Real AAMC stuff to review - CP
1/73
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
74 Terms
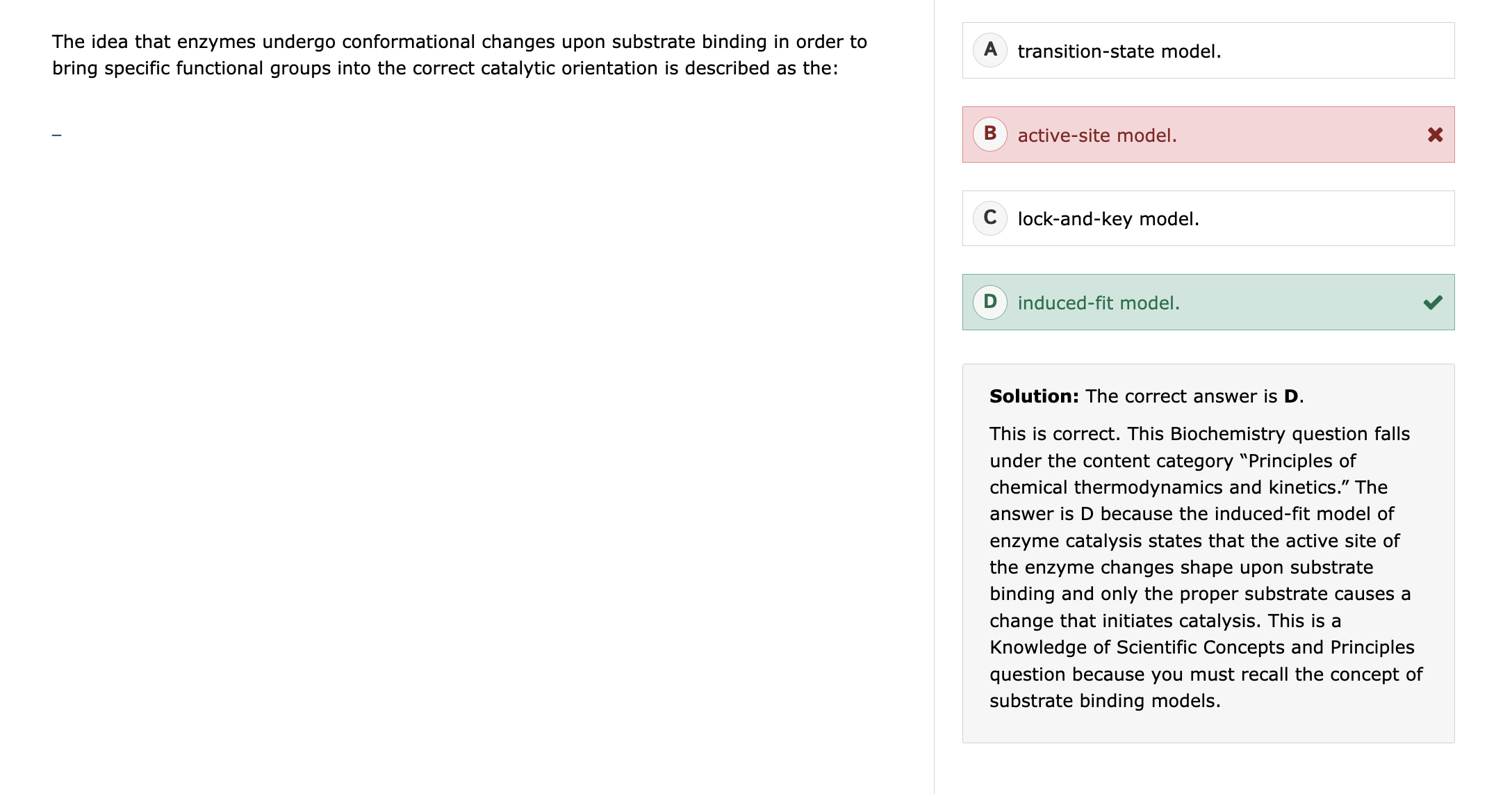
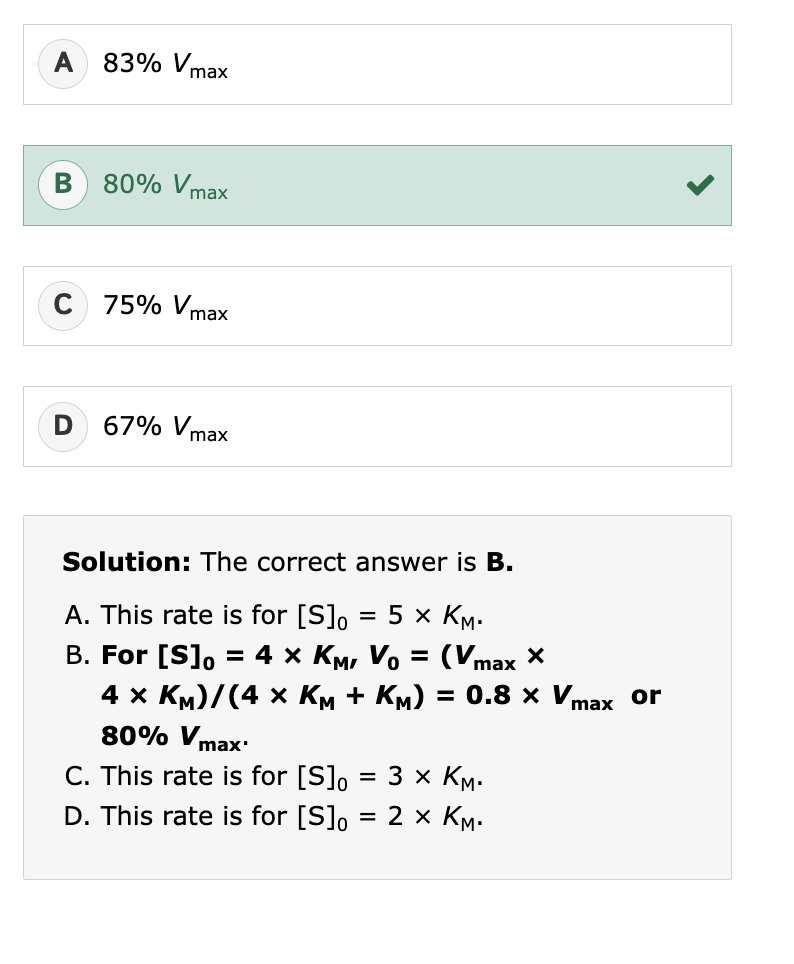
