Chem - topic 2
1/48
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
49 Terms
What are ions?
Ion - a charged particle
Formed when atoms gain or lose electrons to become more stable
Full outer shell = more stable
Losing or gaining electrons requires energy
What is ionic bonding?
Non-metal + metal
Atoms have opposite charges
So are attracted to eachother by electrostatic forces
Ionic bond - really strong
What happens to the electrons in ionic bonding?
Metal atoms lose electrons to become positively charged ions
Non-metal atoms gain electrons to become negatively charged ions
These oppositely charged ions are strongly attracted to one another by electrostatic forces
What is covalent bonding?
Non-metal + non-metal
Share a pair of electrons between the atoms
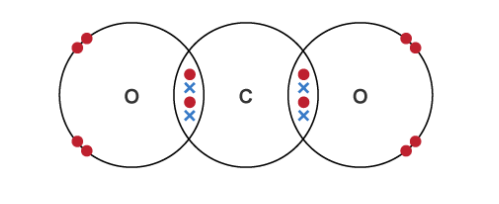
What happens to the electrons in covalent bonding?
Non-metal atoms share pair(s) of electrons to make covalent bonds - only in outer shell
Held together by electrostatic forces of attraction between the positively charged nuclei of the bonded atoms and the shared pair of electrons - covalent bonds are strong
What is metallic bonding?
The electrostatic attraction between positive metal ions and delocalised electrons
What is the structure ionic compounds?
Giant ionic lattice
Held together by strong electrostatic forces of attraction in all directions between oppositely charged ions
What are the properties of ionic compounds?
High melting and boiling points - a large amount of energy is required to break the strong ionic bonds
When solid, ions are held in place (not free to move) so cannot conduct electricity
When molten or aqueous - can conduct electricity as ions are free to move and so charge can flow
Examples of simple molecular substances
Hydrogen (H2) - single covalent bond
Chlorine (Cl2) - single covalent bond
Oxygen (O2) - double covalent bond
Nitrogen (N2) - triple bond
Methane (CH4) - four covalent bonds
Hydrogen Chloride (HCl) - single covalent bond
What are the properties of simple molecular substances? (small covalent molecules)
Held together by strong covalent bonds but weak intermolecular forces
Usually liquids or gases at room temperature - intermolecular forces are weak, so energy is not needed to break the bonds
Very low melting and boiling points - little energy required to break weak intermolecular forces (increase with size of the molecules)
Don't conduct electricity - no free electrons or ions and so no overall electrical charge
What is the effect of increased molecule size?
As molecule size increases:
• Strength of intermolecular forces increases
• More energy required to break them
• Melting/boiling point increases
What is a polymer?
Long chain of monomers (repeating units)
Joined together by strong covalent bonds
Strong intermolecular forces
What are the properties of polymers?
Solid at room temperature - have strong intermolecular forces of attraction
High melting and boiling points - require a lot of energy to break the strong intermolecular forces
What are giant covalent structures?
• All atoms are bonded to each other by strong covalent bonds
• In a giant lattice structure
What are the properties of giant covalent structures?
• High melting and boiling point - hard to overcome strong covalent bonds
• Non conductors of electricity, except graphite
What is the structure of diamond?
Giant covalent structure
Each carbon atom forms covalent bonds to four other carbon atoms
Arranged in giant lattice
What are the properties of diamond?
Solid at room temperature because they have high melting and boiling points - unlike simple molecular substances (small covalent molecules), it is the strong covalent bonds that must be broken when they are melted / boiled
Extremely hard substance - contains millions of carbon atoms joined by covalent bonds
Cannot conduct electricity - all of the outer electrons are in covalent bonds so diamond has no free electrons to carry electrical charge
What is the structure of silicon dioxide?
• Giant covalent molecule - huge number of strong covalent bonds
• Each silicon atom is covalently bonded to 4 oxygen atoms
• Each oxygen atom is covalently bonded with 2 silicon atoms
What are the properties of silicon dioxide?
Very hard
High melting/boiling point
Does not conduct electricity
What is the structure of graphite?
Each carbon atoms forms covalent bonds to three other carbon atoms
Arranged in layers of hexagonal rings (no covalent bonds between layers, only weak intermolecular forces)
Each atom has one delocalised electron
What are the properties of graphite?
Soft and slippery - carbon atoms form heaxagonal rings. The hexagonal rings of carbon atoms are arranged into layers. There are no covalent bonds between these layers so the layers can slide over eachother
High melting and boiling point - requires lots of energy to break the strong covalent bonds
Conducts electricity and heat - carbon atoms have four electrons in their outer shell. In graphite, the carbon atom forms covalent bonds to three other carbon atoms. Each carbon atom has one electron in its outer shell that is not in a covalent bond. This means graphite has delocalised electrons which are free to move and conduct electricity and heat
What are the uses of graphite?
