AQA A level Chem 3.2 Alkanes
1/25
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
Pent-1-ene is a member of the alkene homologous series.
Pent-1-ene can be separated from other alkenes.
State the physical property of alkanes that allows them to be separated from a mixture by fractional distillation (1)
Different boiling points (1)
State the meaning of the term structural isomerism (2)
Same molecular formula with a different structural formula (2)
Explain how NO is produced in the engine of a motor vehicle (2)
Nitrogen and oxygen react (1)
under high temperature (1)
Suggest why Calcium oxide reacts with Sulphur dioxide (1)
Calcium oxide is basic/SO2 is acidic (1)
Butane obtained from crude oil may contain trace amounts of an impurity. When this impurity burns it produces a toxic gas that can be removed by reacting it with calcium oxide coated on a mesh
Suggest why calcium oxide is coated on a mesh (1)
So there’s a larger surface area (1)
Cetane has a melting point of 18oC and a boiling point of 264oC . In polar regions vehicles that use diesel fuel may have ignition problems. Suggest one possible cause of this problem with the diesel fuel(1)
Cetane solidifies (1)
State one type of useful solid material that could be formed from alkanes (1)
Polymers/plastic (1)
State two characteristics of a homologous series (2)
Same general formula (1)
Similar chemical properties (1)
Suggest why the branched chain isomer has a lower boiling point than an alkane (2)
There are weaker vdw forces between the branched chain isomers (1)
Branched chain isomers have a lower contact surface area (1)
There is a risk of gas explosions in coal mines. This risk is mainly due to the presence of methane. If the percentage of coal-mine methane (CMM) in the air in the mine is greater than 15%, the explosion risk is much lower. CMM slowly escapes from the mine into the atmosphere.
Suggest one reason why there is a much lower risk of an explosion if the percentage of CMM is greater than 15%. (1)
Not enough oxygen / air (1)
There is a risk of gas explosions in coal mines. This risk is mainly due to the presence of methane. If the percentage of coal-mine methane (CMM) in the air in the mine is greater than 15%, the explosion risk is much lower. CMM slowly escapes from the mine into the atmosphere.
State why it is beneficial to the environment to collect the CMM rather than allowing it to escape into the atmosphere. (1)
CMM/methane is a greenhouse gas/ contributes to global warming (1)
Methane can be obtained from crude oil. Some of this crude oil contains an impurity called methanethiol (CH3SH). This impurity causes environmental problems when burned. Write an equation to show the complete combustion of methanethiol.(1)
CH3SH + 3O2 →CO2 + 2H2O + SO2(1)
State why high temperatures are necessary for cracking reactions to occur.
Give one reason why thermal cracking reactions are carried out in industry. (2)
To break the (C-C and/or C-H) bonds (1)
To make products which are in greater demand / higher value / make alkenes (1)
Write an equation for the incomplete combustion of pentane to form a solid pollutant.
Suggest why this solid pollutant is an environmental problem. (2)
C5H12 + 3O2 → 5C + 6H2O (1)
Causes global dimming / exacerbates asthma / causes breathing problems / makes visibility poor / smog (1)
Octane and Isooctane can be separated in the laboratory.
Name a laboratory technique that could be used to separate isooctane form a mixture of octane and isooctane.
Outline how this technique separates isooctane from octane (3)
(Fractional) distillation (1)
Isooctane has a lower boiling point so boils first (1)
Isooctane condenses and is collected separately (1)
Platinum, palladium and rhodium are metals used inside catalytic converters. A very thin layer of the metals is used on a honeycomb ceramic support. Explain why a thin layer is used in this way (2)
Increase / maximise / produce large surface area so more reaction can be catalysed (1)
To reduce amount of metals needed which lowers cost (1)
The alkanes nonane and 2,4-dimethylheptane are structural isomers with the molecular formula C9H20
They are found in crude oil and can be separated by fractional distillation.
