Proton Motive Force Old Exam Questions
1/10
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
If you isolate mitochondria and place them in buffer of low pH, they begin to synthesize ATP. Why?
Lowering external pH increases the proton gradient across the inner membrane, increasing the chemiosmotic force by driving the ATP synthase
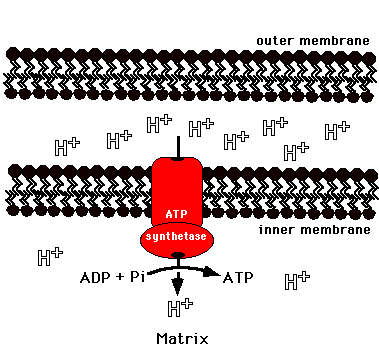
What feature determines the ELECTROSTATIC component of the chemiosmotic potential?
Z (the proton charge)
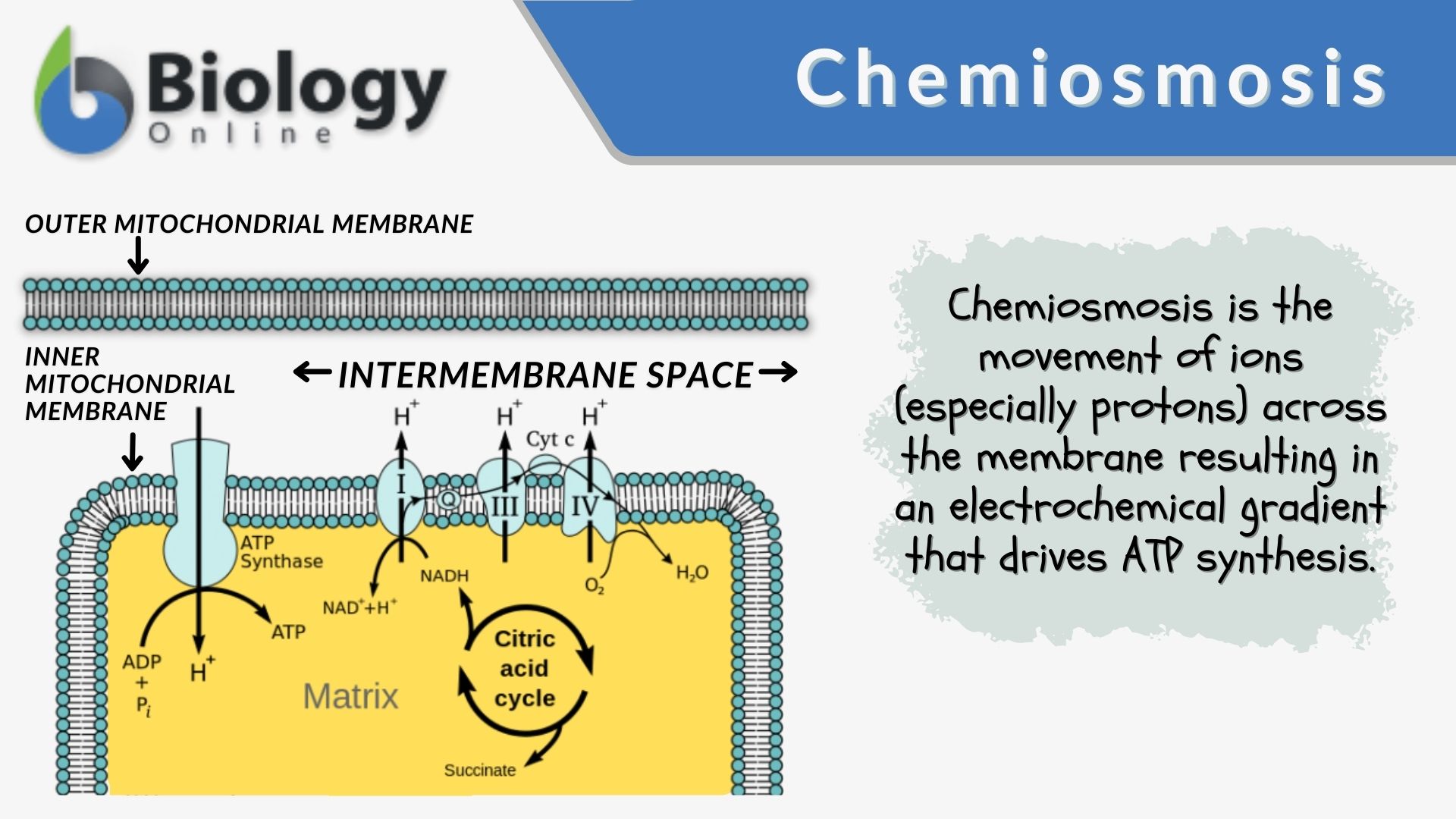
What is the primary function of chemical uncouplers?
