Chemistry 12 - Chapter 5: Applications of Acid-Base Reactions
1/45
Earn XP
Description and Tags
my memory is failing me so i gotta hit the knowt cards again
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
Hydrolysis
reaction of an ion with water to produce either the conjugate base of the ion and hydronium ions or the conjugate acid of the ion and hydroxide ions
Reaction type 1: Neutral Salts
“A salt containing the anion of a strong monoprotic acid and the cation of a strong base will produce a neutral solution in water because neither of the ions undergoes hydrolysis”
ion from strong acid + ion from strong base
alkali ions and alkaline earth ions do not hydrolyze (except Be)
this is because they are cations of strong bases
anions of strong monoprotic acids do not hydrolyze
this produces a neutral solution in water
Reaction type 2: Basic Salts
“A salt containing the anion of a weak monoprotic acid and the cation of a strong base will produce a basic solution in water because the anion acts as a weak base, producing hydroxide ions, and the cation does not react"“
ion from weak acid + ion from strong base
conjugate base of weak acid has a measurable ability to accept protons from water (hydrolyzes)
cation does not react because it is from a strong base
this results in a basic solution with pH > 7
Reaction type 3: Acidic Salts I
“A salt containing the cation of a weak base and the anion of a strong monoprotic acid will produce an acidic solution in water because the cation acts as a weak acid producing hydronium ions and the anion does not react”
conjugate base of strong acid + conjugate acid of weak base
conjugate bases of strong acids don’t react in water
conjugate acids of weak bases DO hydrolyze and act as a weak acid in water
this results in an acidic solution with pH < 7
Reaction type 4: Acidic Salts II
“A salt containing the anion of a strong monoprotic acid and a small highly charged metal cation will produce an acidic solution in water because the hydrated cation acts as a weak acid producing hydronium ions and the anion does not react.”
small highly charged cation = hydrated due to small size and high charge density
results in increased tendency to give protons away when hydrated
anion does not react
this results in an acidic solution with pH < 7
Reaction type 5: Salts containing weakly acidic cations and weakly basic anions
“A salt is containing a weakly acidic cation and a weakly basic anion will produce a solution that is:
acidic if Ka for cation > Kb for anion
basic if Ka for cation < Kb for the anion
neutral if Ka for cation = Kb for anion”
both ions hydrolyze and influence pH
write equilibrium equations and compare Ka to Kb
Reaction type 6: Salts containing the Cation of a Strong Base and an Amphiprotic Anion
“A salt containing a cation of a strong base and an amphiprotic anion will produce a solution that is:
acidic if Ka for the anion > Kb for the anion
basic if Ka for the anion < Kb for the anion”
only a partial neutralization of the amphiprotic anion occurs
Definition of a buffer
An acid-base buffer is a solution that resists changes in pH following the addition of relatively small amounts of a strong acid or strong base.
Components of a buffer
A buffer solution normally consists of a weak acid and its conjugate weak base in appreciable and approximately equal concentrations so that it has the ability to shift left or right across the equilibrium to respond to either acidic or basic stress.
Acidic buffer
Buffers a solution in the acidic range of the pH scale.
[H3O+] = Ka [HA]/[A-]
The hydronium ion concentration, and therefore the pH, of a buffer solution depends on two factors:
the Ka value of the weak acid
the ratio of the concentration of that weak acid to its conjugate base in the solution
If the concentrations of the acid and its conjugate base are equal, the hydronium concentration in the buffer solution is equal to the Ka value for the weak acid ([H3O+] = Ka and pH = pKa)
At a constant temp, Ka is constant so only the weak acid/conjugate base concentration ratio affects [H3O+]. If ratio increase, [H3O+] increases and vice versa, but changes in [H3O+] are only slight.]
When diluting a buffer solution, both concentrations of weak acid and conjugate base are reduced equally. Therefore, ratio remains constant so [H3O+] and pH doesn’t change when diluted. However, the number of moles of weak acid and conjugate base available changes.
