PHAN LAB Midterm Liquid Dosage Form
1/51
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
Quality Control
Its main objective in the Pharmaceutical Industry is to test the drugs in their various stages of production, verifying that they can proceed to the next stage and release the manufacturing process following the regulations and specifications required for consumption.
Quality Control
In the physical-chemical laboratory, controls of all types are carried out, from the simplest to the most complex. We can mention simple analyses of the product’s appearance, DELIVERABLE VOLUME, VISCOSITY, Ph, and so on.
Official test
Unofficial test
Two types of quality control tests
Weight variation test
Drug content (assay)
Disintegration time test
Dissolution test
Moisture content
Official Quality Control Tests
Thickness
Hardness
Friability
Organoleptic characters (Color, odor, taste)
Unofficial Quality Control Tests
Organoleptic evaluation
assesses the sensory characteristics of products, such as their taste, smell, appearance, and texture.
Deliverable volume
General chapters: <698>
<698>
Deliverable volume General Chapter
<698> Deliverable Volume
designed to provide assurance that oral liquids will, when transferred from the original container, deliver the volume of dosage form that is declared on the label.
<698> Deliverable Volume
The tests apply whether the products are supplied as liquid preparations or liquid preparations that are constituted from solids upon the addition of a designated volume of a specific diluent.
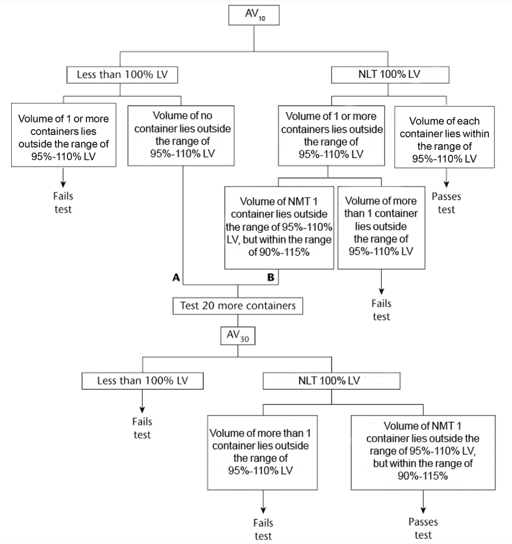
The average volume of liquid obtained from the 10 containers is NLT 100%, and the volume of each of the 10 containers lies within the range of 95%–110% of the volume declared in the labeling. If (A), the average volume is less than 100% of that declared in the labeling, but the volume of no container is outside the range of 95%–110%, or if (B), the average volume is NLT 100% and the volume of NMT 1 container is outside the range of 95%–110%, but within the range of 90%–115%, perform the test on 20 additional containers. The average volume of liquid obtained from the 30 containers is NLT 100% of the volume declared in the labeling; the volume obtained from NMT 1 of the 30 containers is outside the range of 95%–110%, but within the range of 90%–115% of the volume declared on the labeling.
Acceptance criteria: for single-dose container
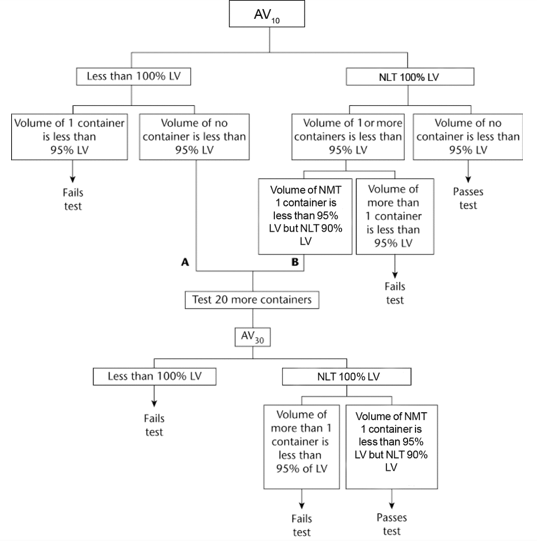
The average volume of liquid obtained from the 10 containers is NLT 100%, and the volume of no container is less than 95% of the volume declared in the labeling. If (A), the average volume is less than 100% of that declared in the labeling, but the volume of no container is less than 95% of the labeled amount, or if (B), the average volume is NLT 100% and the volume of NMT 1 container is less than 95%, but is NLT 90% of the labeled volume, perform the test on 20 additional containers. The average volume of liquid obtained from the 30 containers is NLT 100% of the volume declared in the labeling; the volume of liquid obtained from NMT 1 of the 30 containers is less than 95%, but NLT 90% of that declared in the labeling.