Both can be used in fuels or cracked to form other products
State the general formula of an alkane containing n carbon atoms
Deduce an expression for the relative molecular mass of an alkane in terms of n (2)
General formula CnH2n+2 (1)
Expression 14n+2 (1)
Suggest one environmental disadvantage of using calcium oxide to remove sulphur dioxide from exhaust gases (1)
CaO requires lots of energy to be made (1)
The production and use of CFCs have been banned in many countries because they decrease the amount of ozone in the upper atmosphere. State why ozone in the upper atmosphere is important for life on Earth. (1)
Absorbs/removes ultraviolet/UV radiation that is harmful/causes (skin) cancer/causes (cell) mutations (1)
A sample of butane has a volume of 20 cm3 at room temperature and pressure.
The sample is burned completely in 1350 cm3 of air.
The final mixture is cooled to room temperature and pressure.
C4H10 +6.5O2 ⟶ 4CO2 +5H2O
Calculate the total volume of gas in the final mixture. Assume that air contains 21% by volume of oxygen. (4)
Initial volume O2 = 0.21 × 1350 = 283.5 (cm3) (1)
Volume of O2 remaining = M1 - (6.5 × 20) = 153.5 cm3(1)
Volume of CO2 formed = 20 × 4 = 80 cm3(1)
Total volume of gas left = M2 + M3 + (0.79 × 1350) = 1300 cm3 (1)
Natural gas is used in power stations to produce electricity.
Natural gas contains sulfur impurities. Sulfur dioxide forms when these impurities are burned.
State an environmental problem caused by sulfur dioxide.
Give the formula of a compound that is used to help remove sulfur dioxide from the combustion products.
Environmental problem _______________________________________
Formula of compound ________________________________________(2)
Acid rain (1)
CaO or CaCO3 (1)
Carbon dioxide is a product from the combustion of petrol in cars. Carbon dioxide acts as a greenhouse gas when it absorbs infrared radiation.
Give one reason why carbon dioxide absorbs infrared radiation. (1)
(C=O) bonds vibrate (at IR frequency) / (IR/it makes) bonds vibrate (1)
Nitrogen monoxide (NO) is formed when petrol is burned in cars. State one environmental problem that NO causes.
State what is used to remove NO from the exhaust gases formed in petrol-fuelled cars.
Environmental problem _______________________________________
Removal of NO _____________________________________________(2)
acid rain / respiratory problems (1)
catalytic converters (1)
Which statement is correct about thermal cracking?
A A pressure between 100 and 200 kPa is used.
B Aromatic hydrocarbons are the major products.
C C–C bonds are broken.
D Zeolite catalysts are used. (1)
C (1)
Sulfur dioxide is produced in the combustion of fossil fuels. The total emissions of sulfur dioxide in the UK have fallen dramatically since 1970.
Sulfur dioxide is now removed from the flue gases in power stations by reaction with calcium oxide.
CaO + SO2 → CaSO3
In 1970, the total UK emissions of sulfur dioxide were 6.49 million tonnes
(1 tonne = 1000 kg).
Calculate the mass, in kilograms, of calcium oxide needed to react with this mass of sulfur dioxide. Give your answer in standard form. (2)
(2)
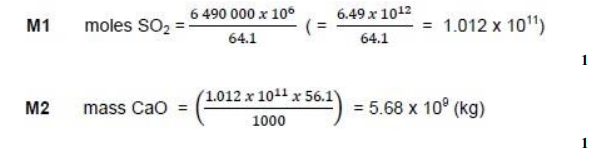
Isooctane (2,2,4-trimethylpentane) is an important component of petrol
used in cars.
When isooctane is burned, the enthalpy change is −47.8 kJ g−1
Isooctane is a liquid at room temperature with a density of 0.692 g cm−3
Calculate the heat energy released, in kJ, when 1.00 dm3 of isooctane
burns in excess oxygen.
Give your answer to the appropriate number of significant figures. (2)
mass of isooctane = 692 (g) (1)
3.31 × 104 or 33100 (kJ)