Biology OCR A Level A - Biological molecules
1/179
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
180 Terms
What is a condensation reaction?
Reaction that occurs when two molecules are joined together with the removal of water
What is a hydrogen bond?
An intermolecular bond that occurs when two separate molecules of an oxygen and hydrogen of different polarity (delta positive and delta negative) bond to each each other to form the hydrogen bond
What is a hydrolysis reaction?
Reaction that occurs when a molecule is split into two smaller molecules. Requires water
What is a monomer?
A small molecule which bind to other monomers to form a polymer.
What is a polymer?
A large molecule made from many smaller molecules called monomers
What is a covalent bond?
An electrostatic attraction between the nuclei of both atoms and the shared electron pair(s). Only can occur between two non-metals.
What elements do carbohydrates contain?
carbon, hydrogen, oxygen
What is the only difference between alpha and beta glucose?
On alpha glucose, the H is above the ring on carbon 1
On beta glucose, the H is below the ring on the carbon 1
What elements do lipids contain?
carbon, hydrogen, oxygen
What elements do proteins contain?
Carbon, hydrogen, oxygen, nitrogen (sometimes sulphur)
What elements do nucleic acids contain?
carbon, hydrogen, oxygen, nitrogen, phosphorus
What are the properties of water?
- cohesive (hydrogen bonds) and therefore provides surface tension.
- Low viscosity (flows easily)
- high specific heat capacity
- high heat of vaporization
- less dense as a solid than a liquid
What does water being cohesive mean?
There is surface tension and therefore:
- columns of water in plant vascular tissue are pulled up the xylem tissue from the roots. Allowing efficient transpiration
- Insects like pond-skater can walk on water
What does water being liquid at room temperature mean?
- habitats for living things in rivers, lakes and seas occurs
- Major component of tissues in organisms
- reaction medium for chemical reactions, such as in blood
- Provides an effective transport system
Why is it important that water is a good solvent?
- Water is polar, therefore;
- Molecules and ions can move around and react together in water.
- Molecules and ions can be transported in water (helps organisms with things like blood)
Why is water's density unique?
Wates is less dense as a solid (ice) than a liquid (water), therefore;
- Organisms (such as a frog) can be buoyant in water
-aquatic organisms have a stable environment to live in underneath ice
-Ice acts as insulation for the temperature below, promotes life
Why is a high specific heat capacity of water important?
Living organisms need a stable temperature as they depend on enzyme controlled reactions to happen.
Organisms can provide homeostasis of temperature with relative ease
Why is water's latent heat of vaporisation important?
- Water requires a lot of energy for it to evaporate because of the hydrogen bonds
- therefore an organism can rid of excessive heat by expelling it through evaporation of water
- great cooling effect during sweating
What is a carbohydrate?
a group of molecules containing C, H and O. They can form hexoses and pentoses.
What is a glycosidic bond?
when two monosaccharides are joined from their carbons by an oxygen atom.
What are the differences between hexose and pentose sugars?
Pentose sugar is a five carbon sugar
Hexose sugar is a six carbon sugar
Is ribose a pentose or hexose sugar?
Pentose sugar
Is glucose a pentose sugar?
No, glucose is a hexose sugar
What are the differences between lactose and maltose?
1. Lactose contains only glucose and galactose monomers (maltose are made of just glucose)
2. Lactose contains beta glucose, maltose contains alpha glucose
3. Lactose has flipped (180 degrees) of each monomer, maltose has its monomers in the same oreintation.
Similarities between lactose and maltose?
