3.3.1 Organic chemistry intro
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
What is structural isomer?
A compound which has the same molecular formula but a different structural formula.
What is stereoisomer?
A compound which has the same structural formula but the atoms are arranged differently in space
What is the empirical formula?
Simplest ratio of atoms of each element in a compound
What is the molecular formula?
Actual number of atoms of each element in a compound
What is a homologous series?
Family of compounds which contain the same functional group and so have similar chemical properties
What is a hydrocarbon?
A compound containing hydrogen and carbon only
What does saturated mean?
A compound which contains only single covalent bonds
What does unsaturated mean?
A compound which contains one or more double covalent bonds
What is organic chemistry the chemistry of?
Carbon-containing compounds (except CaCO3, CO2, CO)
How is carbon chemistry extensive?
There are well over ten million compounds containing carbon and hydrogen, due to carbon being able to form rings and very long chains which may be branched. Carbon forms covalent bonds
Click the image showing the different ways in which hexane can be presented

Click the image showing the details of the following homologous series you need to know.
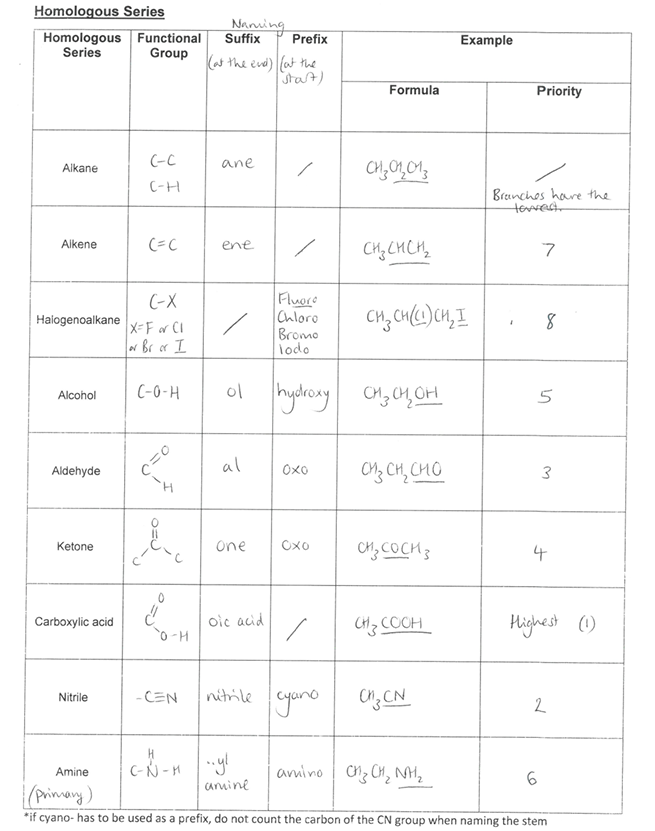
What is Nomenclature?
System of naming compounds, with the names consisting of 3 main parts:
The stem: how many carbons there are in longest chain bearing functional group
The prefix (before stem): position of side chains / functional groups. Side chains are often alkyl groups. An alkyl group is an alkane with a hydrogen atom removed. A general alkyl group is denoted by the letter R
The suffix (after stem): functional group present e.g. ANE, ENE, OL, OIC ACID

What is the general formula of an alkane?
