6 and an EF of CH.
Properties of Benzene
Expected to undergo addition reactions and possess 6 carbon bond lengths averaging between single and double bonds (due to 3 of each), yet shows high stability from its oscillating structure and fails to react as predicted. Non-conductor of electricity- although electrons move within the ring they are delocalised, so they benze reacts differently from alkanes, alkenes and cyclic hydrocarbons.
Isomers
Compounds with the same molecular formula but different arrangement of atoms (structural formula).This leads to different physical properties (MP/BP) and chemical properties (reactivity, pH).
Chain/Positional Isomerism
Compounds have the same functional group but present in different positions or are with different fucntional groups e.g. propanal vs propanone
Geometric/Stereoisomerism
Cis or trans isomerism occurs in alkenes where there is restricted rotation about the C=C bond and each double-bonded carbon is attatched to two different groups.
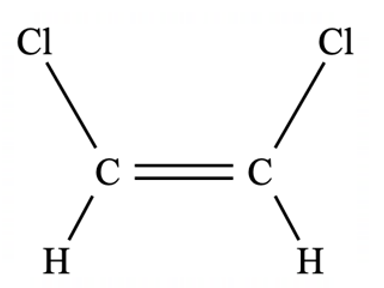
What Are Cis Isomers
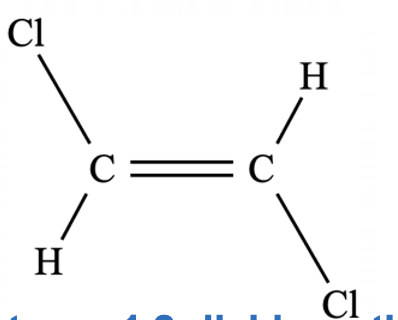
What Are Trans Isomers
Explain why do trans isomers generally have a lower boiling point than cis isomers
As the molecule is symmetrical, the dipoles cancel out and the molecule is non-polar, meaning only dispersion forces are present. The other two molecules are polar and also have dipole-dipole forces.
What Are The Isomers Of Eachother
aldehydes and ketones, esters and carboxylic acids
What are dipole-dipole forces and …
What is the hydrogen bonding
What is the dispersion forces…
The strength of dispersion forces rule
In general the more electrons a particle has, the stronger the dispersion forces. (Means requires more energy to break IMF, increasing the intermolecular forces)
Ionic Bonds
Ion-dipoles
What functional groups have hydrogen bonding as the dominant force
Alcohols, amides, amines, carboxylic acids
What functional groups have dipole-dipole forces as the dominant force
Ketones, aldehydes, esters and halogens
What functional groups have dispersion forces only as the intermolecular force between its molecules
Cyclic, alkenes, alkanes and alkynes
What are the three factors that affect the strength of the forces between molecules from the same homologous series
The number of electrons in molecule, the surface area of the molecule (only considered when comparing molecules with a similar number of electrons, the accessibility of the functional group (only considered when the same functional group is moved to a different position in the carbon chain).
The states of homolgous series under standard conditions
CH4 to C4H10 (gaseous), C5H12 TO C17H36 (liquids), C8H38 (soilds). This is because the boiling points of alkenes increase primarily due to increase in the size (molar mass) of molecules.
What affects the surface area of molecules
A straight-chain molecule will have a boiling point higher than isomer with branches (methyl and ethyl groups) due to the greater surface area in contact, thus the greater dispersion forces, between adjacent molecules.
What are the key steps in explaining boiling/melting points of molecules?
List ALL forces in each of the chemicals 1. Discuss the dispersion forces using: a. the number of electrons and b. the differences in surface area if there is branching (methyl/ethyl groups) and the number of electrons is very similar 2. Describe the effect of the functional groups. Discuss accessibility only if the same functional group is present but in different positions on the carbon chain. 3. A summary statement about the “sum of forces” and the energy required to break these, and the comparison of melting/boiling points.
Explain the solubility theory
When a solvent and solute are mixed to form a solution, several things must occur. Forces of attraction exist between solute particles (solute-solute forces). Forces of attraction exist between solvent particles (solvent-solvent forces). There must be forces of attraction between the solute and solvent particles (solute-solvent forces) that are strong enough to: separate the solvent molecules and separate the solute molecules.
Explaining solublity in terms of strength of IMF
1. Identify all solute-solute forces, and identify the predominant force. 2. Identify all solvent-solvent forces, and identify the predominant force. 3. Identify all solute-solvent forces, and identify the predominant force. 4. Explain how the forces between the solute and solvent are/are not strong enough to overcome the existing forces between solute particles AND existing forces between solvent particles.
