2. Experimental Techniques (for WA1)
1/118
Earn XP
Description and Tags
Paper Chromatography + Collection and drying of gases
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
119 Terms
two or more elements chemically combined in a fixed composition
compounds
two or more substances physically mixed together in any proportion
mixtures
components of a ________ can be seperated by physical means
mixture
can only be broken down into its elements or its simpler compounds by chemical processes
compounds
the chemical properties of a _______ are the same as those of its components
mixtures
physical and chemical properties of a ______ are different from its constituent elements
compounds
methods to determine purity: b________ ________ ___________, __________ ________ _________, ________ __________
boiling point determination, melting point determination, paper chromatography
importance of measuring purity: ensures that only _________ substances are used esp in the food industry
permitted
importance of measuring purity: presence of _________ can have ______ effects in a ______ / _________
impurities, harmful, drug, medicine
importance of measuring purity: presence of _________ can _____ with _______ pure substances are involved
impurities interfere reactions
purity: for example the addition of salt to water ________ its boiling point but the addition of salt to water ________ its freezing point
raises lowers
purity: the ________ the amount of ________, the larger the change in melting or boiling points
greater impurities
a pure substance has a ________ meling and/or boiling point under fixed conditions
fixed
chromatography:____________ ________ may be used to reveal some colourless substances e.g. conjugated systems
ultraviolet light
locating agent is a _______ that can ____ with ________ substances in order to make them _______
chemical react colourless visible
retention factor is the ratio between the ________ _________ by the _________ and the ________ ________ by the ________
distance travelled substance distance travelled solvent
methods of paper chromatography: ________ method is faster due to the effect of _________
descending gravity
chromatography: ________ ______ should be near the ____ of the paper to ensure that the dyes are ________ separated
solvent line top fully
paper chromatography: ______ of solution and dyes on the starting line should be _______ and ________ to ensure that the dyes are _________ __________
spots small concentrated distinctly separated
chromatography: the more soluble the substance, the ______ and _______ it travels
further faster
chromatography is used to _______ and _______ _______ amounts of substances that are ________ in _______.
seperate identify small dissolved solvents
chromatography: why does the container need to be closed with a lid? (to __________ ________ _______ and _______ ___ ________ ________ is ________)
to minimise solvent evaporation and drying up before process is completed
chromatography: will the results of the experiment be the same if a different solvent is used? (the result will be _______ as the ______ have _________ ________ in _________ _________)
the result will be different as the solutes have different solubilities in different solvents
chromatography: why should the start line be above the solvent level? (So that the _______ will ____ __________ in the ________ _______ the _________ in the _____ can be ___________)
so that the sample will not dissolve in the solvent before the components in the sample can be seperated
chromatography: why should the spots and dyes on the starting line be small? (to _______ that there will _____ be any __________ of the _________)
to ensure that there will not be any overlapping of the components
paper chromatography: why should the solvent front be near the end of the paper at the end of the experiment? (to ________ _________ _________ of the _________ in the _______)
to ensure maximum seperation of the components in the sample
chromatography: how can chromatograpy be applied to colourless substances? (use a ________ _______)
use a locating agent
chromatography: what are the advantages of using chromatography (over other seperation techniques)? (only a _____ amount of _______ is _______. _________ to __ __ and ______, in the case of ______ _____________)
only a small amount of sample is required. inexpensive to set up and conduct, in the case of paper chromatography
paper chromatography: why should the base line be drawn using a pencil instead of a pen? (the _______ will ____ _________ the _______ _____, unlike ____ which contains _______ _____. the _________ ______ from the ____ will ________, thus ________ with _____ from the test sample)
the solvent will not seperate the pencil mark, unlike ink which contain soluble dyes. the seperated dyes from the ink will smudge, thus interfering with dyes from the test sample
how does the two solutes separate from each other in chromatography? (A _____ that is ____ _______ in the solvent will travel up the filter paper a ________ distance than a solute that is _____ _________ in the solvent)
a solute that is more soluble in the solvent will travel up the filter paper a greater distance than a solute that is less soluble in the solvent
how does chromatography works? (the ________ solutes in the _____ have _________ solubilities in the _____ solvent)
the different solutes in the mixture have different solubilities in the same solvent
why should spots be above the solvent in chromatography? (to _______ the spots from being _________ from the paper)
to prevent the spots from being removed from the paper
why is an ink pen not used as the ink for chromatography?