Basic buffer
Buffers a solution in the basic range of the pH scale.
[OH-] = Kb [B]/[HB+]
The hydroxide ion concentration, and therefore the pH, of a buffer solution depends on two factors:
the Kb value of the weak base
the ratio of the concentration of that weak base to its conjugate acid in the solution
If the concentrations of the base and its conjugate acid are equal, the hydroxide concentration in the buffer solution is equal to the Kb value for the weak base ([OH-] = Kb and pH = pKb)
At a constant temp, Kb is constant so only the weak acid/conjugate base concentration ratio affects [OH-]. If ratio increase, [OH-] increases and vice versa, but changes in [OH-] are only slight.]
When diluting a buffer solution, both concentrations of weak base and conjugate acid are reduced equally. Therefore, ratio remains constant so [OH-] and pH doesn’t change when diluted. However, the number of moles of weak base and conjugate acid available changes.
Henderson-Hasselbach Equation
pH = pKa + log ([base]/[acid])
Buffer capacity
The amount of acid or base a buffer can neutralize before its pH changes significantly.
After adding an equal amount of H3O+ or OH- ions, the concentration ratio and hence the pH changes more for a solution of a low-capacity buffer than a high-capacity buffer.
To cause the same pH change, more strong acid or strong base must be added to a high-capacity buffer than to a low-capacity buffer
Buffer preparation
A buffer is only effective with the concentration ratio is between 0.1-10. Any further outside the range and the buffer can no longer maintain its pH level when a small amount of strong acid or base is added (10 > [A-]/[HA] > 0.1). Therefore, buffer pH range = pKa + or - 1.
Because a buffer is most effective when the component concentration ratio is closest to 1, the best weak acid will be the one whose pKa is closest to that target pH value.
Once the acid is chosen, the Henderson-Hasselbach equation is used to choose the appropriate ratio of [A-]/[HA] that achieves the desired pH.
For most applications, concentrations between 0.05M and 0.5M are sufficient.
Titration
Any reaction considered to be a titration must satisfy three criteria:
Only one reaction can occur between the solutes in the two solutions
The reaction between those solutes must go rapidly to completion (no equilibrium)
There must be a way of signaling the point at which the complete reaction has been achieved in the reaction vessel (equivalence point or stoichiometric point).
Equivalence point
The equivalence point in an acid-base titration occurs in the reaction vessel when the total number of moles of H3O+ from the acid equals the total number of moles of OH- from the base.
where the chem. indicator changes color
Titration considerations
The volume of added solution required to reach the equivalence point depends only on the moles of the acid and base present and the stoichiometry of the reaction.
The pH at the transition point is dependent only on the chemical nature of the indicator and is independent of the equivalence point. The pH at the equivalence point is dependent only on the chemical nature of the reacting species.
A titration should always be repeated as an accuracy check. Trials should agree with each other within 0.02 mL (less than a drop).
Standard solution
A solution of known concentration used when carrying out a titration.
It can be prepared if the solute is a stable, non-delinquescent, soluble compound available in a highly pure form (aka primary standard).
If not available as a primary standard and the solute readily undergoes reaction with atmospheric water vapor or carbon dioxide, the solution must be standardized to accurately determine concentration for use in titration. This is accomplished by titrating the solution in question against a primary standard.
Acidic primary standard examples
potassium hydrogen phthalate (KHC8H4O4): a monoprotic acid often abbreviated simply as KHP
oxalic acid dihydrate (C₂H₂O₄ * 2 H₂O): diprotic acid
Basic primary standard examples
anhydrous sodium carbonate (Na2CO3): accepts two protons in a reaction with an acid
Acid-base indicators
Weak (usually monoprotic) organic acids whose conjugate pairs display different and normally intense colors.
Acidic form and color predominate when [H3O+] is relatively high and the equilibrium favors the reactant side.
Basic form and color predominate when [H3O+] is relatively low and the equilibrium favors the product side.