Explaining solubility in terms of energy
1. Identify all solute-solute forces, and identify the predominant force. 2. Identify all solvent-solvent forces, and identify the predominant force. 3. Identify all solute-solvent forces, and identify the predominant force. 4. Explain whether the energy released when forces form between the solute and solvent is bigger/smaller than the energy required to overcome the existing forces between solute particles AND existing forces between solvent particles. If energy released is greater than energy required, it is soluble.
Explaining slight solubility
Strong enough to partially overcome the existing forces between… OR The energy released is similar to the energy required to overcome the existing forces between…
Combustion
Organic molecules burnt in O2 gas to form CO2, C or CO + H2O
Complete Combustion General Formula
Organic molecule (g) + O2 (g) —> CO2 (g) + H2O (g)
Incomplete Combustion General Formula
Organic molecule (g) + O2 (g) —> C (s) + H2O (g)
What is the substitution reaction
Alkanes can have a hydrogen atom ‘substituted’ by another atom requiring energy (light) to break covalent bonds in atom.
What is an addition reaction
Alkenes (and alkynes) have a molecule ‘added’ to them to make one molecule as the product. The molecule added could be hydrogen gas (hydrogenation) requiring a Ni, Pd or Pt catalyst, halogen (halogenation) or water (hydration) requiring a H2SO4 or H3PO4 catalyst.
Alkenes
Alkenes are unsaturated as other atoms can be added on due to the double bond between two carbon atoms. If an orange bromine solution/water (aq) is added it will quickly react to decolourise the bromine solution.
Alkanes
Alkanes are saturated as no other atoms can be added on due to only single bonds between carbon atoms. If an orange bromine solution/water (aq) is added it will slowly react to decolourise the bromine solution.
Oxidation
Alcohols can be oxidised to aldehydes, ketones and carboxylic acids by oxidising agents