Acceptance criteria: for multiple-dose container
viscosity
General chapters: <912>
<912>
Viscosity General chapters
Viscosity
The principle of the method is to measure the force (torque) acting on a rotor when it rotates at a constant angular velocity or rotational speed in a liquid.
Rotational rheometers/viscometers
are used for measuring the viscosity of fluids, both Newtonian and non-Newtonian.
Shear-thinning or pseudoplastic
These fluids become less viscous as the shear rate increases. Examples include ketchup, yogurt, and certain types of paint.
ketchup, yogurt, and certain types of paint.
Shear-thinning or pseudoplastic examples
Shear-thickening or dilatant
These fluids become more viscous as the shear rate increases.
cornstarch and water mixtures (oobleck) and some types of industrial slurries.
Shear-thickening or dilatant Examples
Thixotropic
These fluids become less viscous over time when subjected to constant stress or shear. However, their viscosity increases again once the stress is removed. Examples include certain types of clay suspensions and printing inks.
Rheopectic
These fluids become more viscous over time when subjected to constant stress or shear. Their viscosity decreases when the stress is removed.
Cylinder spindle
Disk-shaped spindle
Two types of spindle viscometer
pH
General chapters: <791>
<791>
pH General chapter
pH
is a measure of the acidity or alkalinity of a solution.
pH
is a measure of the concentration of hydrogen ions (H⁺) in a solution.
"power of hydrogen"
pH stands for
Acids
were substances that had a sour taste, were corrosive, and reacted with substances called bases.
bases
Substances that had a bitter taste, made skin slippery on contact, and reacted with acids
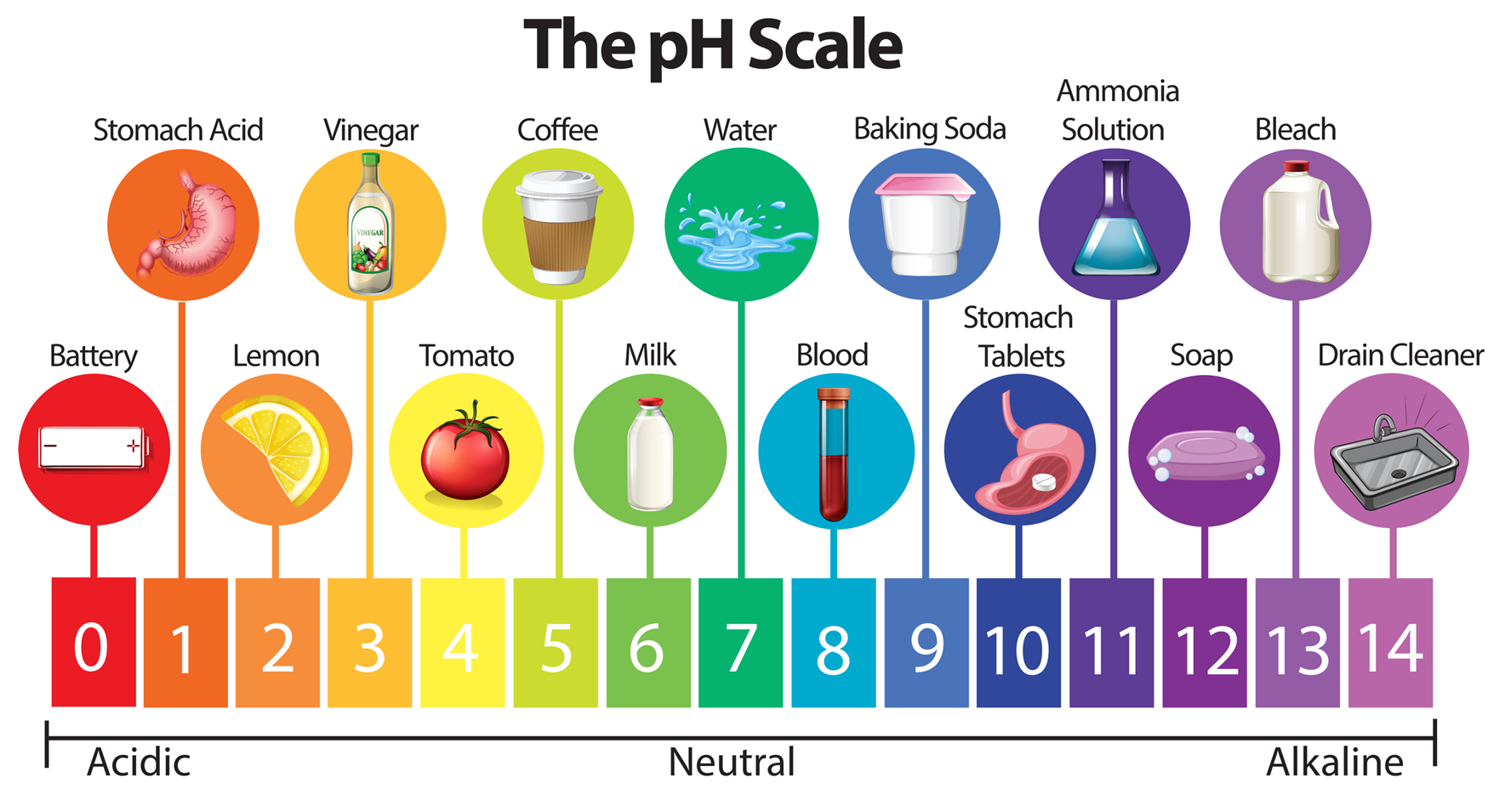
pH scale
pH meter
is a scientific instrument used to measure the pH of a solution accurately.
a pH probe,
an electronic meter, and
a reference electrode.
pH meter consists of
