cpat module 1
1/46
Earn XP
Description and Tags
inflammation, neuroinflammation, immunology
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
47 Terms
acute inflammation major cell type
neutrophils
chronic inflammation major cell type
monocytes / macrophages
inflammation can be the
cause of disease rather than the pathogen directly
acute injury blood test
high leukocytes
increased proportion of neutrophils (neutrophilia)
healing depends on
the degree of injury
no bacterial entry = fastest
bacteria = minutes to days
tissue damage = weeks
host defences against micro organisms
barriers → epithelial, secretions, preformed non specific effectors
activation of immune systems - macrophages, phagocytosis, complement
antigen movement
switch to adaptive immunity
switch from. preformed non specific innate effectors to macrophage and phagocytosis activation
takes more than 6 hours
after macrophage activation, antigen drains to lymph nodes, further exposure to NK cells and macrophages
humoral and cellular immunity activation
roles of inflammation
defence against micro organisms
elimination of damaged cells, inanimate foreign particles
initiation of healing processes (early problems lead to long healing impairment)
inflammation
the reaction of vascularised living tissues to local injury or infection, characterised by the movement of fluid and leukocytes from the blood into the affected tissue
acute inflam
rapid onset
relatively short lived
sterotypic response to injury or infection
movement of fluid and neutrophils out of the blood and into affected tissue
can evolve to chronic, but its rare
chronic inflam
prolonged inflammation due to injury or infection
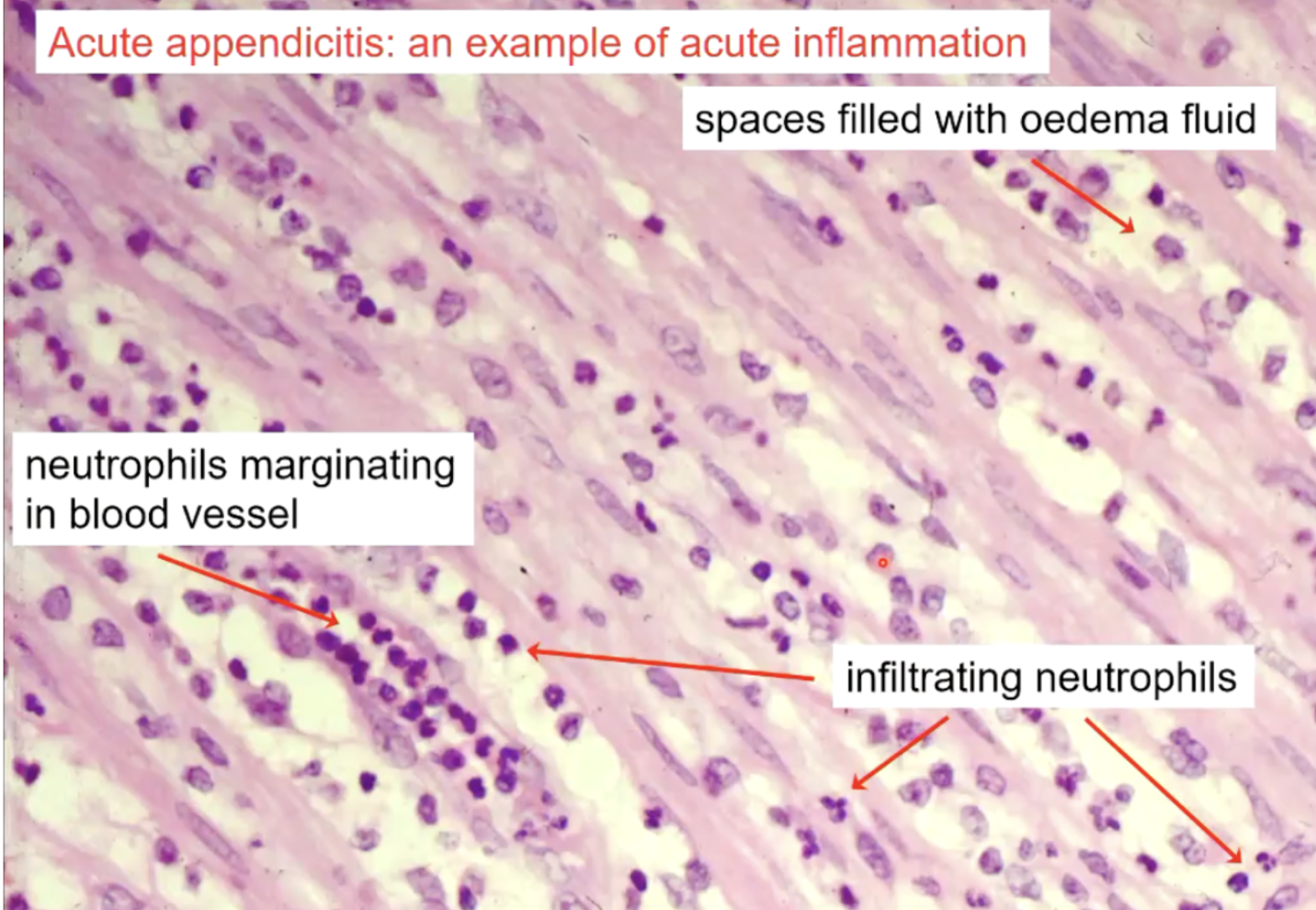
appendicitis histology
acute inflammation
spaces filled with oedema fluid
neutrophils marginate in blood vessels, they also infiltrate the tissue
positives of inflammation
promotes host survival
initiates adaptive and innate immunity
host defence against micro-organisms
initiates healing
negatives of inflammation
deleterious effects on host survival
acute - perforation of wall can kill patients, meningitis (intra cranial pressure)