What are the oxidising agents used
Acidified potassium dichromate solution and acidifed potassium permanganate solution
What would be observed in a oxidation reaction involving acidifed potassium dichromate solution
A orange solution turns deep green
What would be observed in a oxidation reaction involving acidifed potassium permanganate solution
A purple solution turns pink/colourless
The four oxidation equations
1. oxidation of a primary alcohol to an aldehyde (mild conditions), 2. oxidation of a primary alcohol to a carboxylic acid (strong conditions), 3. oxidation of a secondary alcohol to a ketone (any conditions) 4. oxidation of a tertiary alcohol has no reaction under any condition
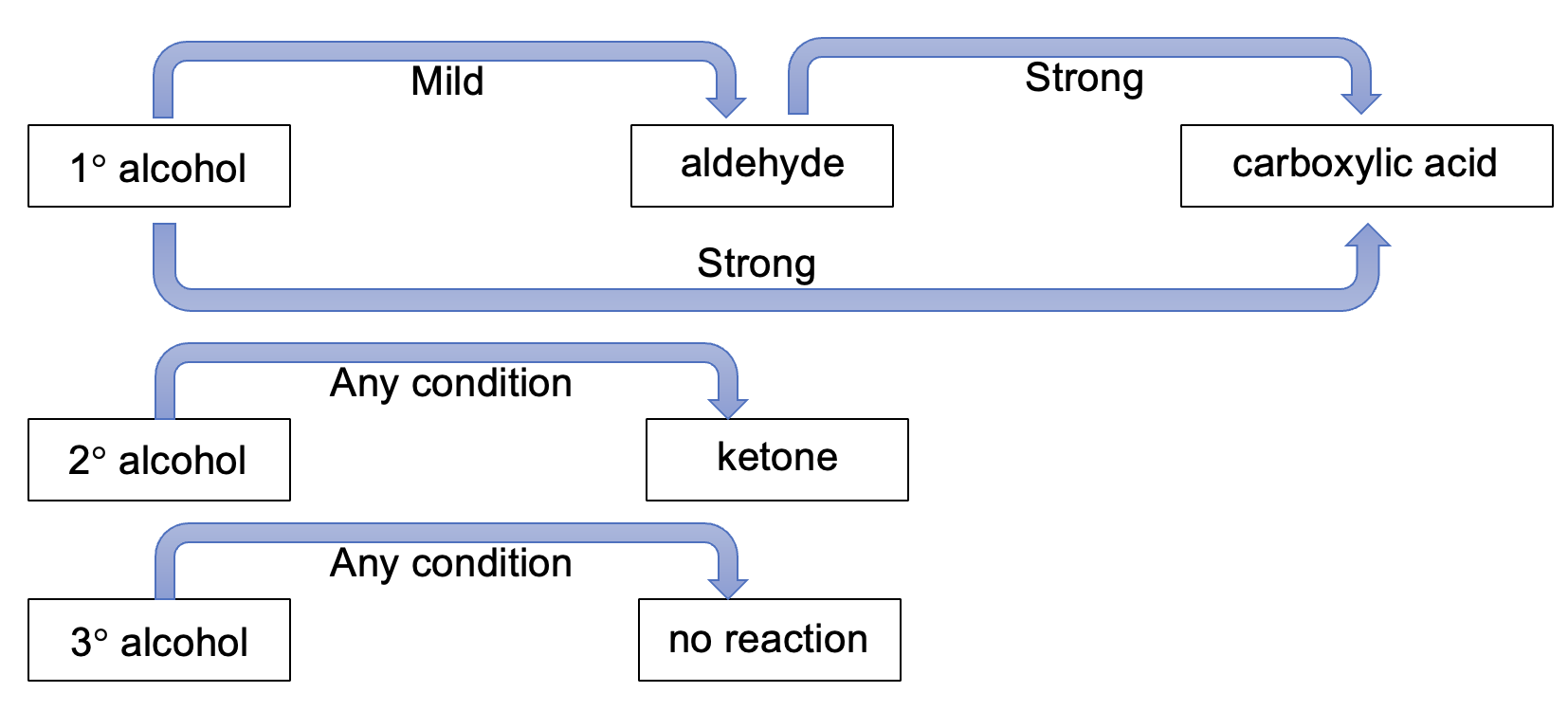
Describe the mild conditions for an oxidation reaction to occur
Acidified potassium dichromate that is unheated, not in excess and/or dilute
Describe the strong conditions for an oxidation reaction to occur
Acidified potassium dichromate that is heated, in excess and/or concentrated. Acidifed potsasium permanganate under any condition.
Esterification
Condensation reaction- alcohols and carboxylic acids will react to form esters and water, requires sulfuric acid catalyst. The ‘yl’ is determined by the alcohol reactant and the ‘oate’ is determined by the carboxylic acid reactant
Esterification General Formula
Carboxylic acid + alcohol —> ester + water, where sulfuric acid presents as a catalyst
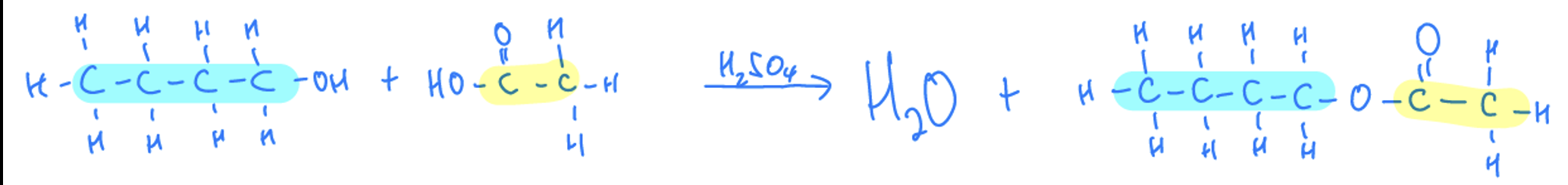
Describe the smell of an ester
Esters usually have a "sweet smell" and will be used as food additives to give a nice odour like in lollies and bubble gum.
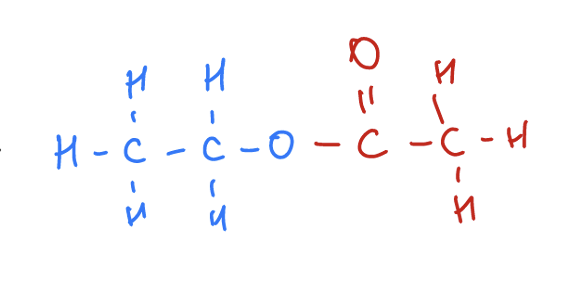
The purpose of ethyl ethanoate
Used as nail polish remover (solvent) alternative to acetone
What is the hydrolysis of esters
Typically the reverse of esterification- splitting an ester into a carboxylic acid and an alcohol using water.
Acidic hydrolysis