gas-liquid chromatography, the first peak to emerge will be:
the least polar, most volatile compound
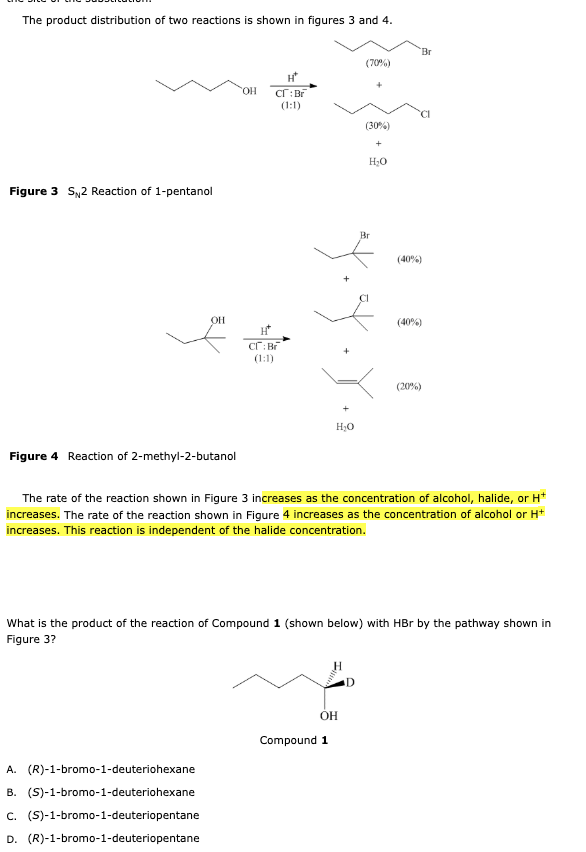





standard atmospheric pressure is:
Atmosphere (atm) – 1 atm