Intermediate color appears when acidic and basic forms exist in a 1:1 ratio
pH range = pKa + or - 1
Ka and pKa are inversely related; indicator acid strength decreases as we move down the table
First color change
Where [In-]/[HIn] ratio = 0.10
Transition point
Where the indicator is halfway through its color change (intermediate color)
[HIn] = [In-]
Ka = [H3O+] at this point because [In-]/[HIn] ratio = 1
pKa = pH at the transition point
Universal indicator
Several different indicators that each go through a different color change over a different pH range combined into a single indicator solution.
the colors of component indicators at each pH combine to display virtually all colors of the visual spectrum
Titration curve
A plot of the pH of the solution being analyzed versus the volume of titrant added.
Strong acid-strong base titration curves
Acid is strong so initial [H3O+] is high and the pH starts out low. As long as there is excess strong acid in the flask, the pH will remain low and increase only very slowly as the NaOH is added during the titration.
The slow increase in pH continues until the moles of NaOH added almost equal the moles of H3O+ initially present in the acid. When the titration is within one to two drops of the equivalence point, the slope of the curve increases dramatically. The next drop of titrant neutralizes the last of the acid at the equivalence point and then introduces a tiny excess of OH- ions into the flask. At that time, the line becomes almost vertical and the pH rises by six to eight units almost immediately.
Following the steep rise in pH at the equivalence point, the pH then increases slowly as excess OH- is added.
![<ul><li><p>Acid is strong so initial [H3O+] is high and the pH starts out low. As long as there is excess strong acid in the flask, the pH will remain low and increase only very slowly as the NaOH is added during the titration.</p></li><li><p>The slow increase in pH continues until the moles of NaOH added almost equal the moles of H3O+ initially present in the acid. When the titration is within one to two drops of the equivalence point, the slope of the curve increases dramatically. The next drop of titrant neutralizes the last of the acid at the equivalence point and then introduces a tiny excess of OH- ions into the flask. At that time, the line becomes almost vertical and the pH rises by six to eight units almost immediately.</p></li><li><p>Following the steep rise in pH at the equivalence point, the pH then increases slowly as excess OH- is added.</p></li></ul>](https://knowt-user-attachments.s3.amazonaws.com/6cba7262-0a92-4227-b4c8-cf4f02f1494e.jpg)