CnH2n+2
Can have unbranched or branched chains
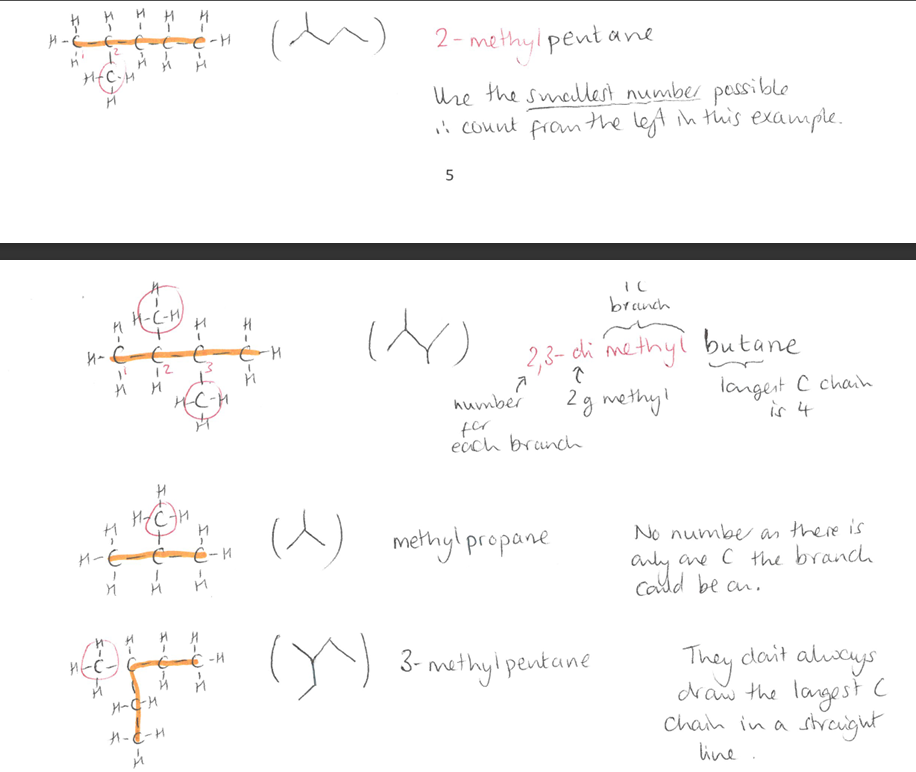
Give examples on naming branched alkanes.
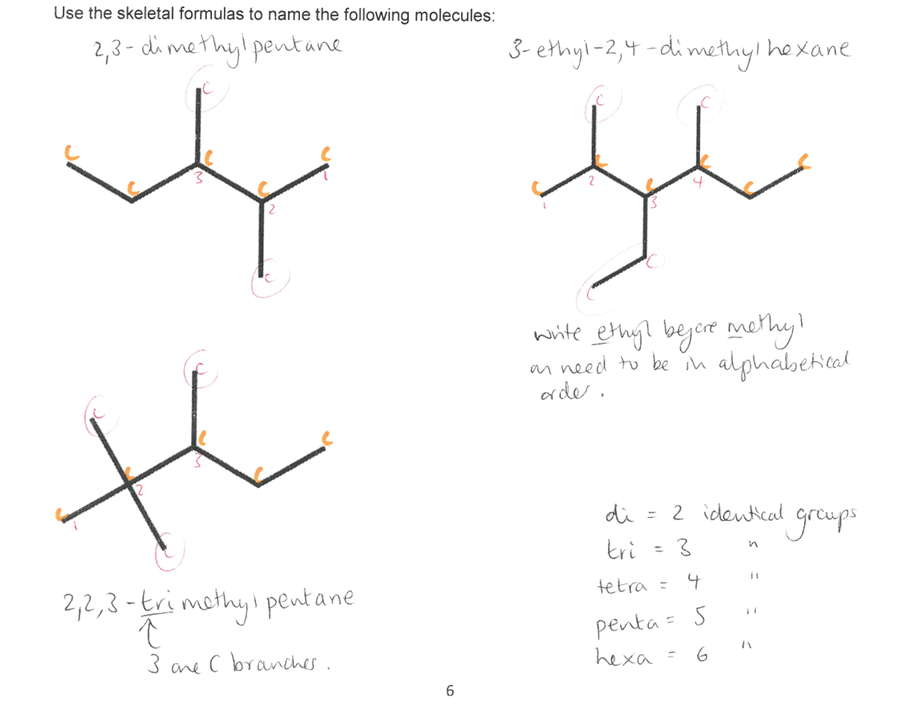
Give examples on naming branched alkanes ONLY using the skeletal formula.