chronic - tissue destruction in TB, incapacitation in arthritis, initiates cancer
major cell types in inflammation
neutrophils - kill pathogen
endothelial - regulates movement of proteins from blood into tissues
macrophages - degrade fibrin and debris, kill pathogen, secrete cytokines
fibroblasts - secrete collagen for healing
acute inflammation can occur in response to
sterile injury
injury with infection
infection without injury
in all cases, processes are the same
cardinal signs of inflammation
redness
heat
swelling
pain
as described by aulus celsus
these are visible/tangible signs of the physiological response to injury
heat + redness
occurs due to increased blood flow (hyperaemia)
redness aka erythemia
swelling
due to fluid movement from blood into tissue (exudation), movement of cells
pain
mediated by pain receptors
increasing sensitivity of pain receptors = hyperalfesia
all inflam mediators, can reset the sensitivity level of the pain receptors
Oedema
accumulation of fluid extra vascularly in tissues
Exudate
oedema fluid with high protein content, resulting from increased endothelial permeability to plasma proteins in inflammation
pus
inflammatory exudate containing viable and dead neutrophils, cell debris, viable and dead micro-organisms, protein, lipid, DNA
hyperaemia
Hyperaemia is the passage of larger-than- normal volumes of blood through a tissue (vasodilation)
Not specific to inflammation – e.g. usually occurs during exercise
Hyperaemia is essential in inflammation – for the formation of the exudate
increased RBCs
Hyperaemia in acute inflammation
Heat” and “redness” are consequences of hyperaemia.
Hyperaemia is an increase in the amount of blood flowing through an area of tissue.
Hyperaemia results from:
vasodilatation of the pre-capillary arterioles
opening up of dormant capillaries to the passage of blood
Vasodilation causes hyperaemia in acute inflammation
Locally produced vasoactive mediators of inflammation act on arteriolar smooth muscle cells.
Relaxation of the smooth muscle cells allows the arteriole to dilate.
More blood is able to enter the microvascular bed.