(Lactose molecule in picture)
- Both contain OH groups
- Both joined by a 1,6 - glycosidic bond
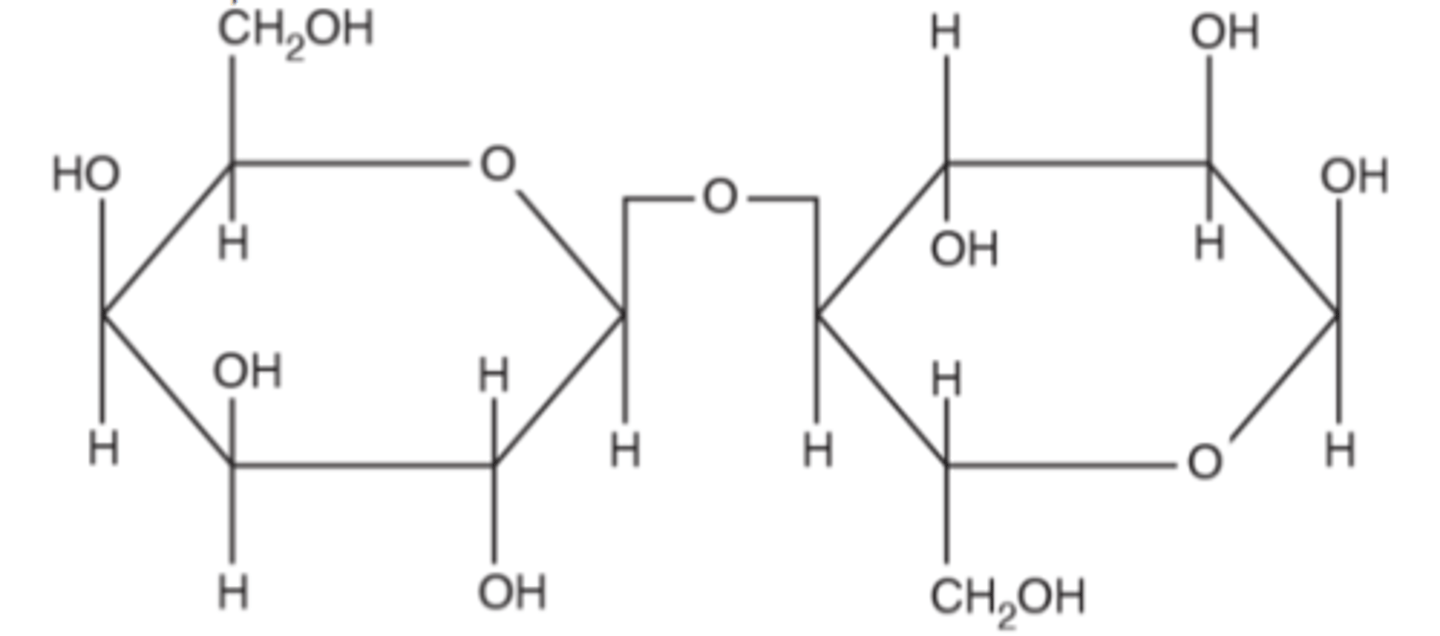
Is maltose a polysaccharide or disaccharide?
Disaccharide. Therefore cannot be regarded as a polymer.
Is starch a polysaccharide or disaccharide?
Polysaccharide.
How does the structure of galactose relate to it's function as a respiratory substrate?
- Contains hydrogen carbon and oxygen elements; contains high calorific value of energy for respiration
- Contains OH groups, soluble in water
What is the main store of polysaccharide in animals?
Glycogen
What is the main store of polysaccharide in plants?
Starch
What is starch formed of?
Amylose and amylopectin.
Why is glycogen and starch particularly good polysaccharides for storage of energy? (properties)
They are both;
- compact, occurring in dense spaces in the cell
- There have branches on each polysaccharide that can be hydrolysed for readily available respiration
- They do not affect the water potential and therefore are osmotically inactive
Which enzyme hydrolyses the 1-4 linkages in starch?
amylase
Which enzyme hydrolyses the 1-6 glycosidic linkages?
glucosidase
In terms of solubility, are polysaccharides more or less soluble in water than monosaccharides?
polysaccharides are less soluble in water than monosaccharides
Why, apart from being a bigger molecule than monosaccharides, are polysaccharides less soluble in water?
- Their polar functional groups that could form hydrogen-bonds are hidden away in the molecule
- Amylose molecule can sometimes form a double helix presenting a hydrophobic external surface
Overall, why is it important that polysaccharides such as glycogen and starch are not soluble in water?
As then they will not affect the water potential of the solution, therefore no osmosis would occur, therefore they will not disrupt normal workings of the cell
What bonds does amylose have?
