CHEM LAB 1212
1/127
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
128 Terms
What must you do before coming to the lab?
Select all that apply.
Write the Chemical Alert Table in the lab notebook.
Review the chemicals and their hazards.
Read the experiment.
Take the Pre-Lab Quiz.
Prepare lab notebook for the experiment.
Corrosive
Irreversibly destroys living tissue or metal upon contact
Toxic
may cause harm through ingestion, inhalation or skin absorption
Irritant
Chemical causes dryness, itching or sensitivity
Used (and rinsed) Beral pipet and Filter paper
Disposed in trash can
Reaction mixture and excess chemica
Disposed in appropriate waste container
Cracked or chipped beaker
Disposed in Broken glass box
Which of the following items of clothing are appropriate to wear in CHEM 1212 Lab to protect your skin?
T-shirt
Scrub pants
Lab Apron
Tank top
Midriff top
Knee length shorts
Jeans
Leggings
Which of the following are appropriate to wear in CHEM 1212 Lab to protect your feet?
Sneakers or tennis shoes, Boots, Loafers
Where is the eye wash station located?
Mounted on the wall next to the large sink.
What should you do if a chemical gets into your eye?
Flush with eye-wash solution for 15 minutes.
What should you do if a chemical is spilled on your hand?
Rinse with running water for 15 minutes.
What should you do if a person's hair or clothing catch fire?
Wrap with a fire blanket and then Stop, Drop & Roll.
What should you do if a chemical is splashed on your leg?
Remove affected clothing and rinse with running water for 15 minutes.
What should you do if you spill a chemical on your lab bench?
Clean it up using the spill kit in your lab.
Can you eat or drink in the lab?
NO
When is it okay to remove your chemical splash proof goggles?
When everyone in the lab has put away all chemicals and glassware.
Which of the following are Good Lab Practices (GLPs)?
Use a brush to clean any spilled chemical off the balance., Record all volumes directly into your lab notebook., Rinse your buret with the titrant before using., Always replace the cap back on the solid jar.

What does the GHS icon (below) represent?
Flammable
What is the density of a solid that occupies a volume of 5.89 mL and weighs 5.14 g?
0.873 g/mL
Adding mercury(ℓ) to water results in a solution consisting of two layers.
What describes the relationship between mercury and water?
immiscible
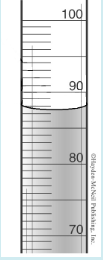
In the picture, what is the correct volume reading?
87.7 mL
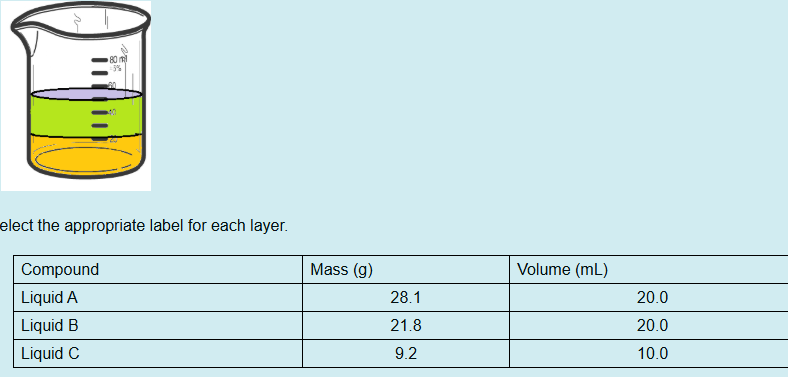
Which chemical species is in the bottom layer?
Liquid A
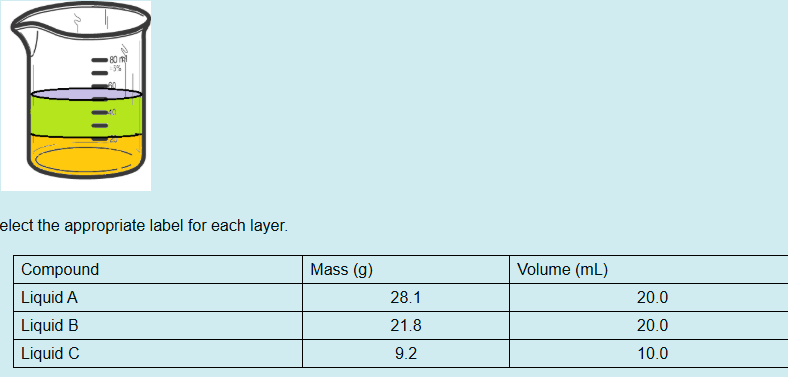
Which chemical species is in the top layer?
Mixture of B & C
Adding water(ℓ) to diesel fuel (ℓ) results in two layers.
What describes the relationship between diesel fuel and water?
immiscible
Which property is an example of a physical property?
A solid’s melting point.
How would the calculation of density for CH2Cl2 be affected if the volume was recorded as 4.29 mL instead of 4.92 mL and the mass was 6.23 g? (REASON)
Since dividing by a smaller number in the denominator (volume) makes the calculated density larger.
How would the calculation of density for CH2Cl2 be affected if the volume was recorded as 4.29 mL instead of 4.92 mL and the mass was 6.23 g? (AFFECT)
It will make the calculated density higher.
Which chemical(s) used in this experiment is/are an oxidizing agent? (Exp. 2)
H2O2
Identify the reaction type for the generic reaction given.
A + B → A+n + B–m
Oxidation-Reduction
Identify the reaction type for the generic reaction given.
ABC → AB + C
Decomposition
What should you do if some of the H2O2-H2SO4 solution comes into contact with your skin?
Wash the skin for 15 minutes with water and notify the TA.
What should you do if NaOH gets into your eye because you took your googles off?
Wash your eye for 15 minutes with the solution from the eyewash station and notify the TA.