what is the general formula of Alkenes and Cyclic Alkanes
CnH2n
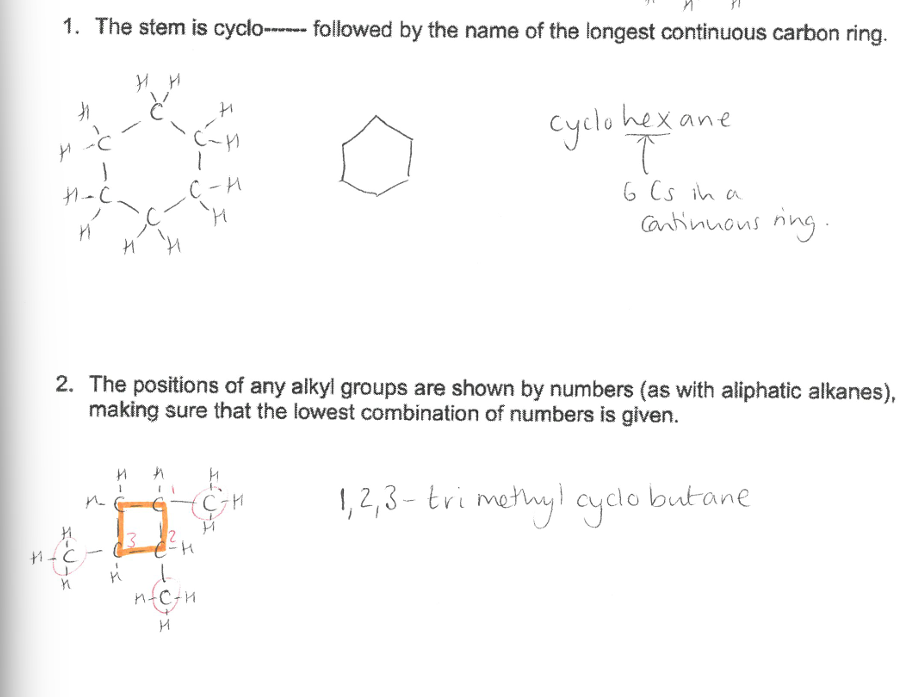
Click the image containing details and examples of Cyclic Alkanes
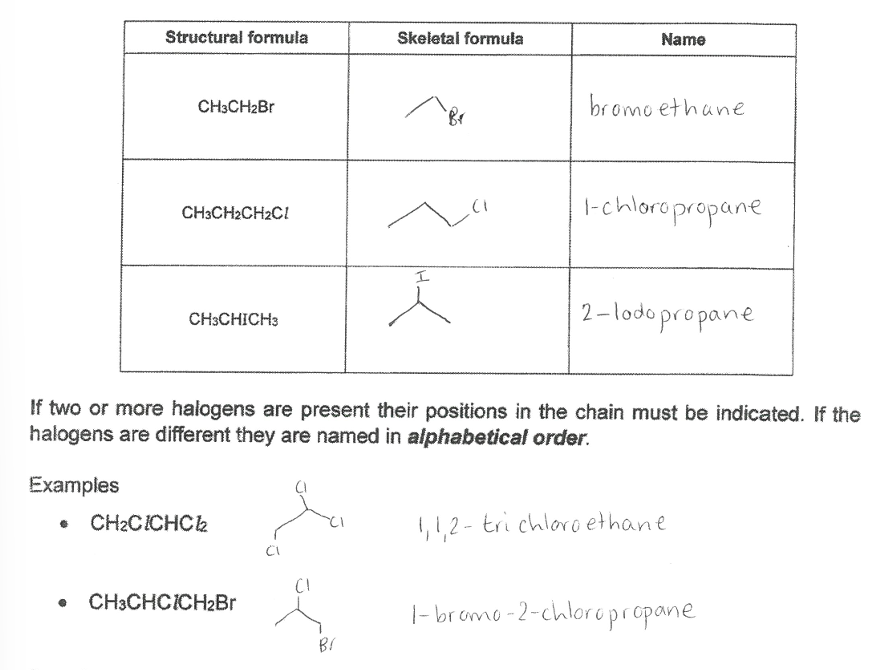
Click the image about halogenoalkanes.
