3.2.1.3 Methods Of Studying Cells
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
preparing a temporary mount
1) add a drop of water to the slide
2) obtain thin section of tissue and place on slide
3) stain specimen (e.g. iodine in KI solution) to make structures visible and to add contrast
4) add coverslip using mounted needle at an angle to avoid trapped air bubbles
artefacts - things not part of cell/specimen e.g. dust
reasons for addition of acid to specimen + coverslip needed
-acid = to break down the cell wall to allow the cells to be easily squashed
-coverslip = creates thin layer of cells to allow light to pass through easily, push down firmly for thin layer but do not push sideways to not break chromosomes
optical microscopes
-uses beams of light to magnify the specimen which are reflected back to observer
-image is focused using two glass lenses: objective and eyepiece
-two wheels used: coarse focusing = moves stage up or down, fine focusing = makes the image more clearer/detailed
advs + disadvs of optical
advs: colour image, cheap, shows living structures
disadvs: lower resolution as wavelength of light too long, 2D image
electron microscope
-produces beams of electrons to produce an image on a fluorescent screen
-electrons have a shorter wavelength than light = higher resolution
transmission electron microscopes (TEM)
-produces a beam of electrons which pass through thin section
-structures absorb electrons and appear dark
-image is focused using magnetic lenses
scanning electron microscope (SEM)
-produces a beam of electrons onto the surface of specimens which is then reflected/bounce back onto a detector and produces an image
-image is focused using electromagnetic lenses
advs + disadvs of electron
advs: high resolution, high magnification, smaller organelles can be observed, for SEM - produces 3D image
disadvs: no colour, requires a vacuum (so only use dead/dehydrated), expensive, more complex preparation, requires thinner specimens, for TEM - only 2D image, for SEM - lower resolution than TEM + only outer surface
magnification
the ability to make small objects seem larger
resolution
the ability to distinguish 2 objects from each other
how to measure the size of a structure
1) place micrometer on stage to calibrate eyepiece graticule
2) line up their scales and count how many graticule divisions there are in eyepiece units
3) length of 1 division = length of division in mm / number of eyepiece units
4) use values to work out actual length of structures
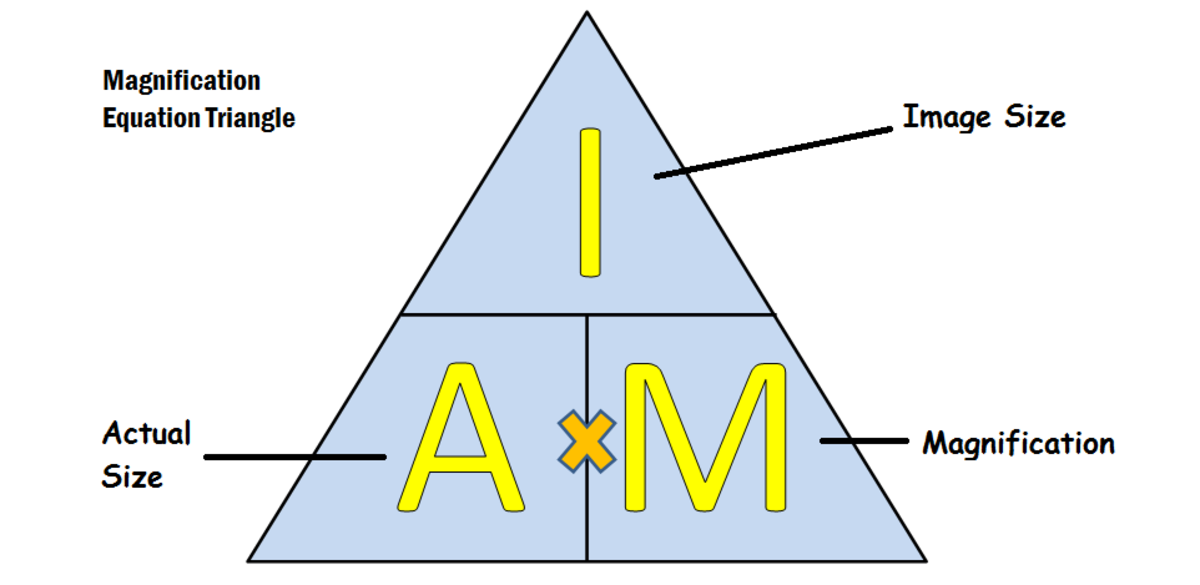
magnification formula
image size / actual size
cell fractionation
1) add tissue to an ice-cold, buffered, isotonic (same water potential as tissue) solution
2) cells are broken open by homogeniser which releases its organelles
3) filter homogenate to remove unbroken cells or large pieces of debris
why doe the solution need to be ice-cold, buffered and isotonic?
-ice-cold = slows enzyme activity to prevent digestion of organelles
-buffered = maintains constant pH so no denatured enzymes
-isotonic = prevents osmosis so no shrinkage of cells
ultracentrifugation
1) tube of filtrate spun at a slow speed in centrifuge
2) the heaviest organelles are forced to the bottom of tube and form a pellet
3) the supernatant (liquid at the top) is filtered into another tube and is spun at a higher speed in second centrifuge
4) the next heaviest organelles form the pellet and process is repeated until each organelle is separated out
heaviest to lightest organelles
nucleus → (chloroplasts if plant) → mitochondria → lysosomes → rER → membrane → sER → ribosomes