Lubricant - reducing friction between moving parts
Pencils
Electrolysis
What is the structure of graphene?
One layer of graphite (one atom thick)
Contains delocalised electrons
What are the properties of graphene?
Strong
Light (can be added to composite materials to improve strength without adding much weight)
Can conduct electricity - delocalised electrons which are free to move and carry electrical charge
High melting and boiling point
What are the uses of graphene?
Electronics and composites
What is the structure of fullerene?
Fullerenes are molecules of carbon atoms with hollow shapes
Carbon atoms arranged in hexagonal rings (can be with five or seven carbon atoms)
What are the uses of fullernes?
Delivering drugs to the body
Industrial catalysts (due to large surface area)
Lubricants in machines
What was the first fullerene discovered and what is its structure?
Buckministerfullerene
Hexagonal rings of carbon atoms
60 carbon atoms (C60)
Spherical shape
What is the structure of nanotubes?
Hollow carbon cylinders formed from fullerenes
What are the properties of nanotubes?
High ratio between length and diameter of nanotube
Can conduct both electricity and thermal energy - have delocalised electrons
High tensile strength (don't break when stretched)
What are the uses of nanotubes? (nanotechnology)
Electronics
Strengthen materials without adding much weight
Advantage of 2D ball and stick model
Shows which atoms are bonded to each other
Disadvantage of 2D ball and stick model
Does not show true shape of the molecule
Advantage of 3D ball and stick model
Shows the shape of a molecule
Disadvantage of 3D ball and stick model
Does not show how it is bonded via electrons
Advantage of dot and cross diagram
Shows electrons from each atom
Disadvantage of dot and cross diagram
It is a 2D representation - don’t show the real shape of molecules
What is the structure of metals?
Giant structure of positive ions arranged in regular layers
Sea of delocalised electrons
Strong forces of electrostatic attraction between positive metal ions and the sea of delocalised negative electrons
What are the properties of metals?
High melting/boiling points - a lot of energy is required to overcome the strong electrostatic forces
Good conductors of electricity and heat - delocalised electrons free to carry charge throughout the metal
Soft -layers of atoms able to slide over each other
Malleable
Ductile
Shiny
What is an alloy?
A mixture of metals
In an alloy the different sizes of atoms distort the layers
This makes it more difficult for the layers to slide over eachother
Alloys are harder than pure metal
What is the particle theory?
Energy needed for a change of state depends on strength of forces between particles
Stronger forces between particles mean higher melting/boiling points
What are the limitations of the particle model?
Model shows no forces between particles
Assumes that all particles are solid spheres
Particles in solids
Strong forces of attractions between particles, holding them close together in fixed positions to form regular lattice arrangement
Particles vibrate about their positions - the hotter the solid becomes, the more they vibrate (slightly expand when heated)
Particles don't move from their positions - keep a defined shape and volume
Cannot flow and cannot be compressed
Particles in liquids
Weak force of attraction between particles
Randomly arranged and free to move, but tend to stick closely together
Constantly moving in random directions - the hotter the liquid gets, the faster they move (expand when heated)
Have definite volume but not definite shape - will flow
Cannot be compressed
Particles in gases
Very weak forces of attraction between particles
Free to move and far apart
Move constantly with random motion - the hotter the gas gets, the faster they move (expand or pressure increases when heated)
Don't keep definite shape or volume - always fill space
Can be compressed
Particles during melting (solid to liquid)
Solid is heated → particles gain more KE and vibrate more
At the melting point, the particles have enough energy to overcome the intermolecular forces holding them in fixed positions
Temperature stays constant while the solid melts → solid becomes a liquid
Particles during boiling/evaporating (liquid to gas)
Liquid is heated → particles gain KE and move faster
At the boiling point, particles have enough energy to overcome the intermolecular forces between them
Temperature stays constant while the liquid boils → liquid becomes a gas
Particles during condensation (gas to liquid)
Gas cools → particles have less KE and move more slowly
Particles no longer have enough energy to overcome intermolecular forces, so they start to stick together
Temperature stays constant while the gas forms a liquid → gas becomes a liquid
Particles during freezing (liquid to solid)
Liquid cools → particles have less KE so move around less
Particles no longer have enough energy to overcome intermolecular forces, so they start to be held in place
Temperature stays constant while the solid forms → liquid becomes a solid