Capillaries that were “dormant” (filled with plasma) start to carry blood
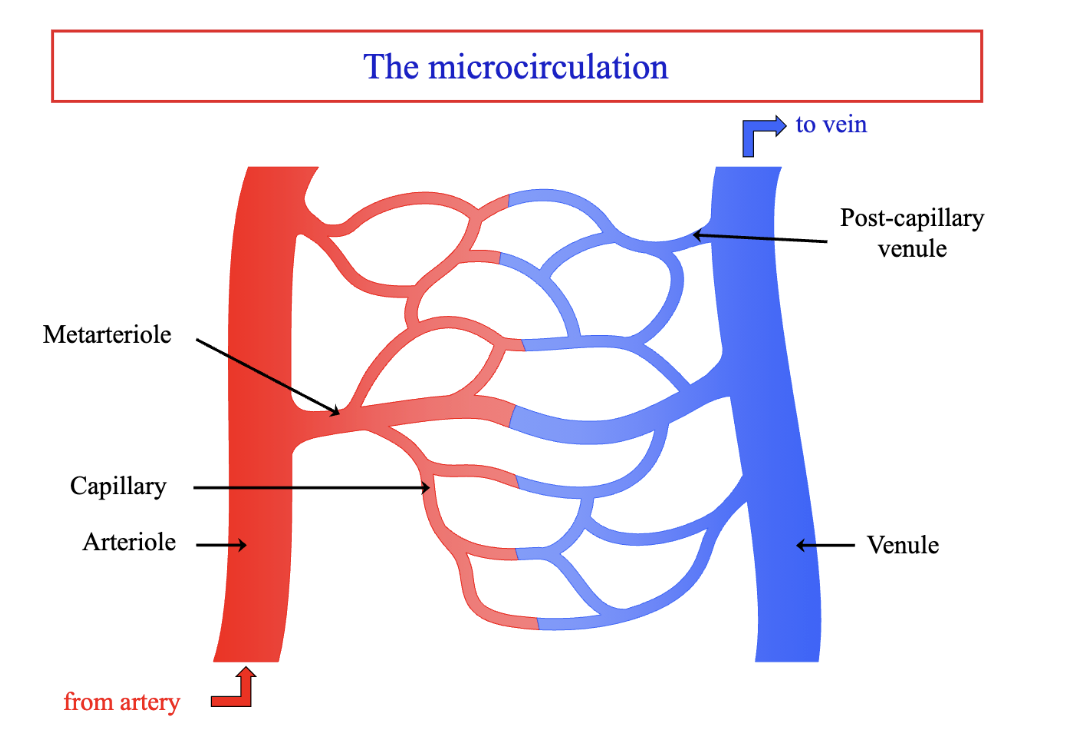
exudation
due to dramatic increase in permeability of endothelium.
After initial hyperaemia, fluid (containing plasma proteins) moves from the blood into the affected tissue.
Increased vascular permeability to proteins must happen for exudation to occur.
The fluid movement leads to slowing of blood flow in the affected area. The vessels appear “congested.”
Swelling results from the increased fluid content of the tissue.(macroscopic)
Accompanying the fluid movement is emigration of neutrophils into the affected tissue
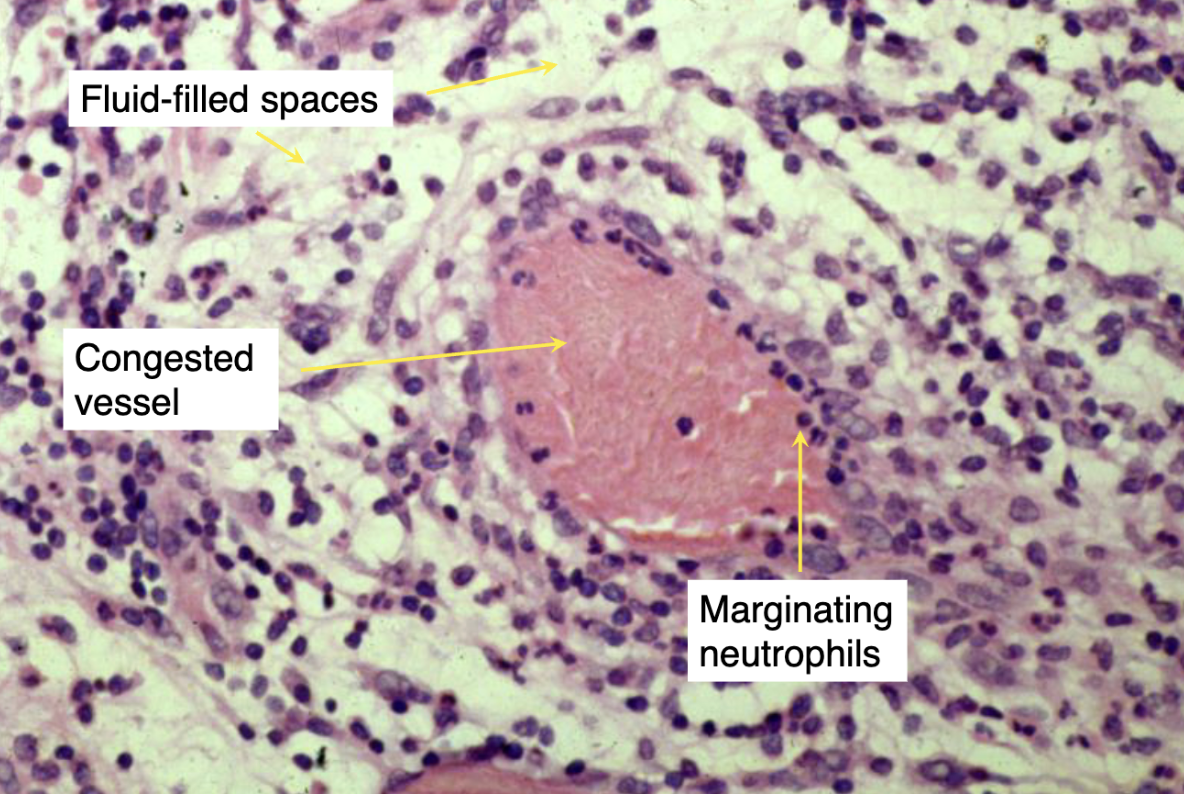
how does exudation occur?