They collapse the H+ gradient across the inner membrane
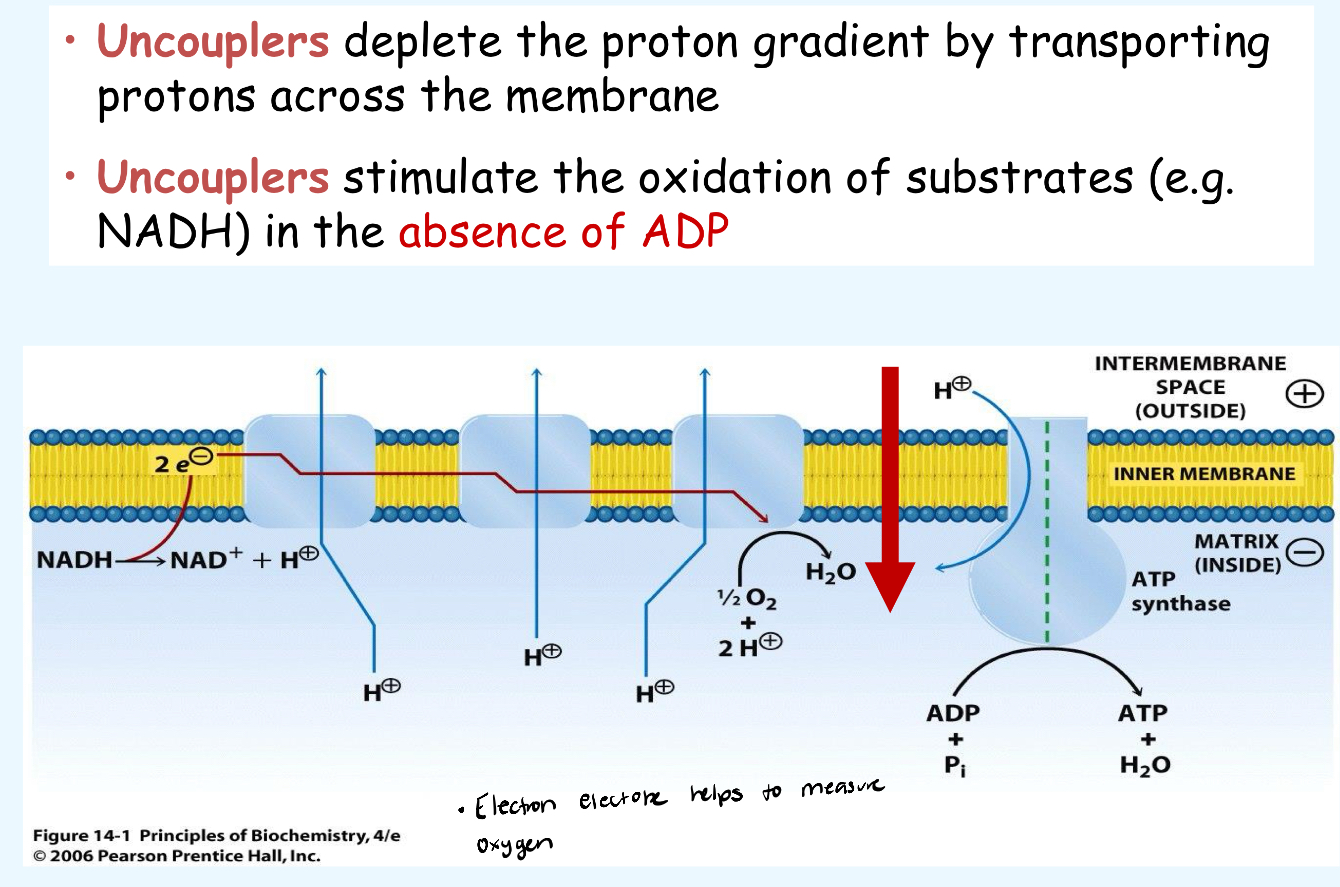
Define the correct P:O ratio:
P:O = (ADP molecules phosphorylation) / (oxygen atoms reduced)

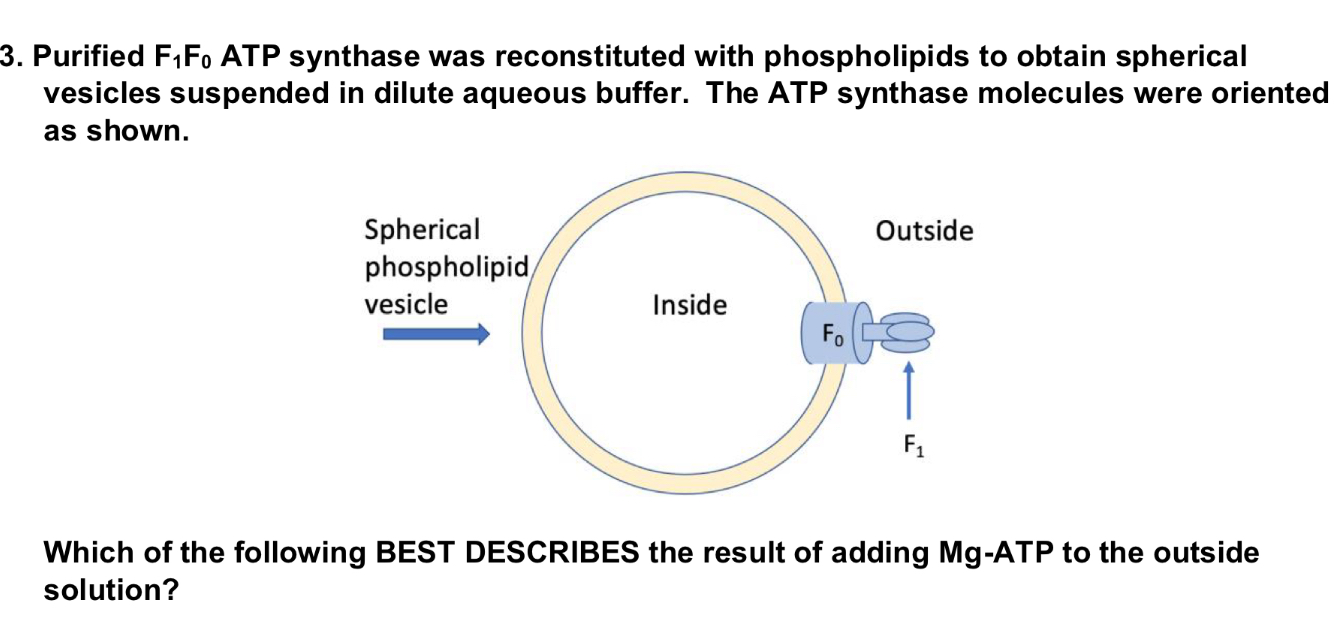
ADP will rise on the outside and the pH will drop on the inside
What are substrates for Complex II?
Succinate and FADH2


What are true statements about the function of the F1F0 ATP synthase?
