AQA Chemistry 5: Energy changes
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
What happens to the amount of energy in a chemical reaction?
The amount of energy in the universe at the end of a chemical reaction is the same as before the reaction takes place
What is an exothermic reaction?
-energy is transferred to the surroundings
-temperature of the surroundings increases
-eg combustion, many oxidisation reactions, neutralisation
-eg self-heating cans, hand warmers
What is an endothermic reaction?
-energy is taken in from the surroundings
-temperature of the surroundings decreases
-eg thermal decompositions, reaction of citric acid and sodium hydrogencarbonate
-eg sports injury packs
When do chemical reactions occur?
When reacting particles collide with each other with sufficient energy
What do reaction profiles show?
-relative energies of the reactants and products in a reaction
-how energy changes over the course of the reaction
What does an exothermic reaction profile look like?
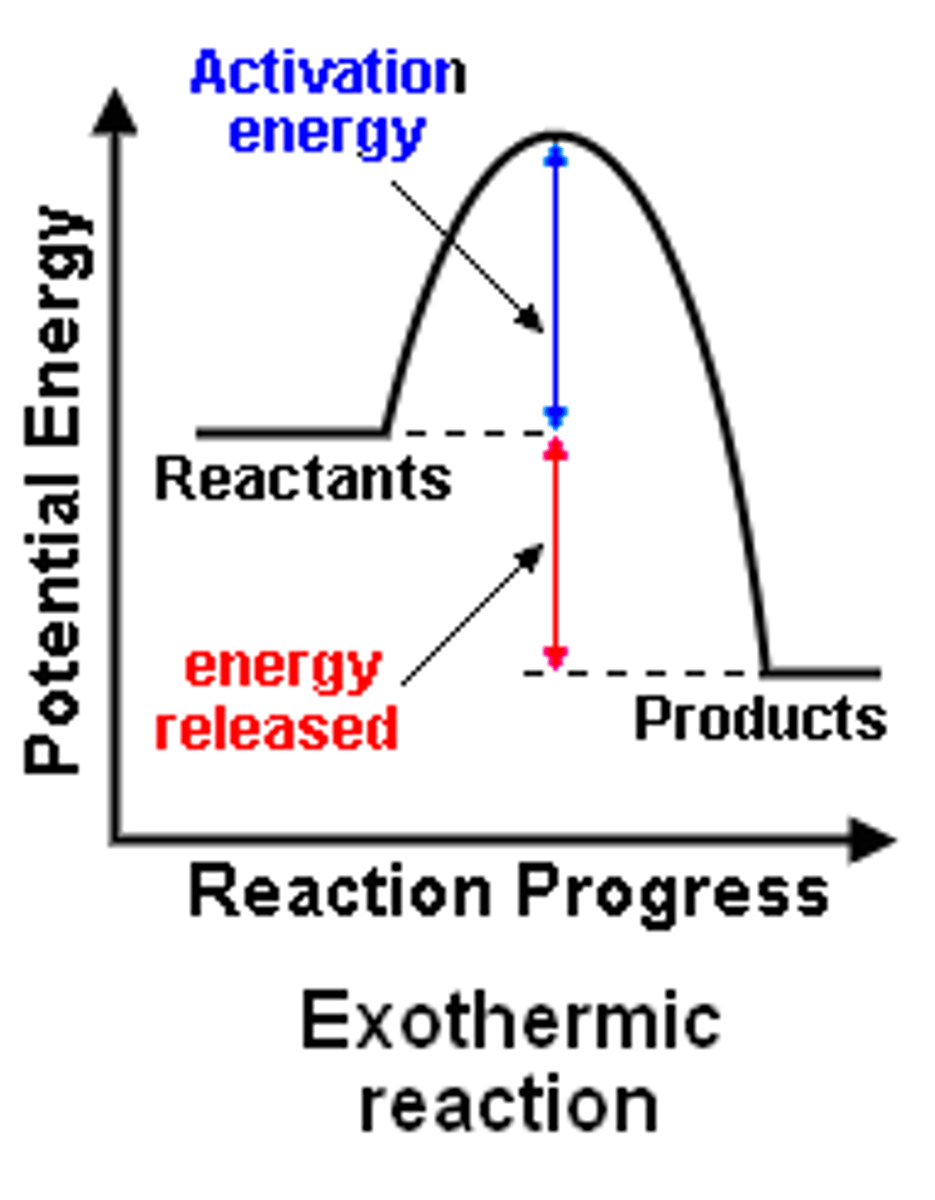
What does an endothermic reaction profile look like?