coefficient in front of KOH(s)
2

coefficient in front of H2SO4(aq)
1

Classification of reaction
Double displacement

coefficient in front of H2O (liq)
2

coefficient in front of K2SO4(aq)
1
In this reaction, you observed several different Cu species which had different physical attributes.
Select the appropriate chemical formula(s) that corresponds to a sky blue solution.
CuSO4
What is the mole to mole ratio between Cu(C2H3O2)2·H2O and NaC7H4SO3N·H2O?
1 mol Cu(C2H3O2)2·H2O : 2 mol NaC7H4SO3N·H2O

What is the theoretical yield?
9.52

What is the actual yield?
8.25

What is the percent yield?
86.6
Cooper, Zach and Nate did this experiment dissolving 3.50 g of Cu(C2H3O2)2·H2O and adding 7.90 g NaC7H4SO3N·H2O (assume present in excess).
What is the theoretical yield of the product, Cu(C7H4SO3N)2(H2O)4·2 H2O(s)?
Molar Mass of Copper(II) acetate monohydrate = 199.65 g/mol
Molar Mass of Sodium saccharinate monohydrate = 223.18 g/mol
Molar Mass of product = 535.59 g/mol
9.39 g
Zach and Nate calculated the theoretical yield as 6.57 g of Cu(C7H4SO3N)2(H2O)4·2 H2O.
They recovered 4.53 g of Cu(C7H4SO3N)2(H2O)4·2 H2O.
What is the percent yield?
68.9%
In a similar experiment, Cooper, Zach & Nate started with 4.18 g of Cu(C2H3O2)2·H2O and 9.78 g of NaC7H4SO3N·H2O and obtained 10.84 g of Cu(C7H4SO3N)2(H2O)4·2H2O
Molar Mass of Copper(II) acetate monohydrate = 199.65 g/mol
Molar Mass of Sodium saccharinate monohydrate = 223.18 g/mol
Molar Mass of product = 535.59 g/mol
The theoretical yield of the product is
11.21g
In a similar experiment, Cooper, Zach & Nate started with 4.18 g of Cu(C2H3O2)2·H2O and 9.78 g of NaC7H4SO3N·H2O and obtained 10.84 g of Cu(C7H4SO3N)2(H2O)4·2H2O.
Molar Mass of Copper(II) acetate monohydrate = 199.65 g/mol
Molar Mass of Sodium saccharinate monohydrate = 223.18 g/mol
Molar Mass of product = 535.59 g/mol
The percent yield of the product is
96.7%
For the procedural error, indicate if the error will affect the actual yield of copper(II) saccharinate product and if it does, will it raise or lower the actual yield:
Scraping the filter paper so hard that small bits of filter paper are with the crystals on the watch glass
Raises the actual yield
For the procedural error, indicate if the error will affect the actual yield of copper(II) saccharinate product and if it does, will it raise or lower the actual yield:
Not pulling air through the crystals (to dry crystals)
Raises the actual yield
Which statement best explains what happens to the percent yield if the crystals (product) were washed with hot water?
The actual yield will decrease since part of the product will dissolve and not be recovered as a solid.
Since actual yield is in the numerator, the percent yield will also decrease.
Which definition corresponds to excess reagent?
The reactant that is present as extra material when the reaction is complete.
Which definition corresponds to aliquot?
A small portion of a chemical species.
Which definition corresponds to coordination compound?
A chemical species that contains a metal ion that is coordinated to ligands.