ink is a mixture which can dissolve in certain solvents and spread up the chromatogram
what is sprayed on the chromatogram to make the colourless spots visible? (2 words)
locating agent
separate substances with different solubility
chromatography
The method to collect a gas from a preparation depends on two factors: (_ of the _ in _, _ of the _ as compared to _)
solubility of the gas in water, density of the gas as compared to air
method for gases that are insoluble or have low solubility in water
displacement of water
what gases can be collected using displacement of water
carbon dioxide hydrogen oxygen
use displacement of water for gases that are _ or have _ _ in _
insoluble low solubility water
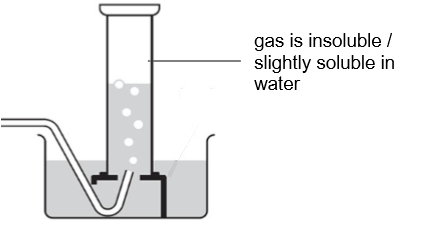
what method is this
displacement of water
method for gases that are more dense than air
downward delivery
what gases can be collected using downward delivery
chlorine hydrogen chloride gas
use downward delivery for gases that are _ _ than _
more dense air
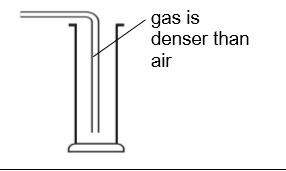
what method is this
downward delivery
method for gases that are less dense than air
upward delivery
what gases can be collected using upward delivery
ammonia gas hydrogen gas
use upward delivery for gases that are _ _ than _
less dense air
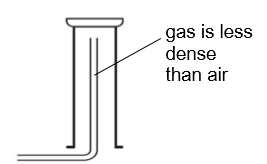
what method is this
upward delivery
gas can also be collected and measured using a _ _
gas syringe
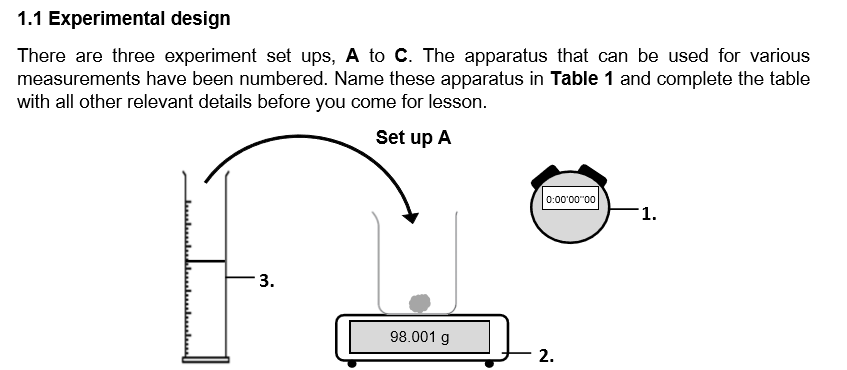
What do you think is the purpose of this set up? Which apparatus is essential for this purpose? (_ the _ of a _ by _ the decrease in _ _ _. _ is an essential apparatus for investigating experiments involving _.)
Measure the rate of a reaction by determining the decrease in mass over time. Stopwatch is an essential apparatus for investigating experiments involving rate.
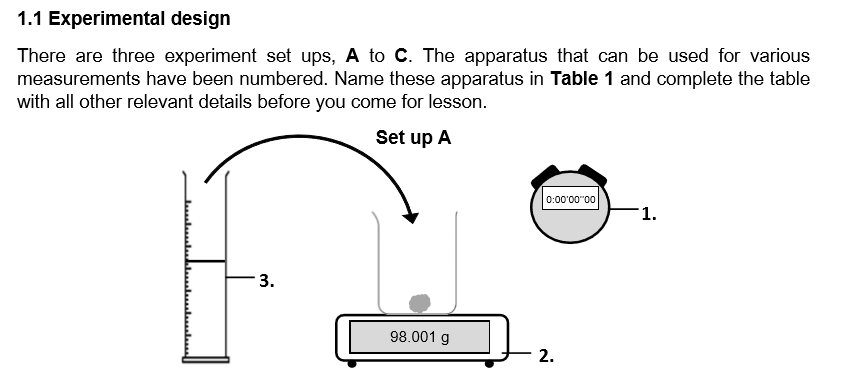
Can i use a beaker to measure the same volume of solution? (_ a beaker is _ _ for any _ of _ as it is _ an _ instrument.)