Mild or moderate acute inflammation: Through inter-endothelial gaps in post-capillary venules only (contraction of endothelial cells).
Two patterns:
immediate but transient
delayed and prolonged
more severe = damage to endothelial of all microvessels - immediate prolonged response
forces at the vessel walls
hydrostatic P → pushes out of the vessel
osmotic P → pushes into the vessel
normally → balance between these forces allows control of fluid movement
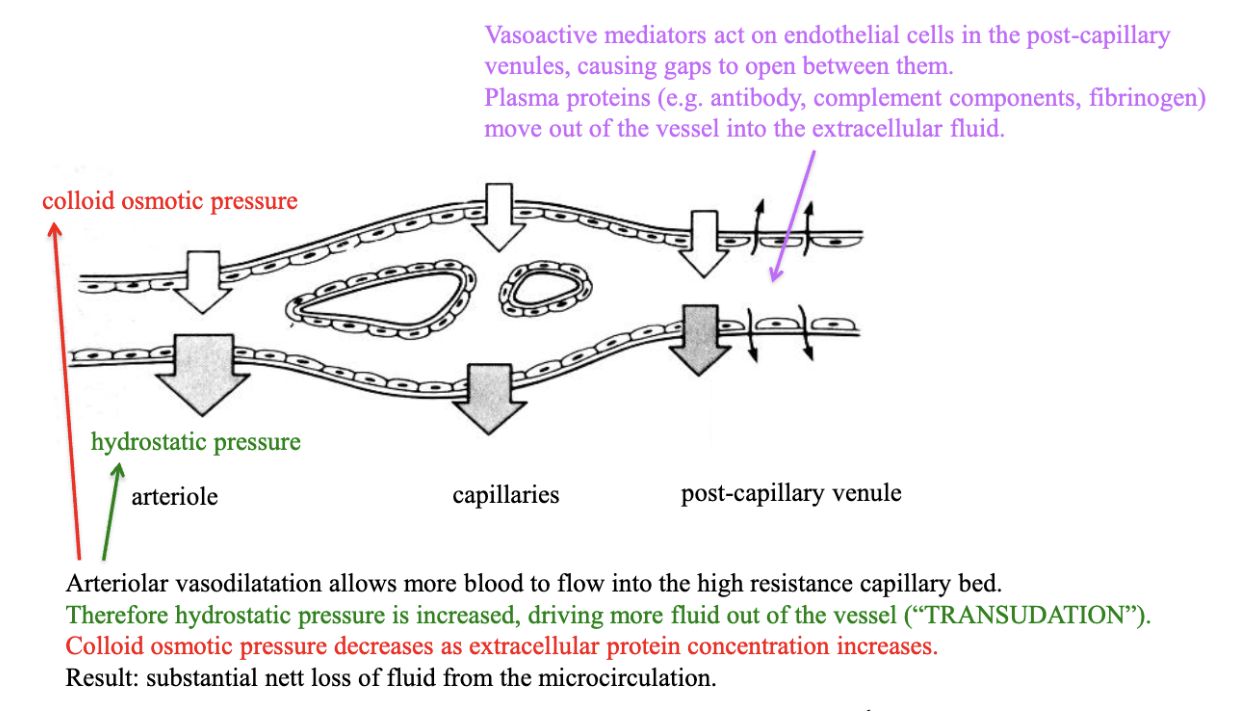
movement of fluid in acute inflammation
artiole → vasodilation allows more blood flow, hydrostatic pressure increased which drives fluid out of the vessel (transudation). Colloid osmotic pressure decreases as EX protein conc increases. — substaintial net loss of fluid from microcirculation
post capillary venule - vasoactive mediators act on endothelial cells in the venules, causing gaps to open between them. Plasma proteins (eg, antibodies) move out of vessel into EX fluid.