The theoretical yield is …
3.93 g

The actual or experimental yield is …
3.43 g

The chemical, C2H8N2, is the …
limiting reagent

The chemical, NiCl2· 6 H2O, is the
chemical present in excess
What color is expected for the product, [Ni(C2H8N2)3]Cl2 (s), if C2H8N2 is the limiting reagent?
Pink-violet
For the procedural error, indicate if the error will affect the actual yield of [Ni(C2H8N2)3]Cl2 product and if it does, will it raise or lower the actual yield:
Rinsing the crystals with water
Lowers the actual yield
For the procedural error, indicate if the error will affect the actual yield of [Ni(C2H8N2)3]Cl2 product and if it does, will it raise or lower the actual yield:
Added an extra 5 mL of acetone in step 7 of Part II
No effect
Ethyl, Ann & Ami reacted 3.45 g of NiCl2·6 H2O with 10.00 mL of 4.00 M C2H8N2.
They recovered 2.47 g of product, (Ni(C2H8N2)3)Cl2. The theoretical yield of the product is
4.13 g
Ethyl, Ann & Ami reacted 3.45 g of NiCl2·6 H2O with 10.00 mL of 4.00 M C2H8N2.
They recovered 2.47 g of product, (Ni(C2H8N2)3)Cl2. The percent yield of the product is
59.8%
Which definition corresponds to acid-base indicator?
A chemical species that changes colors at different pH values.
In today’s experiment, which chemical species is the acid-base indicator?
(C6H5OH)2C2O2C6H4 (phenolphthalein)
Which chemical(s) will be in the buret? (Exp. 5)
NaOH
Cedrick & Astrid titrated a 20.00 mL aliquot of grapefruit juice with a 0.165 M NaOH solution to the end point.
The initial buret reading was 1.72 mL and the final buret reading was 15.51 mL.
H3C6H5O7(aq) + 3 NaOH(aq) → Na3C6H5O7(aq) + 3 H2O(l).
What is the volume of NaOH titrated?
13.79 mL of NaOH
Final Buret volume - Initial Buret Volume = volume titrated
Cedrick & Astrid titrated a 20.00 mL aliquot of grapefruit juice with a 0.165 M NaOH solution to the end point.
The initial buret reading was 1.72 mL and the final buret reading was 15.51 mL.
H3C6H5O7(aq) + 3 NaOH(aq) → Na3C6H5O7(aq) + 3 H2O(l).
What is the mass of citric acid in the juice sample?
0.146 g of H3C6H5O7
Cedrick & Astrid titrated a 25.00 mL aliquot of grapefruit juice with a 0.117 M NaOH solution to the end point. The initial buret reading was 1.82 mL and the final buret reading was 21.33 mL.
They calculated the mass of citric acid in the juice as 0.1462 g. What is the amount mg of citric acid present per mL of juice?
5.85 mg/mL
Sid Trick titrated a 17.00 mL aliquot of juice. It required 24.61 mL of 0.1320 M NaOH to reach the equivalence point. Analyze his results and answer the questions below. The reaction and the molar mass are given below.
H3C6H5O7(aq) + 3 NaOH(aq) → Na3C6H5O7(aq) + 3 H2O(ℓ)
Molar Mass of H3C6H5O7 = 192.12 g/mL
How many grams of citric acid were present in the aliquot?
0.2080 g
Sid Trick titrated a 17.00 mL aliquot of juice. It required 24.61 mL of 0.1320 M NaOH to reach the equivalence point. Analyze his results and answer the questions below. The reaction and the molar mass are given below.
H3C6H5O7(aq) + 3 NaOH(aq) → Na3C6H5O7(aq) + 3 H2O(ℓ)
Molar Mass of H3C6H5O7 = 192.12 g/mL
How many mg of citric acid were present per mL of juice?
12.24 mg/mL
For the error, indicate if the error will affect the mg of citric acid present per mL of juice, and if it does, will it raise or lower the value of the mg citiric acid/ mL of juice?