Each proton binds an Asp residue in an F0 channel
Rotation of F0 drives conformational changes in the F1 structure
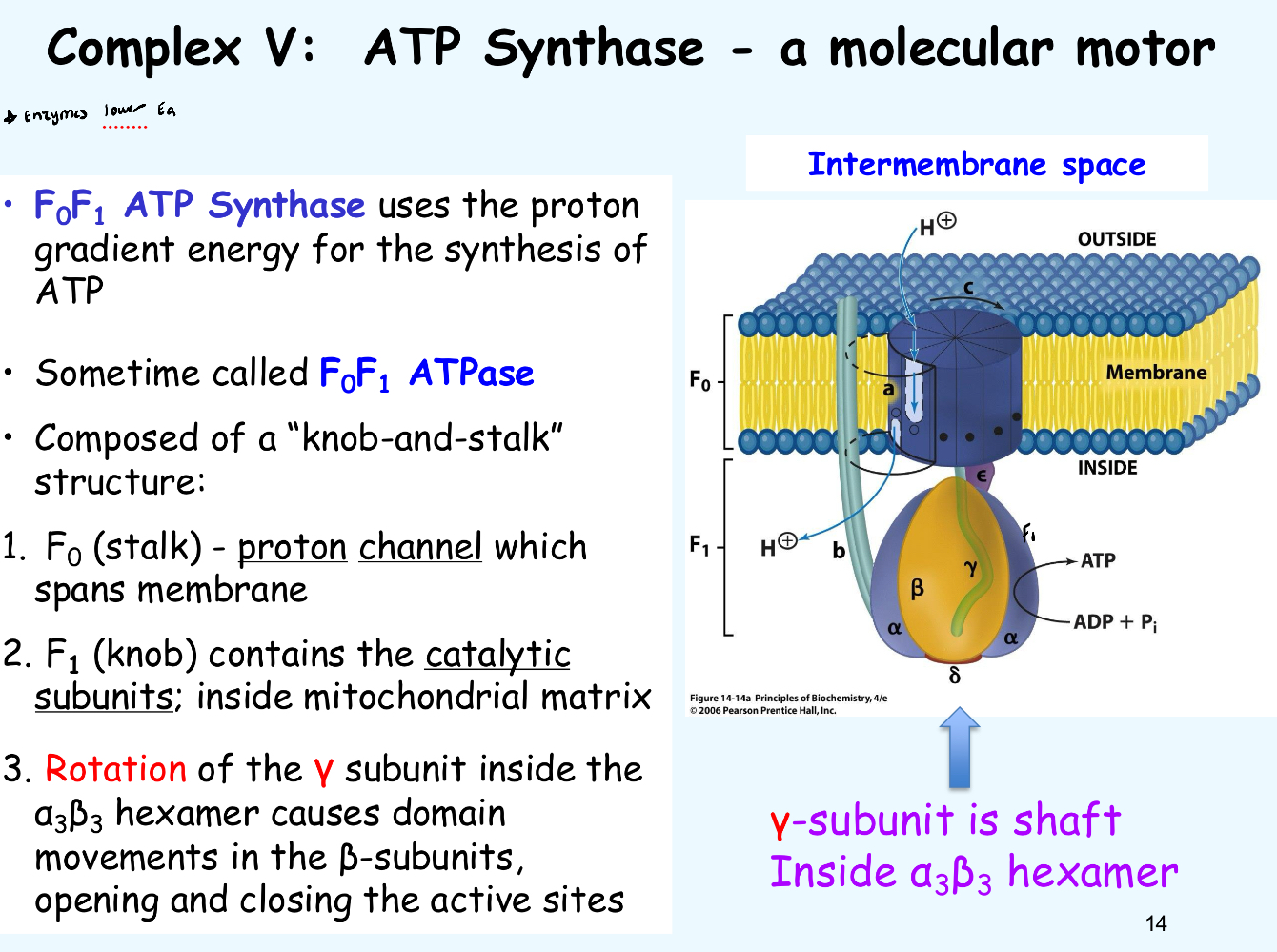

High H+ in the intermembrane space
High ADP in the intermembrane space
High ATP in the matrix

Electron transport coupled to proton export from the matrix
Proton translocation across the inner membrane coupled with ATP synthesis
An intact inner membrane
How many protons are trans located by complex I for each electron pair supplied by NADH?
4

Under aerobic conditions, a mechanism is needed to deliver cytoplasmic reducing equivalents to the mitochondrial electron transport system. What statement correctly describes this process?
NADH is oxidized by cytosolic glycerol-3-P-Dehydrogenase
Ubiquinone is converted to ubiquinol
Dihydroxyacetone phosphate is reduced to glycerol 3-phosphate