OCR A Level Chemistry- Acids and Redox
0.0(0)
Studied by 6 peopleCard Sorting
1/36
Earn XP
Description and Tags
Last updated 7:09 PM on 11/3/22
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
1
New cards
What is an acid?
substances that release H+ ions when dissolved in water. Proton donors
2
New cards
What is a strong acid?
an acid that fully dissociates when dissolved in water
3
New cards
Give 3 examples of strong acids
HCl, H2SO4, HNO3
4
New cards
Define weak acid
acids that partially dissociate when dissolved in water
5
New cards
Give an example of a weak acid
CH3COOH
6
New cards
What is the difference between a base and an alkali?
an alkali is a base that dissolves in water, releasing OH- ions
7
New cards
Give 3 examples of common alkalis
NaOH, KOH, NH3
8
New cards
How can you identify a salt in a reaction?
an ionic compound formed when the H+ ion of an acid is replaced by a metal ion or ammonium ion
9
New cards
What are the products of a reaction between an acid and a metal carbonate?
salt, water and carbon dioxide
10
New cards
What are the common bases?
metal oxides, metal hydroxides, metal carbonates and alkalis
11
New cards
What are the products of a reaction between an acid and a metal oxide?
salt and water
12
New cards
What is a titre?
the volume of acid needed to neutralise the alkali
(the vol of solution added from the burette)
(the vol of solution added from the burette)
13
New cards
why do you invert the volumetric flask containing the standard solution?
to mix it thoroughly
if not, conc is not consistent throughout the mixture
if not, conc is not consistent throughout the mixture
14
New cards
What are the products of a reaction between an acid and an alkali?
salt and water
15
New cards
Give the ionic equation of a neutralisation reaction
H+ + OH- = H2O
16
New cards
What is formed from the reaction of an acid with ammonia?
ammonium salts
17
New cards
What are titrations used for?
Finding out exact conc. or vol. of acid needed to neutralise a quantity of alkali
finding conc./vol of a solution
identifying unknown chemicals
finding purity of a substance
finding conc./vol of a solution
identifying unknown chemicals
finding purity of a substance
18
New cards
What precise apparatus is used in a titration?
burette and pipette
19
New cards
methyl orange in acid v alkali
red in acid
yellow in alkali
yellow in alkali
20
New cards
phenolphthalein in acid v alkali
colourless in acid
pink in alkali
(pale pink when neutralised from acid to alkali)
pink in alkali
(pale pink when neutralised from acid to alkali)
21
New cards
What is a standard solution?
a solution of known concentration
22
New cards
Describe the preparation of a standard solution
1) Work out mass of substance needed to be weighed by multiplying the concentration by the volume to get the mole. Then, multiply the mole by the molar mass to get the mass.
2) Dissolve substance in a beaker with distilled water. Then, pour solution into a volumetric flask, including the rinsings from the beaker. Add distilled water until bottom of meniscus is on the graduation line
2) Dissolve substance in a beaker with distilled water. Then, pour solution into a volumetric flask, including the rinsings from the beaker. Add distilled water until bottom of meniscus is on the graduation line
23
New cards
Describe how to carry out an acid-base titration
1) Add 25cm^3 of the solution of known concentration into a conical flask
2) Place flask on white tile and add indicator (e.g. methyl orange)
3) Add solution of unknown concentration and volume into a burette
4) Turn on tap, swirling the flask until there is a colour change signalling the end-point
5) Calculate the volume of solution added. Repeat titration until there are at least 2 concordant results
2) Place flask on white tile and add indicator (e.g. methyl orange)
3) Add solution of unknown concentration and volume into a burette
4) Turn on tap, swirling the flask until there is a colour change signalling the end-point
5) Calculate the volume of solution added. Repeat titration until there are at least 2 concordant results
24
New cards
What is the process to calculate unknown concentration?
1) Write a balanced equation for the reaction
2) Calculate the amount in moles of the known solution that reacted
3) Use stoichiometry to calculate the amount in moles of the unknown solution that reacted
4) Calculate the unknown concentration by dividing the moles by the volume
2) Calculate the amount in moles of the known solution that reacted
3) Use stoichiometry to calculate the amount in moles of the unknown solution that reacted
4) Calculate the unknown concentration by dividing the moles by the volume
25
New cards
monoprotic, diprotic, polyprotic
1 mole of HCl produces 1 mole of H+ ions
1 mole of H2SO4 produces 2 moles of H+ ions
1 mole of H3SO4 produces 3 moles of H+ ions
1 mole of H2SO4 produces 2 moles of H+ ions
1 mole of H3SO4 produces 3 moles of H+ ions
26
New cards
How can the percentage uncertainty be calculated?
((uncertainty * number of readings) / measured value ) * 100
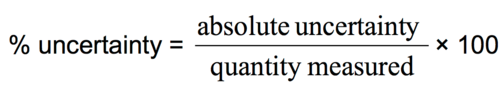
27
New cards
Define oxidation number
a number assigned to an element in chemical combination which represents the number of electrons lost or gained by an atom of that element in the compound
28
New cards
What is the rule for assigning an oxidation number to a pure element?
the oxidation number is zero
29
New cards
What is the rule for assigning an oxidation number to monatomic ions?
the oxidation number is equal to ionic charge
30
New cards
What are the rules for assigning oxidation numbers for compounds?
fluorine= -1
hydrogen= +1
oxygen= -2
hydrogen= +1
oxygen= -2
31
New cards
Describe the common exceptions for assigning oxidation numbers to hydrogen and oxygen
hydrogen- in a metal hydride, it has an oxidation number of -1
oxygen- when it is bonded to fluorine, it has an oxidation number of +2; in a peroxide, it has an oxidation number of -1
oxygen- when it is bonded to fluorine, it has an oxidation number of +2; in a peroxide, it has an oxidation number of -1
32
New cards
What does the sum of the oxidation numbers equal?
the overall charge of the compound (e.g. in a polyatomic ion, it equals the overall charge on the ion)
33
New cards
What are Roman numerals used for?
show the oxidation number of elements that can have different oxidation numbers in different compounds or ions
34
New cards
What is a redox reaction?
a chemical reaction involving both oxidation and reduction
35
New cards
What is oxidation?
the loss of electrons and increase in oxidation number
36
New cards
What is reduction?
the gain of electrons and decrease in oxidation number
37
New cards
What do half equations show?
the electron transfer in a redox reaction by splitting the overall equation into two halves