Role of Exudation
fluid → dilutes toxins, increases flow into lymphatics
plasma proteins → antibody, complement components, fibrin, all moved into tissue for repair
neutrophils moved to destory micro-organisms
types of inflammatory mediators
vasoactive
chemotactic
vasoactive mediators
increase blood flow and vascular permeability to protiens
aka cause exudation
chemotactic mediators
recruit and stimulate inflammatory cells
results in either acute or chronic inflammation
ex, vasoactive mediators
Amines - histamine
Lipid-derived mediators • prostanoids • leukotrienes
Plasma-derived mediators • complement fragments 3a and 5a (“C3a and C5a”) • kinins
histamine source
mast cell degranulation
platelets
role of histamine in inflammatory response
vasodilation of arterioles
increase vascular permeability in post capillary venules
NB: there are a number of clinical anti-histaminic drugs, but these have little anti-inflammatory activity.
formation of lipid derived mediators of inflammation
membrane phospholipid phosphilpase A2 is activated by inflammatory stimuli
→ arachidonic acid -> cyclo-oxygenases or lipoxygenases
cyclo-oxgenases → prostanoids
lipooxygenases → leukotrienes
corticosterooids block
phospholipase A2, so development of prostanoids and leukotrienes
NSAIDs block
cyclo-oxygenases So prostanoids
zileuton blocks
lipooxygenases so leukotrienes
summary of vasoactive mediators
vasodiltion → histamine, kinins, prostaglandin E2 and I2
increased vasc perm → histamine, Kinins, C3a, C5a, leukotrienes B4 and C4
sensitisation of pain receptors
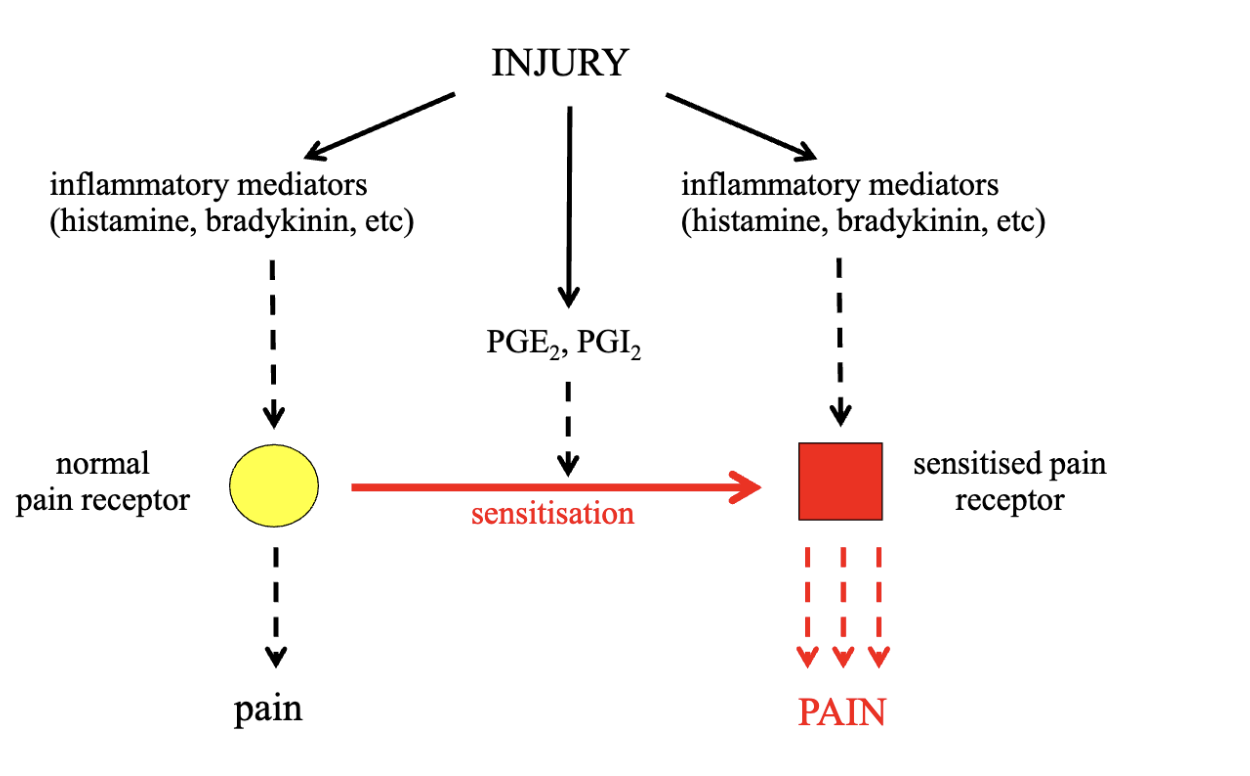
done by prostaglandins