No a beaker is not used for any measurement of solutions as it is not an accurate instrument.
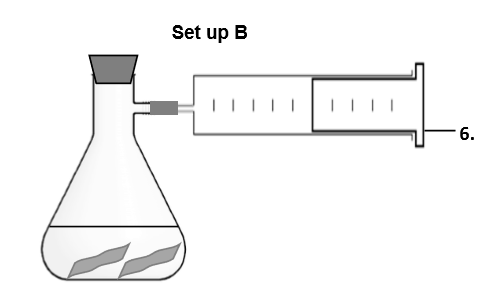
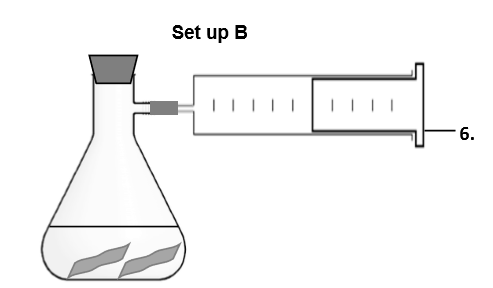
What do you think is the purpose of this set up? Is there another way to achieve this purpose? (_ and _ _ of gas. A _ _ can be used for measuring the _ of a gas _.)
Collect and measure volume of gas. A gas syringe can be used for measuring the volume of a gas accurately.
What do you think is the purpose of this set up? Is there another way to achieve this purpose? (_ and _ _ of gas. _ _ _ method is another way to _ the _ of gas produced if the gas is _ in _.)
Collect and measure volume of gas. Displacement of water method is another way to measure the volume of gas produced if the gas is insoluble in water.
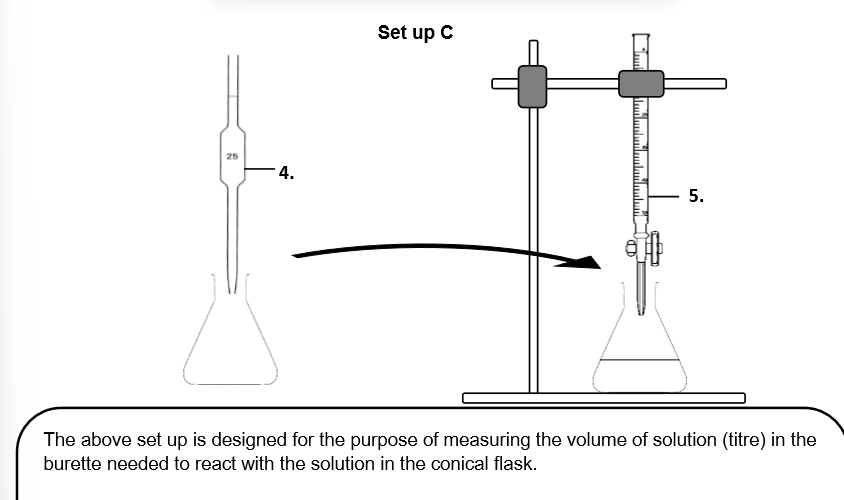
Can I use a beaker to dispense the liquid instead of apparatus '5.'? (No a beaker _ _ _ as it cannot _ the _ of a liquid _ .)
No a beaker cannot dispense liquid as it cannot measure the volume of a liquid accurately.
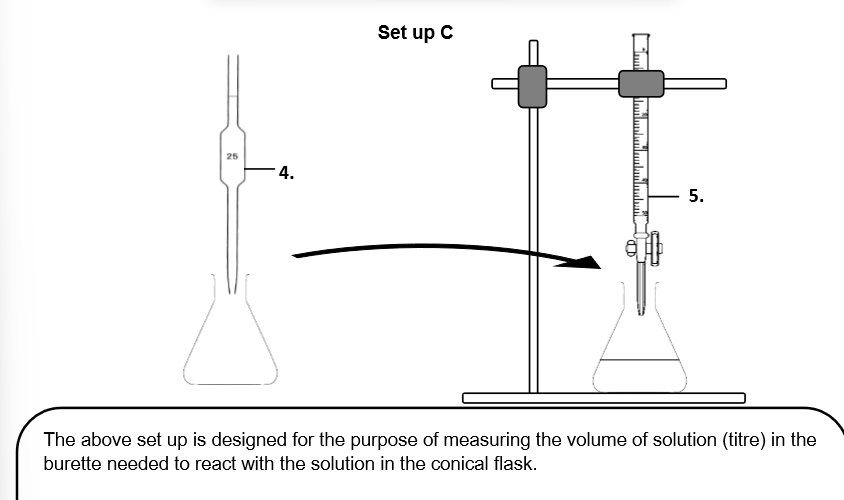
Can I use a measuring cylinder to measure out the 25.0cm^3 instead? (_ the measuring cyclinder is _ _ than a _.)