Four key stages of strong acid-strong base titration curves
Stage 1: pH prior to adding any titrant
starts off low pH due to excess H3O+
Stage 2: pH approximately halfway to the equivalence point
some of the acid has been neutralized by the added base, and the volume has increased
Stage 3: pH at the equivalence point (mol H3O+ = mol OH-)
the titration of a strong monoprotic acid by a strong base will produce a solution with a pH of 7 at the equivalence point because neither of the ions present in the product salt can undergo hydrolysis to affect the pH
because of the large jump in pH that occurs with a single drop of titrant at the equivalence point means that we can choose from indicators with transition points as low as pH 5 and as high as pH 9 for this type of titration
Stage 4: pH beyond equivalence point
solution becomes increasingly basic as excess OH- ions are added
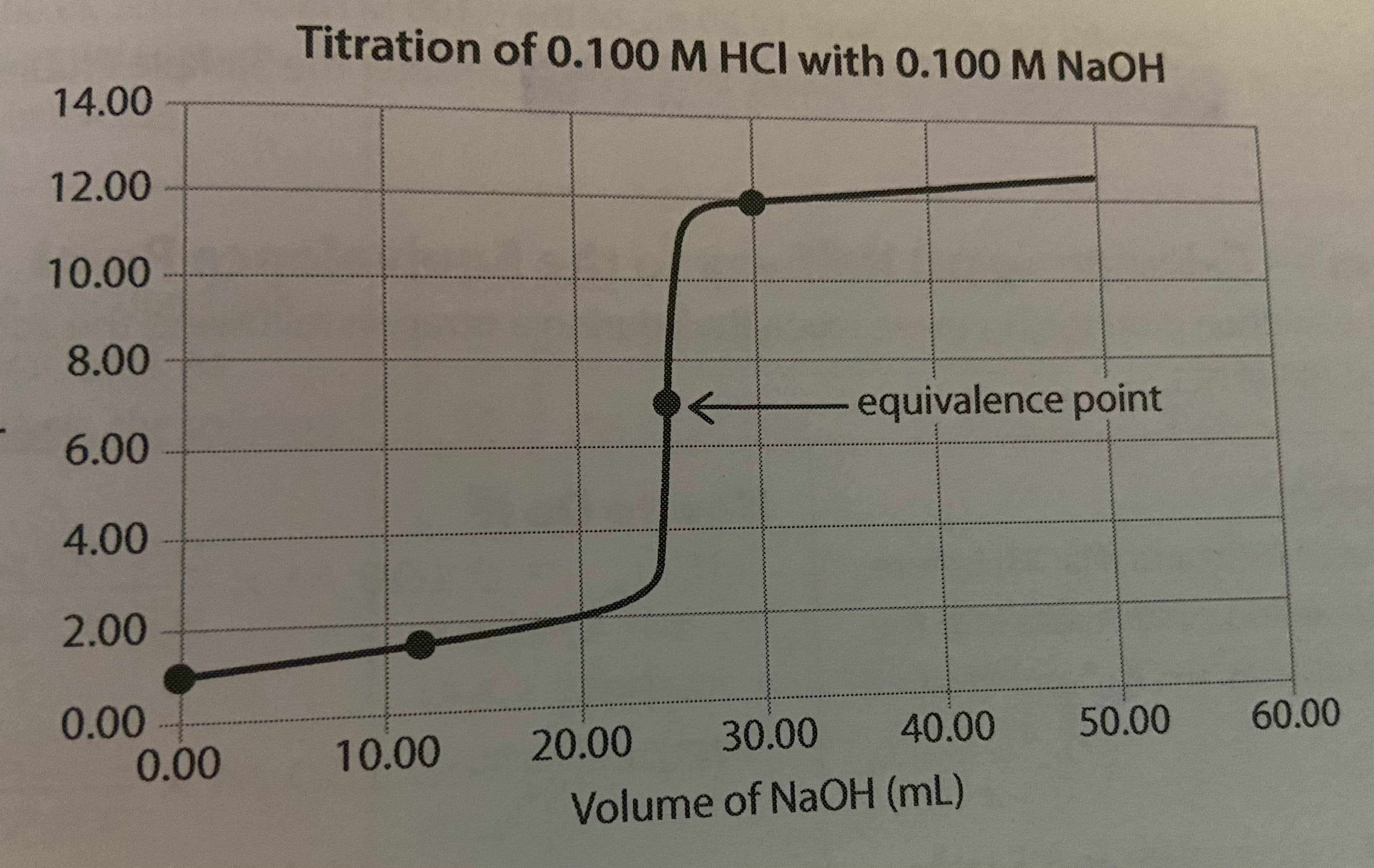
Strong base titrated against strong acid in buret (reverse of strong acid-strong base titration curves)
Titration curve is inverted, but net ionic equation and important features are effectively the same.
Weak acid-strong base titration curves
The predominant species reacting with the hydroxide ion from the strong base is the intact molecular acid
Because the acid is weak and thus only ionizes to a slight extent, the initial [H3O+] is lower and the initial pH is higher than for a strong acid.
There is an initial small jump in pH but then the pH increases more slowly over a portion of the curve called the buffer region just before the steep rise to the equivalence point. However, the pH in this region still changes more quickly than it does for a strong acid-strong base titration. The buffer region occurs because large enough quantities of both the weak acid and its conjugate base exist in the reaction flask.
The steep rise to the equivalence point occurs over a smaller pH range than when a strong acid is titrated with a strong base and the pH at the equivalence point is greater than 7. Even though the anion and cation are present at the equivalence point, the anion is a conjugate base of a weak acid so it hydrolyzes are raises the pH slightly.