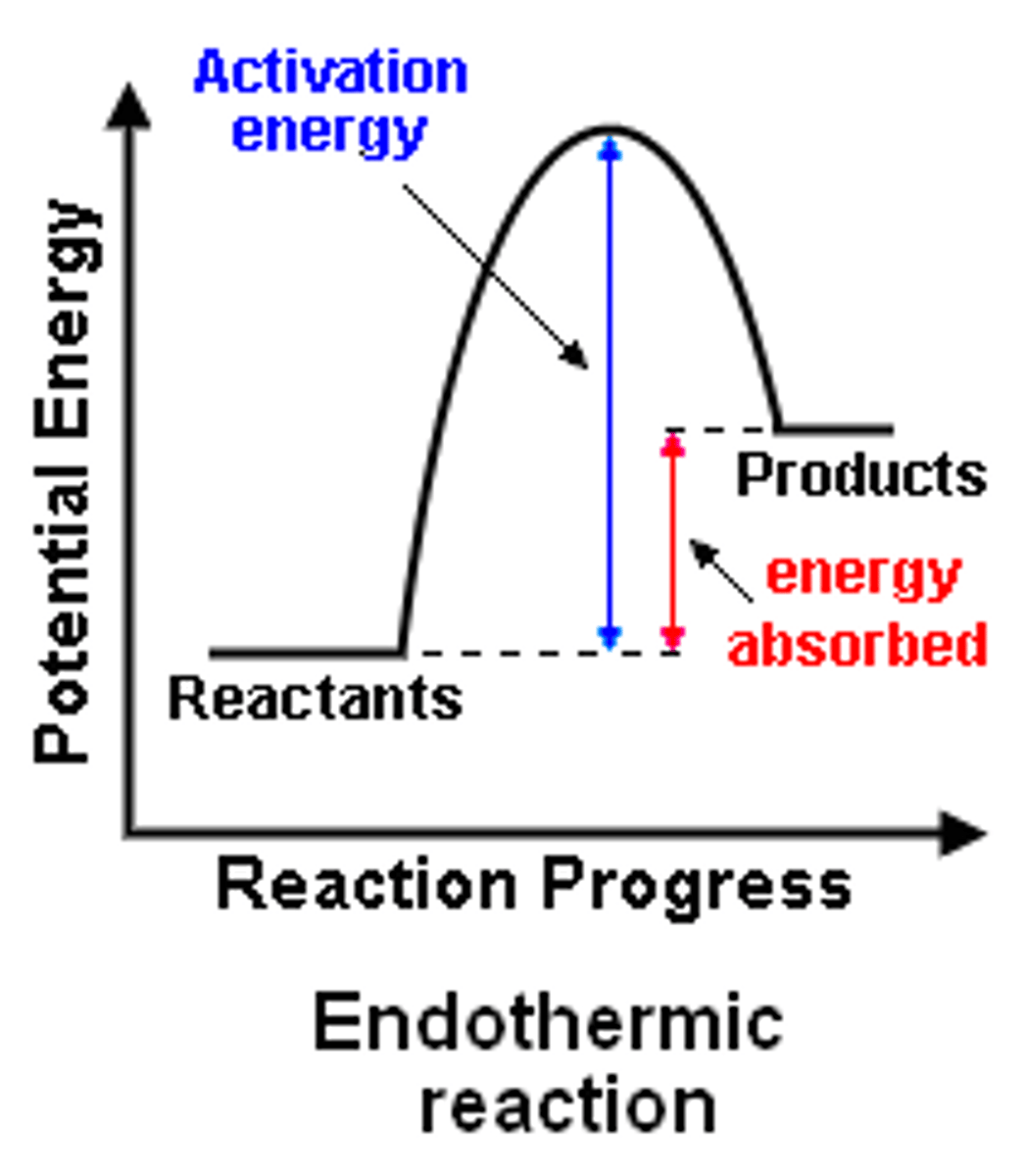
[HT only] What happens to bonds during chemical reactions?
• energy must be supplied to break bonds in the reactants
• energy is released when bonds in the products are formed.
[HT only] How do you calculate bond energy?
1. Add together the bond energies for all the bonds in the reactants - this is the 'energy in'.
2. Add together the bond energies for all the bonds in the products - this is the 'energy out'.
3. Calculate the energy change = energy in - energy out.
[HT only] What is the overall energy change of a reaction?
The difference between the sum of the energy needed to break bonds in the reactants and the sum of the energy released when bonds in the products are formed
[HT only] In an exothermic reaction, is bond energy positive or negative?
-positive
-the energy released from forming new bonds is greater than the energy needed to break existing bonds
[HT only] In an endothermic reaction, is bond energy positive or negative?
-negative
-the energy needed to break existing bonds is greater than the energy released from forming new bonds
[Triple only] What is a cell?
Cells contain chemicals which react to produce electricity
[Triple only] What does the voltage produced by a cell depend on?
-type of electrode
-type of electrolyte
[Triple only] How can a simple cell be made?
A simple cell can be made by connecting two different metals in contact with an electrolyte.
[Triple only] What is a battery?
Batteries consist of two or more cells connected together in series to provide a greater voltage
[Triple only] What is the difference between non-rechargeable and rechargeable cells and batteries?
non-rechargeable cells and batteries:
-the chemical reactions stop when one of the reactants has been used up.
-Alkaline batteries are non-rechargeable.
-rechargeable cells and batteries:
-can be recharged because the chemical reactions are reversed when an external electrical current is supplied.
[Triple only] What is a fuel cell?
Fuel cells are supplied by an external source of fuel (eg hydrogen) and oxygen or air.
[Triple only] How does fuel in a fuel cell produce a potential difference?
The fuel is oxidised electrochemically within the fuel cell to produce a potential difference.
[Triple only] What does the reaction in a hydrogen fuel cell involve?
the oxidation of hydrogen to produce water.
[Triple only] What are the pros and cons of using hydrogen fuel cells?
Hydrogen fuel cells offer a potential alternative to rechargeable cells and batteries.
+ easy to maintain, no moving parts
+ small size
+ water is only chemical product
- expensive to manufacture
- need a constant supply of hydrogen gas
- hydrogen gas is flammable
[Triple only] What are the pros and cons of using alkaline cells (batteries)?
+ cheap to manufacture
- may end up in landfill once used
- expensive to recycle
[Triple only] What are the pros and cons of using rechargeable cells?
+ can be recharged many times before being recycled, reduces use of resources
- more expensive than alkaline cells to manufacture