Not subtracting the initial buret volume (1.20 mL) from final buret volume (18.43 mL)
Raises the value of mg citric acid/mL juice
For the error, indicate if the error will affect the mg of citric acid present per mL of juice, and if it does, will it raise or lower the value of the mg citiric acid/ mL of juice?
Under-titrating the end point (stopping before the endpoint is reached)
Lowers the value of mg citric acid/mL juice
A common error in titration is over-titrating.
Which of the following statement(s) is/are TRUE for the citric acid titration?
Over-titrating means too much titrant was added to the reaction.
Since the volume of titrant is too large, dividing by this larger number in the calculation of citric acid makes the mass of citric acid too low.
Over-titrating means too much aliquot was added to the reaction.
Only I is TRUE.
You will conduct titrations again in Experiment 6. What will be used to measure the volume of titrant added to the solution?
drop counter
What will be used to measure pH?
pH meter connected to a LabQuest
What indicator will be used to visually observe the endpoint of the titration?
Indicator is not used.
In which ports on the LabQuest should you connect the drop counter and pH probe, respectively? It is important that you do not plug the probe into the incorrect port.
DIG, CH
A titration required 11.5 mL of 0.060 M KOH added to 15.0 mL of HClO2 to reach the equivalence point. What is the concentration of HClO2? What is the pH at the equivalence point?
0.046 M, pH > 7
How did you determine the equivalence point for each titration?
mid-point of rapid-rise section of the plot
What will be the shape of the titration curve if oxalic acid, HO2C2O2H, is titrated with LiOH?
slanted S-shape with an inital pH < 7
In a different experiment, nitric acid (HNO3) is titrated with potassium hydroxide (KOH). What species are in solution at the equivalence point? What is the pH?
KNO3 and H2O only, pH = 7
What is the conductivity classification for an unknown F that dissolves completely in water and doesn’t have any ions present in solution?
Nonelectrolyte
What is the conductivity classification for an unknown B that dissolves completely in water and dissociates completely into ions?
Strong electrolyte
What is the conductivity classification for an unknown H that only partially ionizes in water?
Weak electrolyte
Predict the conductivity of HCOOH, a weak acid.
Weak electrolyte
Predict the conductivity of NH3, a weak base.
Weak electrolyte
In the reaction below, classify the chemical species as the type of electrolyte.
Ba(OH)2(aq) + H2SO4(aq) → BaSO4(s) + 2 H2O(ℓ)
H2O
Nonelectrolyte
In the reaction below, classify the chemical species as the type of electrolyte.
Ba(OH)2(aq) + H2SO4(aq) → BaSO4(s) + 2 H2O(ℓ)
Ba(OH)2
Strong electrolyte
Which statements explain the shape of the titration curve observed in lab?
I. Initially, the conductivity is zero since the reaction has not begun.
II. During the reaction, the conductivity decreases due to the production of nonelectrolytes.
III. During the reaction, the conductivity decreases due increase of ions.
IV. After the endpoint, the conductivity increases due to the excess titrant.
II and IV are TRUE
What is the Molarity of the Ba(OH)2 solution if a 7.50 mL aliquot of the Ba(OH)2 solution required 14.30 mL of a 0.108 M H2SO4 solution to reach the equivalence point?
Ba(OH)2(aq) + H2SO4(aq) → BaSO4(s) + 2 H2O(l)