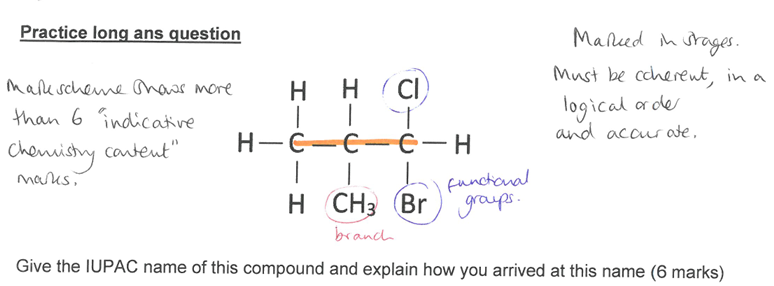
(exam)
longest continuous chain of C atoms is 3 so prop
Only contains single C-C bonds so an alkane
Contains a Cl and Br atom so a halogenalkane
has a IC branch so methyl
Bromo and chloro each need to have a number to show which C atom they are attached to
no number needed for methyl group as it can only go on central C atom
Chloro, bromo, methyl need to be written in alphabetical order so 1-bromo-1-chloro methylpropane
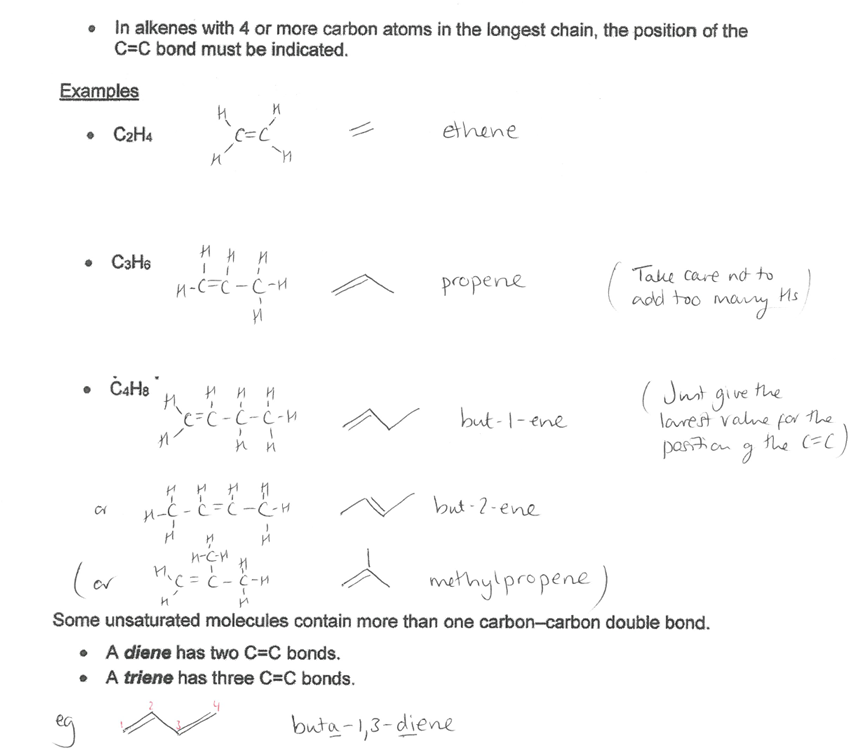
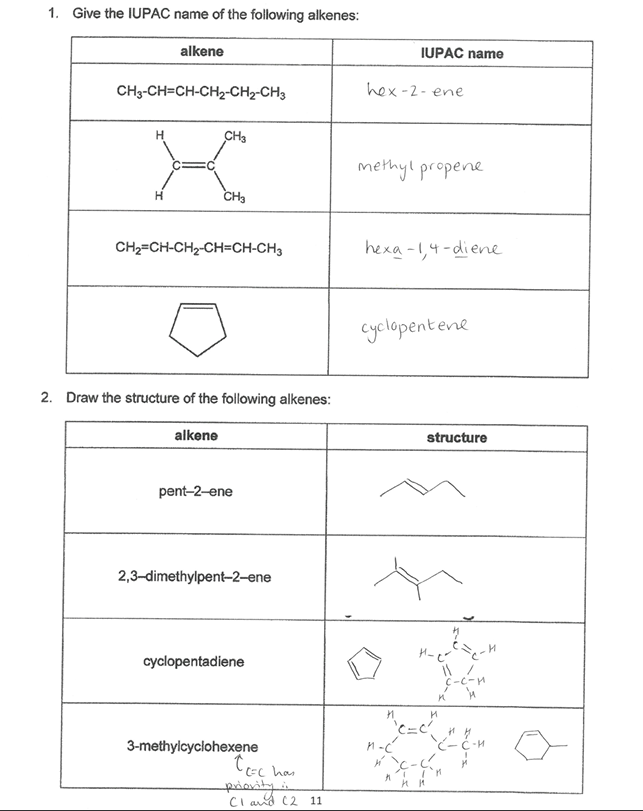
Pick the image of the examples of alkenes
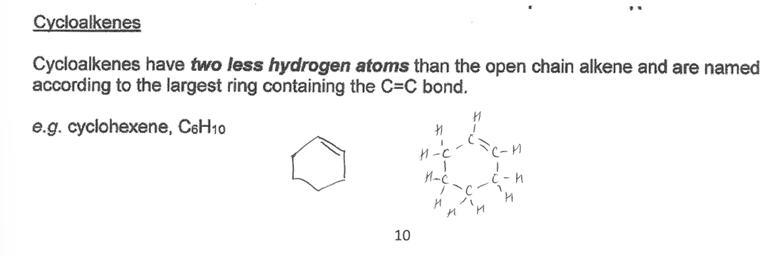
Pick the image about cycloalkenes.