1-4 glycosidic bonds, made of alpha glucose
What is the detailed structure of amylose?
1) Coiled spiral shape, held by hydrogen bonds
2) hydroxyl groups (on carbon 2) are inside the coil allowing
- the property of reduced solubility
- hydrogen bonding to form internally (helps strengthen coil structure)
What bonds does amylopectin have?
1-4 and 1-6 glycosidic bonds
What is the detailed structure of amylopectin?
As amylose, amylopectin also has a coiled shape but has with branches emerging from the spiral
Held together with hydrogen bonds
What bonds does glycogen have?
1-4 and 1-6 glycosidic bonds
What is the detailed structure of glycogen? (relative to amylopectin)
- 1-4 bonded chains are smaller relative to amylopectin, less tendency to coil (less spiral structure)
- Many more branches relative to amylopectin therefore more easily hydrolysed
- More compact to amylopectin
Do cellulose spiral or are do they lie side by side?
Cellulose molecules lie side by side
Why does cellulose not spiral?
They hydrogen and hydroxyl group on carbon 1 are inverted. Every other beta glucose is rotated by 180 degrees, stops the spiralling
What other force (due to hydroxyl groups) in cellulose help with support?
Hydrogen bonding between Beta molecules between chains provides additional strength to the bonding
How can the chain form hydrogen bonds with each other?
There is a hydroxyl group sticking out on carbon 2; allows bonding with other chains of cellulose.
Do cellulose molecules branch?
NO.
What are four properties of cellulose that make it suitable as plant walls?
- Unreactive/inert molecule
- High tensile strength due to criss crossing
- Insoluble in water; doesn't affect the water potential of the cell (osmotically unreactive)
- Prevents pathogens from entering the plant cell
How many cellulose chains are required to from a microfibril?
60 - 70 cellulose chains
How large are microfibrils in diameter?
10 nm - 30nm
How many microfibrils are needed to form a macrofibril?
Up to 400 microfibrils are needed to from a macrofibril
How are macrofibrils layered?
Embedded into pectins forming the plant cell walls.
One extra feature of macrofibrils?
Some microfibrils runs in all directions to add to high tensile strength for the macrofibrils and therefore the cellulose cell wall.
Why is it difficult to digest cellulose?
Glycosidic bonds between glucose molecules are less easy to break
Reiterate the three ways cellulose is adapted to having a high tensile strength
1. Original strength of cellulose due to glycosidic bonds
2. various hydrogen bonding
3. Macrofibrils runs in all different directions therefore there is a criss-crossing and extra strength
What do these reiterations help the cell perform? How do they link to the plant?
- High tensile strength helps the cells from bursting due to being turgid.
* Turgid cells press against each other, giving more strength to the plant
- Macrofibril structure can be reinforced with other substances, i.e. lignin for waterproofing xylem cell, come cells reinforced to have extra support
* Gives the plant even more support
What are between the macrofibrils that allows water and minerals to diffuse in and out of the cell
Gaps in the macrofibrils
What are bacterial cell walls made of? How do they form?
Peptidoglycan, lying in parallel, cross linked chains by peptide bonds.
What are insects and crustaceans exoskeletons made of?
Chitin
What monomer is used to form cross links between these chains?
Acetylglucosamine
What are lipids?
Groups of substances that are soluble in alcohol instead of water despite having -OH groups
What is a macromolecule?
A molecule with a large number of atoms
What does three compounds do phospholipids contain?
A phosphate group, a glycerol and two fatty acids
Three most important lipids? (Relative to you getting that A*)
Triglycerides, steroids and phospholipids
What is the Glycerol structure (IUPAC structure)?
1,2,3 - propantriol
(This will only help people to remember if those people do chemistry as an a-level)
What is the definition of unsaturated?
An organic molecule containing at least one double carbon-carbon bond
Definition of saturated?
Organic molecule containing no carbon-carbon double bonds
Being unsaturated means what to the molecule
There is a kink, therefore the molecule is more fluid
What is an ester bond?
Carbon to oxygen to carbon bond, formed via a condensation reaction between lipids
What are the five functions of triglycerides? Explain each function.