pH probe
is a specialized sensor designed to measure the pH of a solution. It typically consists of a glass electrode with a pH-sensitive membrane at the tip.
pH probe
This membrane is sensitive to the concentration of hydrogen ions (H⁺) in the solution.
pH probe
When this is immersed in a solution, the hydrogen ions in the solution interact with the pH-sensitive membrane, causing a voltage to develop across the glass electrode. This voltage is proportional to the pH of the solution.
Electronic Meter
is the device that reads the voltage generated by the pH probe and converts it into a pH value.
Electronic Meter
It typically includes a display screen that shows the pH reading.
electronic meter
may also have features such as temperature compensation, calibration options, and data logging capabilities.
Litmus paper
is a simple and widely used tool for testing the acidity or alkalinity of a solution.
Litmus paper
It consists of small strips of paper impregnated with a mixture of dyes derived from lichens.
Acids
it ionizes to form hydrogen(+) ions in aqueous solution.
Bases
It ionizes to form hydroxide ions OH(-) in aqueous solution.
Arrhenius theory
Acid → produces H⁺ (hydrogen ions) in solution
Base → produces OH⁻ (hydroxide ions) in solution
Bronsted-lowry theory
According to this theory, an acid-base reaction involves the transfer of a proton from the acid (the proton donor) to the base (the proton acceptor).
Bronsted-lowry theory
This transfer results in the formation of a conjugate acid-base pair.
Ionization constant (Ka)
It indicates the relative strength of the acid or base
stronger (more ionized)
An acid with a Ka of 1 x 10‑1 is ________ than one with a Ka of 1 x 10‑3
weaker (less ionized)
a base with a Ka of 1 x 10-7 is ________ than one with a Ka of 1 x 10-9.
pKw
the negative logarithm of water ion constant,
pKa
negative logarithm of acid dissociation constant
pKb
negative logarithm of base dissociation constant