Pick the image with the examples of alcohols

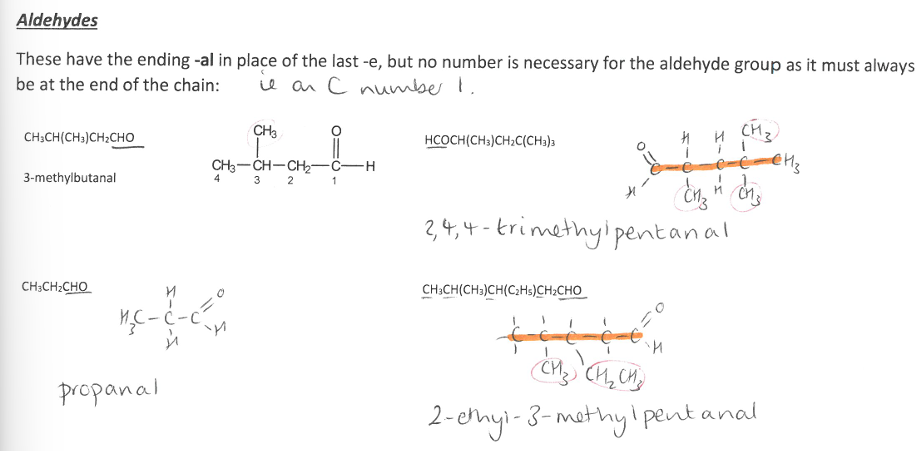
Pick the image about Aldehydes.
Pick the image about Ketones.

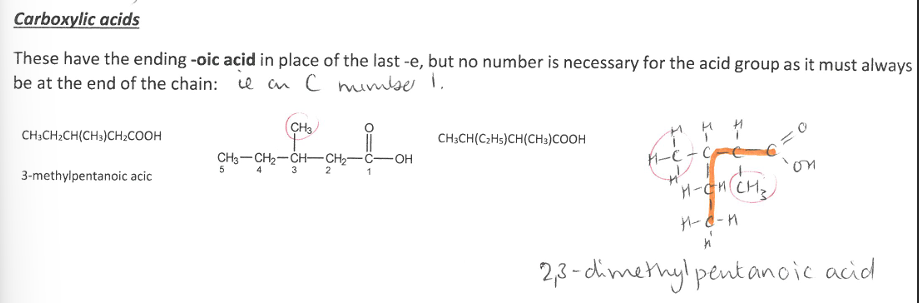
Pick the image about Carboxylic Acids.
Pick the image about examples of combinations of homologous series.

Pick the image about Nitriles.
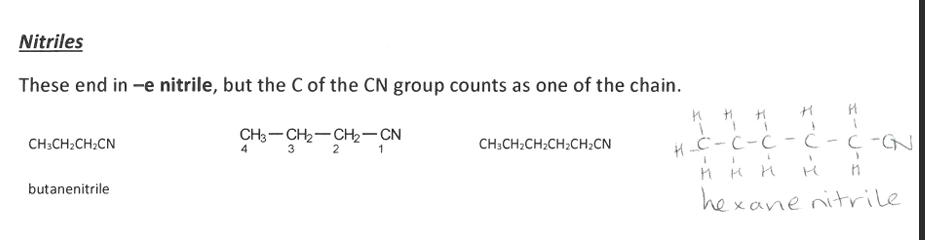
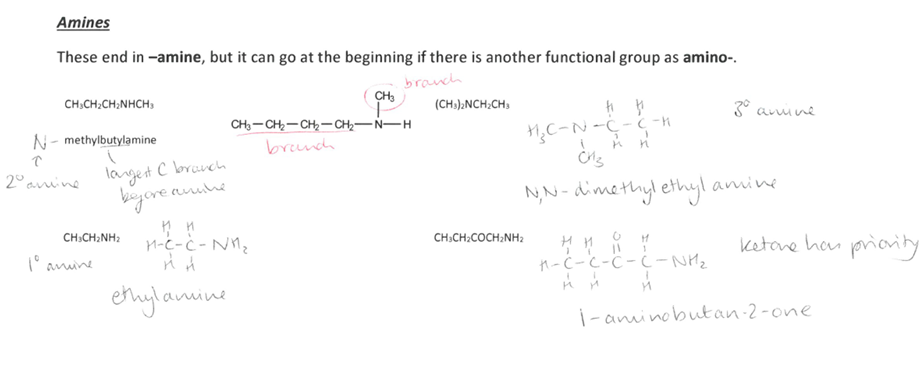
Pick the image about Amines.