- Buoyancy; triglycerides are less dense than water therefore provide buoyancy
- Protection; triglycerides provide protection to important organs, i.e. being a shock absorber
- Insulation: Acts as thermal insulators. Lipids in nerve cells acts as electrical insulators
- Energy store: Does not affect water potential therefore can be stored as an energy store.
- Energy source: Give approx. twice amount of energy than 1g carbohydrates. Used in respiration as a respiratory substrate to form pyruvate and acetyl-group for the Krebs cycle.
Are lipids less or more dense than proteins? How does this relate to buoyancy?
Less dense than proteins, therefore in water they are more buoyant.
Difference between phospholipids and triglycerides?
Triglycerides have three fatty acid tails and a glycerol.
Phospholipids have only two fatty acids tails, a glycerol and a phosphate head group.
How do phospholipids behave in water?
Hydrophilic heads (phosphate ion) with hydrophobic tails. Amphipathic
What are micelles?
Where the hydrophilic head of phospholipid faces outwards and hydrophobic tails faces inwards
What does the micelle structure allow for in cells?
Formation of the phospholipid bilayer
Features of the phospholipid bilayer? (micells)
- Hydrophilic heads face outwards towards the water and hydrophobic tails repel against water facing inwards. There is therefore some stability in the bilayer staying intact.
- Membrane is selectively permeable to small and non-polar substances to diffuse through the phospholipid bilayer.
Is the lipid steroid cholesterol made from glycerols and fatty acids?
No, they are made from isoprene groups
What is the use of cholesterol for the cell membrane?
Regulates the fluidity of the cell membrane and resists changes in temperature, acting as a buffer
Where is cholesterol mainly made in animals?
The liver
What is the exact step by step process of how cholesterol works in cell surface membranes of the human body
- Cholesterol binds to phospholipid fatty acid tails
- Increases the packing of the membrane
- Reduces the fluidity of the membrane
What term is used for a derivative of cholesterol in plants?
Stigmasterol
How is stigmasterol different from normal cholesterol?
There is a double bond in stigmasterol between carbon 22 and 23
What are amino acids?
Monomers of all proteins with the same general formula
What is a peptide bond?
A Nitrogen to carbon bond. Formed through condensation of NH2 and COOH to the removal of water
Three Uses of Proteins?
-Synthesis of hormones, enzymes, antibodies, plasma and muscle proteins, haemoglobin, and most cell membranes
-If needed, can be broken down to an acetyl group as a source of energy for ATP production
-If needed, can be broken down and converted to glucose (gluconeogensis)
What is the primary structure of a polypeptide(s)?
The sequence of amino acids found in a molecule
What is the quaternary structure of a protein?
If there is more than on polypeptide chain formed
What is the secondary structure of a protein?
- Coiling of an amino acid chain.
- Folding of an amino acid chain.
How is the secondary structure of a polypeptide dictated?
Dictated by the hydrogen bond formation between different parts of the chain.
What are the two main secondary structures?
Alpha helix or beta pleated sheet
What is the tertiary structure of a protein?
Overall 3D structure of a protein, including the different bonds that could occur
What types of bonding could occur within the tertiary structure of a protein?
- Ionic bonds
- Disulphide bridges
- Hydrogen bonding
- Hydrophilic/hydrophobic interactions.
In the primary structure what are the two main important components of the sequence of amino acids?
1. Number of amino acids
2. the order of amino acids
How many amino acids are there?
There are 20 different amino acids.
How many amino acids per 10 turns of an alpha helix?
There are 36 amino acids per 10 turns of the alpha helix.
How is an alpha helix presented in an amino acid?
Alpha helix twists.
How is the alpha helix stabilized?
The hydrogen bonds between the CO and the NH group
How is a beta pleated sheet formed?
Zig-zag structure. Held together by hydrogen bonds between the -NH group and the -CO group. Beta-pleated slightly folds
Why are hydrogen bonds weak but can form strong structures?
As as a single unit, hydrogen bonds are weak, however with many many formed, hydrogen bonds are collectively very strong.