Beyond the equivalence point, the pH once again increases slowly as excess OH- is added.
![<ul><li><p>The predominant species reacting with the hydroxide ion from the strong base is the intact molecular acid</p></li><li><p>Because the acid is weak and thus only ionizes to a slight extent, the initial [H3O+] is lower and the initial pH is higher than for a strong acid.</p></li><li><p>There is an initial small jump in pH but then the pH increases more slowly over a portion of the curve called the buffer region just before the steep rise to the equivalence point. However, the pH in this region still changes more quickly than it does for a strong acid-strong base titration. The buffer region occurs because large enough quantities of both the weak acid and its conjugate base exist in the reaction flask.</p></li><li><p>The steep rise to the equivalence point occurs over a smaller pH range than when a strong acid is titrated with a strong base and the pH at the equivalence point is greater than 7. Even though the anion and cation are present at the equivalence point, the anion is a conjugate base of a weak acid so it hydrolyzes are raises the pH slightly.</p></li><li><p>Beyond the equivalence point, the pH once again increases slowly as excess OH- is added.</p></li></ul>](https://knowt-user-attachments.s3.amazonaws.com/bfcbb5a1-dc63-421e-9a43-05a0a63ec485.jpg)
Four key stages of weak acid-strong base titration curves
Stage 1: pH prior to adding the titrant
minimal ionization of the acid means that initial and equilibrium concentrations are approx. equal
Stage 2: pH approximately halfway to the equivalence point
from the beginning of the titration until the equivalence point is reached, the flask contains a mixture of a weak acid and its conjugate base (aka it is a buffer solution)
just before halfway to the equivalence point, the solution pH is always less than the pKa and [H3O+] > Ka
at halfway to the equivalence point, pH = pKa and [H3O+] = Ka
beyond halfway to the equivalence point, pH > pKa and [H3O+] < Ka
Stage 3: pH at the equivalence point
strength of acid does not affect the volume of base required to neutralize it in a titration
the titration of a weak acid by a strong base will produce a basic solution with a pH greater than 7 at the equivalence point because the anion present in the product salt will undergo hydrolysis to produce OH- ions and the cation will not hydrolyze