0.206 M
In this experiment, you will be using the gas constant, R. Which unit is NOT found in R? (Exp. 7)
molarity
In this experiment, you will be using and measuring various chemical species by a variety of units. Which units of measure is used for the volume of hydrogen peroxide?
milliliter
Consider an experiment where 4.55 mL of an unknown H2O2(aq) solution reacted with the yeast at 19.1°C to produce 98.10 mL of gas.
The barometric pressure was 786.1 torr. The vapor pressure of H2O is 16.5 torr at that temperature.
What is the partial pressure of O2 (in atm) in the collected gas?
1.0126 atm O2
Consider an experiment where 4.55 mL of an unknown H2O2(aq) solution reacted with the yeast at 19.1°C to produce 98.10 mL of gas.
The barometric pressure was 786.1 torr. The vapor pressure of H2O is 16.5 torr at that temperature.
How many moles of O2 were produced by the reaction?
0.004142 mol O2
Consider an experiment where 4.55 mL of an unknown H2O2(aq) solution reacted with the yeast at 19.1°C to produce 98.10 mL of gas.
The barometric pressure was 786.1 torr. The vapor pressure of H2O is 16.5 torr at that temperature.
How many moles of H2O2 reacted to produce this amount of O2 ?
0.008284 mol H2O2
Consider an experiment where 4.55 mL of an unknown H2O2(aq) solution reacted with the yeast at 19.1°C to produce 98.10 mL of gas.
The barometric pressure was 786.1 torr. The vapor pressure of H2O is 16.5 torr at that temperature.
What is the Molarity of the H2O2 solution?
1.82 M H2O2
Piers & Aida performed this experiment. Aida read the volume of oxygen collected as 77.5 mL. However, several more bubbles came over and Piers read the volume of oxygen collected as 81.3 mL. Which of the following statements are TRUE about Aida’s calculations?
I. Aida’s calculations will give a lower value of moles of O2(g) than Piers.
II. Aida’s calculations will give a lower partial pressure of O2(g) than Piers.
III. Aida’s calculations will give a higher molarity of H2O2 than Piers.
Only I is true.
Ozzie wanted to do another experiment with a stronger H2O2 solution to check the accuracy of the experiment by calculating the theoretical volume of O2(g) it would produce. Then he could compare his experimental volume of O2(g) to the theoretical volume of O2(g).
He used 5.20 mL of 2.21 M H2O2 and the partial pressure of O2 was 0.9341 atm and the temperature was 298.35 K.
What volume of O2(g) could he theoretically produce (in mL)?
151 mL
Ozzie wanted to do another experiment with a stronger H2O2 solution to check the accuracy of the experiment by calculating the theoretical volume of O2(g) it would produce. Then he could compare his experimental volume of O2(g) to the theoretical volume of O2(g).
He used 5.20 mL of 2.21 M H2O2 and the partial pressure of O2 was 0.9341 atm and the temperature was 298.35 K.
Ozzie wanted to do another experiment using 8.75% (by mass) H2O2.
What is the Molarity of this H2O2 solution ?
2.57 M
% (by mass) = Molarity H2O2 x Molar Mass × (1 / (Density) × (1 L/1000 mL) × 100%
Molarity = % (by mass) / 100 x Density × (1/Molar mass) x (1000 mL/1L)
A 63.0 mg sample of Red 40 dye was dissolved and diluted to a final volume of 0.500 L with deionized water.
What is the concentration of the Red 40 dye solution in ppm?
parts per milliion (ppm) refers to 1 mg of solute in 1.0 L of solution; (mg/L)
126 ppm