CHE 132 Exam 2
1/51
Earn XP
Description and Tags
At this point, just throw me in a pit of wild dogs. It would be better than this shi-
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
Dynamic Equilibrium
The condition where the forward and reverse reaction rates are equal.
Write the rate expressions for H₂ + I₂ ⇌ 2HI
Forward: rate = k[H₂][I₂]
Reverse: rate = k₋₁[HI]²
At equilibrium: kH₂][I₂] = k₋₁[HI]²
What is the general equilibrium expression for aA + bB ⇌ cC + dD?
Kc = products/reactants
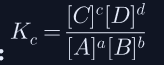
How does reversing a reaction affect K?
The new K is the reciprocal: Krev = 1/Kfwd.
If a reaction is multiplied by n, how does K change?
Knew = (Kold)n
How do you combine equilibrium constants when adding reactions?
Multiply the individual K values.
“A + B + D ⇌ E + F … K = K₁ × K₂”
What is the relationship between Kp and Kc?
Kp = Kc (RT)Δn where Δn = (moles gas products – moles gas reactants). “Kp = Kc(RT)^(Δn)”
Which phases are excluded from equilibrium expressions?
Pure solids and pure liquids.
When is the x ≪ initial approximation valid?
When x (ratio) is less than 5% of the initial concentration
What does a large K mean?
Products are favored; equilibrium lies to the right.
What does a small K mean?
Reactants are favored; equilibrium lies to the left.
What does Q (reaction quotient) < K (reaction constant) mean?
Reaction shifts right (toward products).
What does Q > K mean?
Reaction shifts left (toward reactants).
Factors that make equilibrium shift to the left (reactants)
increasing product concentration, removing reactant, increasing temperature for exothermic reactions, decreasing temperature for endothermic reactions, or decreasing pressure/increasing volume for systems with more moles of gaseous products
Factors that make a reaction shift to the right (products)
increasing reactant concentration, removing products, increasing pressure (if products have fewer gas moles), or changing temperature based on reaction enthalpy (increasing heat for endothermic, decreasing for exothermic)
When a system reaches equilibrium,
the rates of the forward and reverse reactions are equal
A catalyst ___ the rate of the reaction, and it _____ the concentration of the product at equilibrium conditions
increases, does not change
Arrhenius definition of an acid?
Produces H+ (aq) in water
Arrhenius definition of a base?
Produces OH⁻ in water
Brønsted–Lowry acid definition?
Proton (H+) donor
Brønsted–Lowry base definition?
Proton acceptor
What are conjugate acid–base pairs?
Species differing by one proton (HA/A⁻ or H₃O⁺/H₂O).
Strong acids (complete ionization in water)
HCl, HBr, HI, HNO₃, H₂SO₄, HClO₄, HClO₃.
What pH corresponds to [H₃O⁺] = 1.0×10⁻⁷ M?
7.00
How many sig figs appear after the decimal in pH?
Same number as sig figs in [H₃O⁺]. (7.00 M)
How do you find pH of a strong acid?
pH = –log[acid], because it fully dissociates.
What defines a weak acid?
Partial dissociation; most HA remains undissociated.
General weak acid dissociation reaction?
HA + H₂O ⇌ H₃O⁺ + A⁻.
Expression for Ka of HA?
Ka = [H3O+][A-] / [HA]
What does a large Ka mean?
Stronger acid; favors products (more H⁺).
What does a small Ka mean?
Weaker acid; favors reactants.
What is pKa?
pKa = -logKa
Relationship between pKa and acid strength?
Lower pKa = stronger acid, Higher pKa, = weaker acid
What is the general reaction for a weak base in water?
B + H2O ⇄ BH+ + OH-
What does Kb measure?
The extent to which a base produces OH⁻ in water. [BH+][OH-] / [B]
How does Kb relate to base strength?
Larger Kb → stronger base; lower pKb → stronger base.
Lower Kb → weaker base; Higer pKb,→ weaker base
What structural feature is common in weak bases?
A nitrogen atom with a lone pair.
How are Ka and Kb related for conjugate pairs?
Ka ⨉ Kb = Kw
What defines a strong base?
Complete dissociation to produce OH⁻.
Which metal hydroxides are strong bases?
Group 1 hydroxides; Group 2 hydroxides (Ba, Sr, Ca) at <0.1 M (LiOH, NaOH, KOH… Ba(OH)₂, Sr(OH)₂, Ca(OH)₂)
What defines a weak acid?
Partial dissociation; most HA remains undissociated.
Larger Ka means
more acidic, equilibrium lies to the right
Percent dissociation
([H3O+]eq / [HA]initial) ⨉ 100%
Why does dilution increase percent dissociation?
reducing the concentration of a weak electrolyte decreases the rate of the reverse reaction (recombination of ions) faster than the forward reaction (dissociation)
What is a polyprotic acid?
An acid that can donate more than one proton.
How are pH and pOH related?
pH+pOH=14.00
What is the relationship between acid strength and conjugate base strength?
Strong acid → negligible conjugate base; weak acid → weak/medium base
What determines whether a species acts as an acid or base in water?
Whether it donates or accepts a proton relative to water.
What determines which equilibrium controls pH in a mixture?
The reaction with the larger equilibrium constant.
What happens to pH when a strong acid is added to a weak base solution?
The weak base is protonated, reducing [OH^-] and lowering pH.
The stronger the acid…
the weaker the conjugate base
The weaker the acid…
the stronger the conjugate base