because the vertical region around the equivalence point is shorter, we are more limited in choosing indicators
Stage 4: pH beyond the equivalence point
solution becomes increasingly basic as excess OH- ions are added
although conjugate base of weak acid already exists in the flask, added strong base will shift equilibrium further left
![<p>Stage 1: pH prior to adding the titrant</p><ul><li><p>minimal ionization of the acid means that initial and equilibrium concentrations are approx. equal</p></li></ul><p>Stage 2: pH approximately halfway to the equivalence point</p><ul><li><p>from the beginning of the titration until the equivalence point is reached, the flask contains a mixture of a weak acid and its conjugate base (aka it is a buffer solution)</p></li><li><p>just before halfway to the equivalence point, the solution pH is always less than the pKa and [H3O+] > Ka</p></li><li><p>at halfway to the equivalence point, pH = pKa and [H3O+] = Ka</p></li><li><p>beyond halfway to the equivalence point, pH > pKa and [H3O+] < Ka</p></li></ul><p>Stage 3: pH at the equivalence point</p><ul><li><p>strength of acid does not affect the volume of base required to neutralize it in a titration</p></li><li><p>the titration of a weak acid by a strong base will produce a basic solution with a pH greater than 7 at the equivalence point because the anion present in the product salt will undergo hydrolysis to produce OH- ions and the cation will not hydrolyze</p></li><li><p>because the vertical region around the equivalence point is shorter, we are more limited in choosing indicators</p></li></ul><p>Stage 4: pH beyond the equivalence point</p><ul><li><p>solution becomes increasingly basic as excess OH- ions are added</p></li><li><p>although conjugate base of weak acid already exists in the flask, added strong base will shift equilibrium further left</p></li></ul>](https://knowt-user-attachments.s3.amazonaws.com/79a49c77-eba4-4e11-a36f-cc31f3f981f7.jpg)
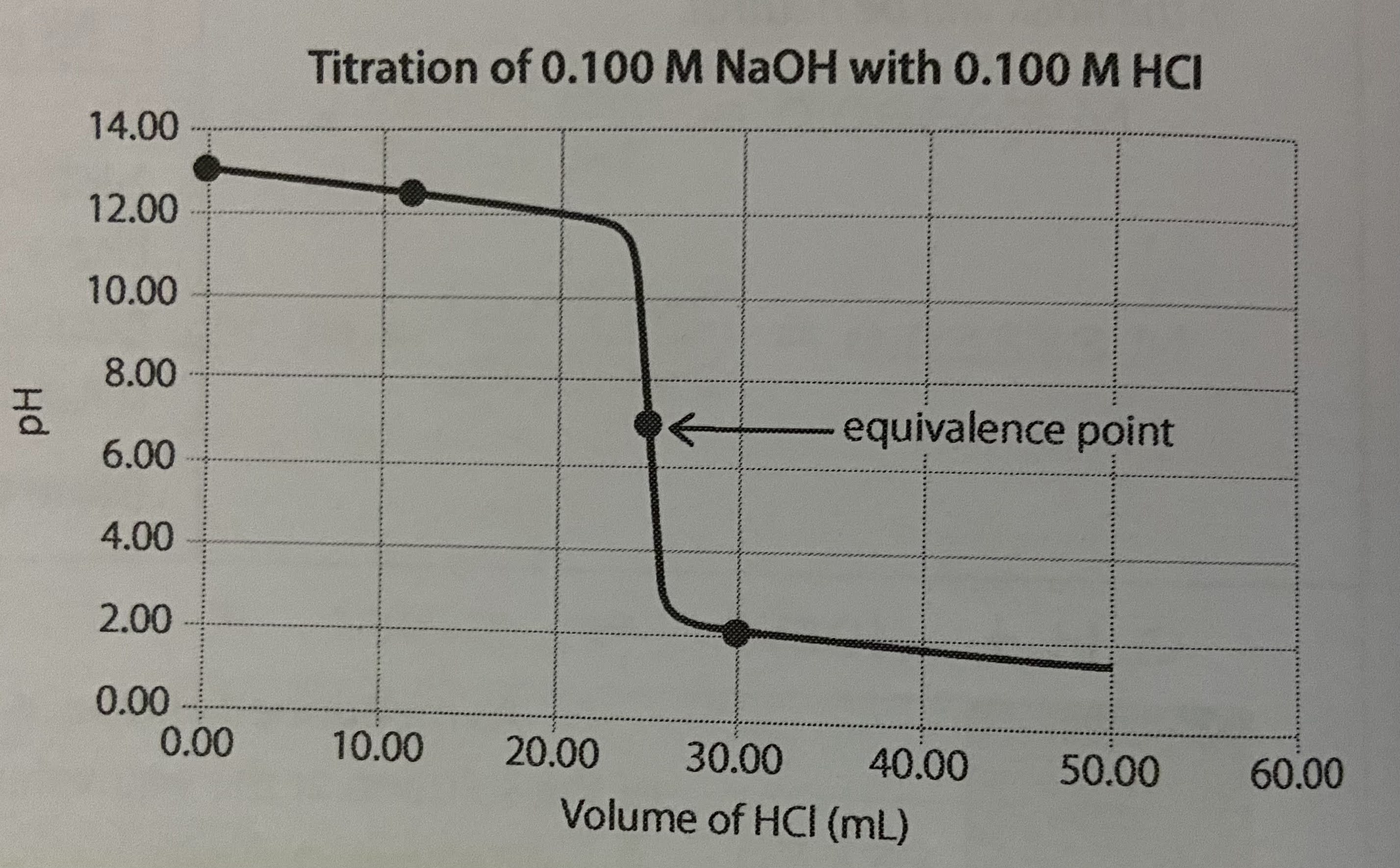
Weak base-strong acid titration curves
Because we begin with a weak base, we expect the initial pH to be above 7 but lower than a strong base
There is an initial small drop in pH as the titration begins but then the pH decreases more slowly over a buffer region during which significant amounts of the weak base and its conjugate acid are present in the flask. However, pH is this region still changes more quickly than it does during a strong base-strong acid titration.