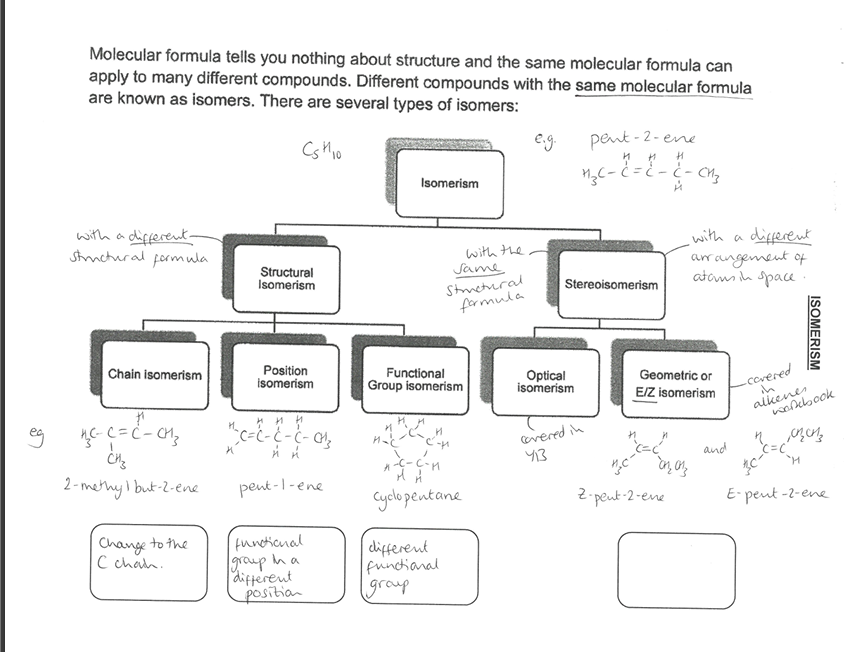
Pick the image of the Isomerism table.
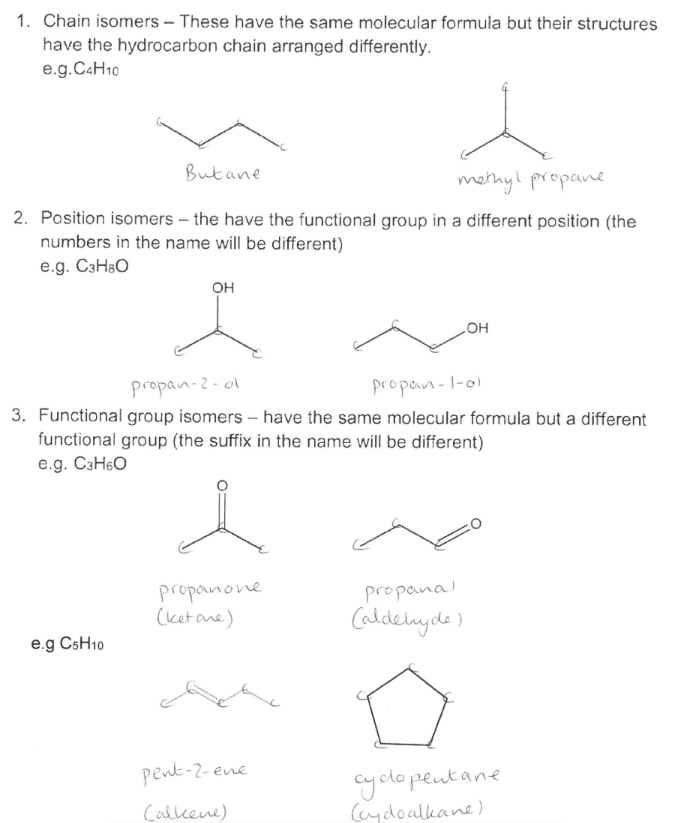
What are the 3 types of structural isomers
What is reaction mechanisms? (part 1)
(1)

What is reaction mechanisms? (part 2)