No the measuring cylinder is less accurate than a pipette.
We can dry a gas by passing it through a _ _ in appropriate apparatus.
drying agent
_ is used to dry acidic gases such as chlorine and hydrogen chloride.
concentrated sulfuric acid
concentrated sulfuric acid is used to dry _ gases such as _ and _ _.
acidic chlorine hydrogen chloride
concentrated sulfuric acid cannot be used to dry _ gases as it _ with them.
alkaline reacts
_ is used to dry alkaline gases such as ammonia gas
quicklime (calcium oxide)
quicklime (calcium oxide) is used to dry _ gases such as _ gas
alkaline ammonia
quicklime (calcium oxide) cannot be used to dry gases as it with them.
acidic reacts
_ can be used to dry most gases such as hydrogen
fused calcium chloride
fused calcium chloride can be used to dry _ gases such as _.
most hydrogen
fused calcium chloride cannot be used to dry _ gas
ammonia
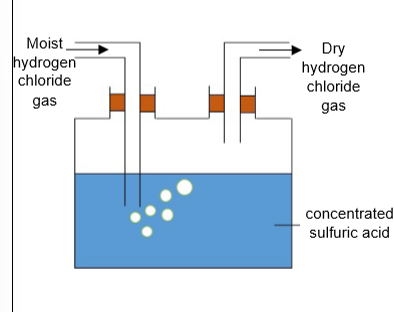
what drying agent
concentrated sulfuric acid
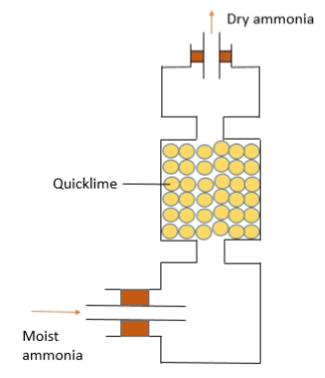
what drying agent
quicklime
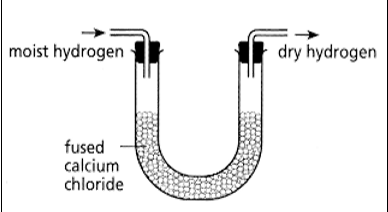
what drying agent
fused calcium chloride
what apparatus is used to measure time?
stopwatch
what apparatus is used to measure temperature?
thermometer
what apparatus is used to measure mass?
electronic balance
what apparatus (most accurate) is used to measure volume of solution or liquid?
burette
what apparatus is used to measure volume of solution or liquid more accurate than measuring cylinder?
pipette
what apparatus (least accurate) is used to measure volume of solution or liquid?
measuring cylinder
what apparatus is used to measure volume of gas?
gas syringe
precision of thermometer including units
0.5'C
precision of electronic balance including units (record what we see) ________ or ______
0.01g or 0.001g
precision of stopwatch including units (record what is asked by qn)
0.01s
precision of measuring cylinder including units (half of smallest division)
0.5cm3
precision of pipette including units
25.0cm3
precision of burette including units
0.05cm3
precision of gas syringe including units
0.5cm3
solubility in water of ammonia
high
solubility in water of hydrogen chloride
high
solubility in water of sulfur dioxide
high
solubility in water of chlorine
high
solubility in water of carbon dioxide
low
solubility in water of carbon monoxide
low
solubility in water of oxygen
low
solubility in water of hydrogen
insoluble
solubility in water of helium
insoluble
solubility in water of nitrogen
insoluble
acidity/basicity of ammonia
basic
acidity/basicity of hydrogen chloride
acidic
acidity/basicity of sulfur dioxide
acidic
acidity/basicity of chlorine
acidic
acidity/basicity of carbon dioxide
acidic
acidity/basicity of carbon monoxide
neutral
acidity/basicity of oxygen
neutral
acidity/basicity of hydrogen
neutral