The steep drop at the equivalence point occurs over a smaller pH range than when a strong base is titrated with a strong acid and the pH at the equivalence point is below 7. This is because the cation is the conjugate acid of a weak base so it hydrolyzes and lowers the pH.
Beyond the equivalence point, the pH decreases slowly as excess H3O+ is added
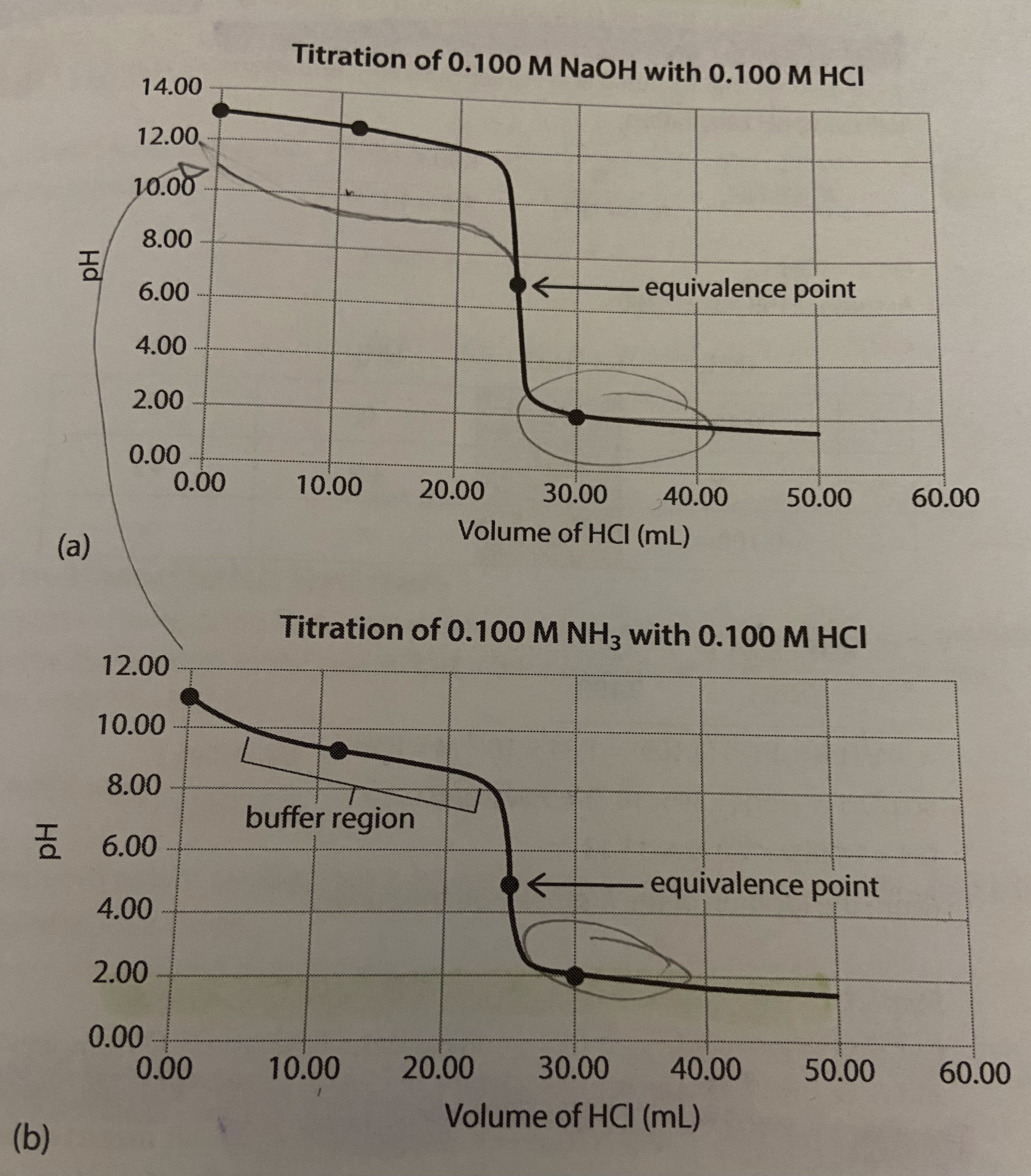
Four key stages of weak base-strong acid titration curves
Stage 1: pH before adding any titrant
pH of a weak base solution is determined given the initial concentration and the Ka value
Stage 2: pH approximately halfway to the equivalence point
from the beginning of the titration until the equivalence point is reached, the flask contains a mixture of a weak base and its conjugate acid (aka a buffer solution)
at exactly halfway to the equivalence point, [H3O+] = Ka and pH = pKa
Stage 3: pH at the equivalence point
all of the base initially present has reacted with the added strong acid
the titration of a weak base by a strong acid will produce an acidic solution with a pH below 7 at the equivalence point because the cation present in the product salt will undergo hydrolysis to produce H3O+ but the anion will not hydrolyze
relatively short vertical region so must be selective with indicators
Stage 4: pH beyond the equivalence point
solution becomes increasingly acidic with addition of H3O+ ions
the presence of the excess H3O+ in the flask is the only significant factor in determining pH (excess HCl added pushes equilibrium further to the left)
Metal oxides in water
Metal oxides formed from group 1 alkali metals and group 2 alkaline earth metals except Be react with water to produce basic solutions
these metal oxides are highly ionic and soluble
these metal oxides referred to as BASIC ANHYDRIDES
metal oxides that are not basic anhydrides are not soluble in water because the oxide ions are locked so tightly in the crystal lattice structure of these compounds that they cannot react with water to generate hydroxide ions
Basic anhydrides in water
dissociate
metal ions are spectators
dissociated oxide ions react with water to convert both itself and the water into hydroxide ions
oxide ion is considered strong base because of its enormous affinity for protons, which means that every oxide ion removes a proton in water
Amphoteric metal oxides
depending on the reaction conditions, they can behave as either acidic oxides or basic oxides
some include BeO, and also Cr2O3, Al2O3, Ga2O3, SnO2, and PbO2
Transition metal oxides
Some transition metal oxides in which the metal has a high oxidation number actually act as acidic oxides
ex. manganese(VII) oxide and chromium(VI) oxide
Acidic anhydrides
non-metal oxides with relatively high oxidation states
ex: SO3, N2O5, Cl2O7
produce acids in water
no oxide ions are released into water, so no hydroxide ions are produced
water molecule binds to molecular oxide to form a molecular acid
Periodic trend in the acid-base properties of the element oxides of the main group elements
In general, as elements become less metallic, their oxides that react with water produce more acidic solutions. This occurs as we move both left to right across a chemical period and bottom to top up a chemical family.
Acid rain
rainwater with pH below 5.3
mostly caused by humankind burning fossil fuels into the air
produced when non-metal oxides react with water in the air to produce acid
Sources of sulfur oxides
combustion of impure coal containing pyrite produces SO2. In polluted air containing ozone, O3, and fine dust particles (especially in the presence of sunlight), oxygen and ozone will oxidize some of the SO2 to SO3. SO3 reacts with water to produce sulfuric acid (strong acid)
Sources of nitrogen oxides
NO gas in air reacts with oxygen gas to produce NO2, which reacts with water in the atmosphere to produce the strong acid HNO3 and the weak acid HNO2
Consequences of acid rain
affects aquatic and terrestrial ecosystems because wind blows acid rain clouds around the area
limestone reacting with acid rain results in bicarbonate/carbonate buffer
Scrubbing
removes sulfuric gases from the atmosphere to prevent/reduce acid rain production