Millimeters of mercury (mmHg or Torr) – 760 mmHg
Pascal (Pa) – 101,325 Pa


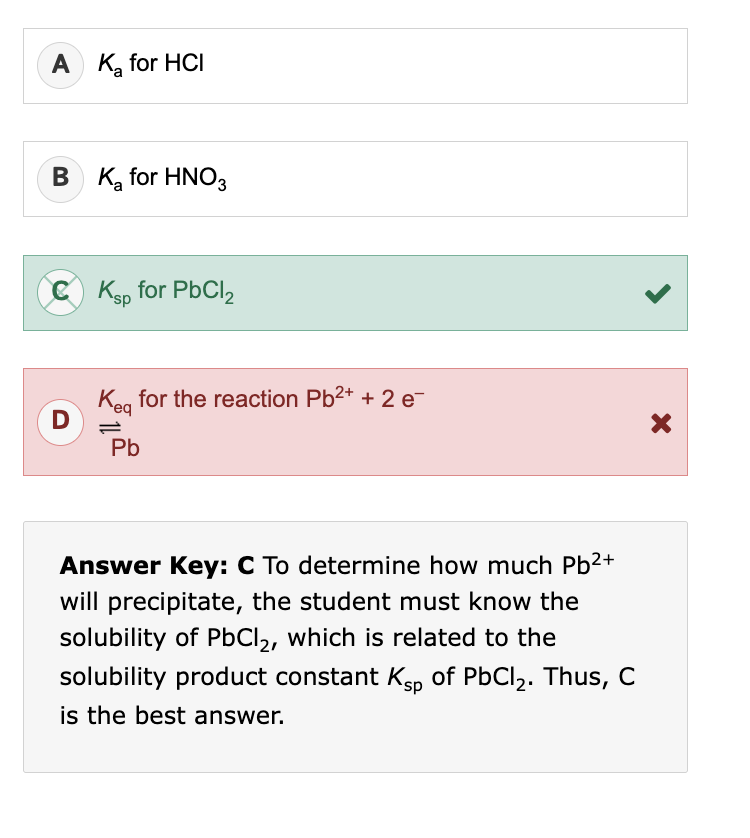
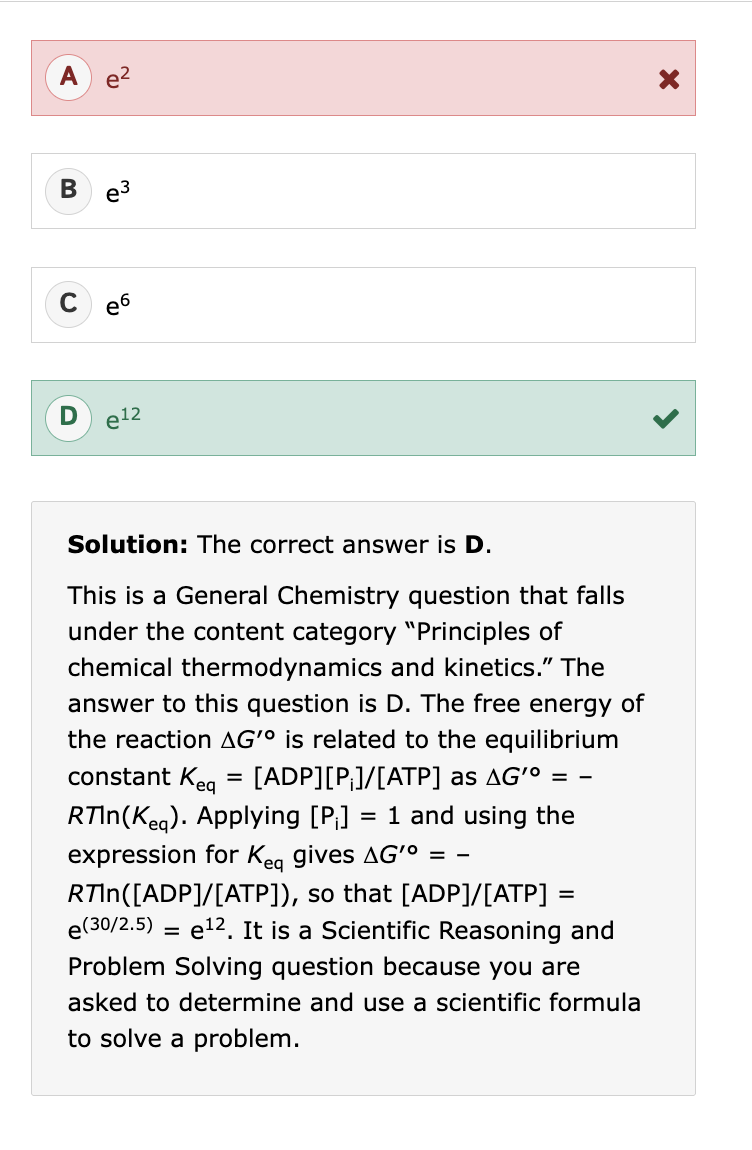
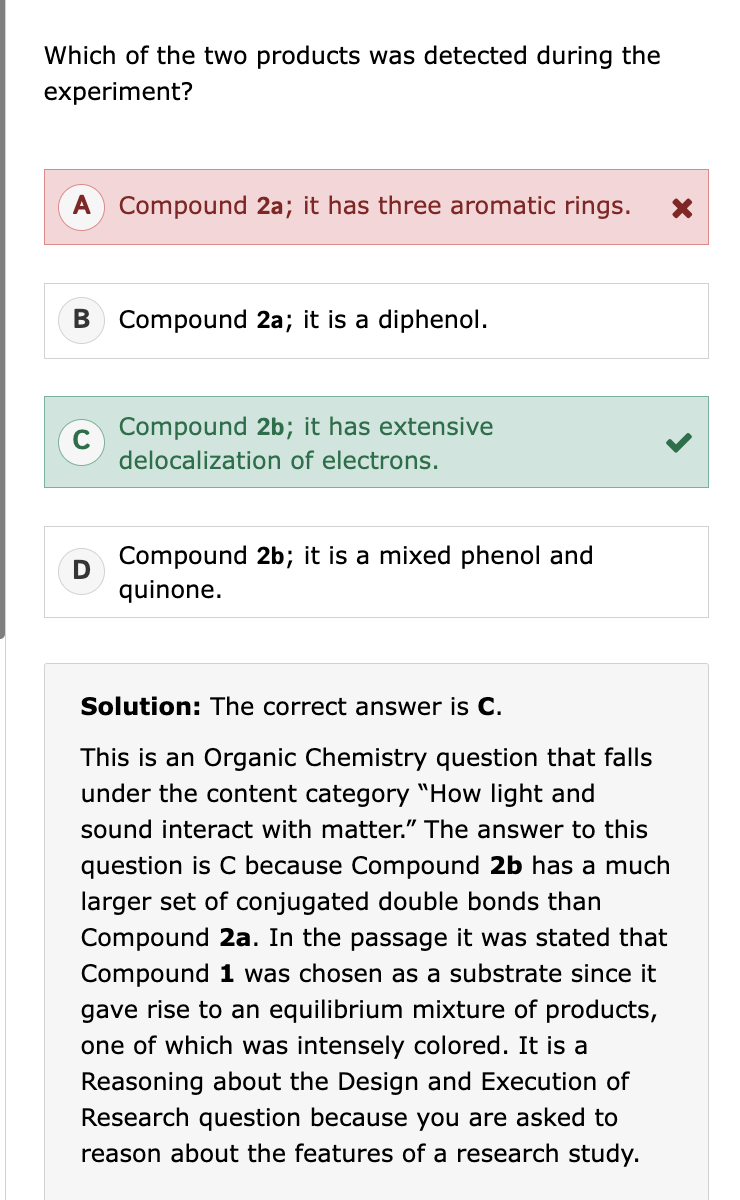
“Oxidation of self-assembled, micellular Compound 1 with iodine resulted in a solid material (Compound 2) that was stable to alkaline solutions (Equation 2)” this means:
S-S bonds
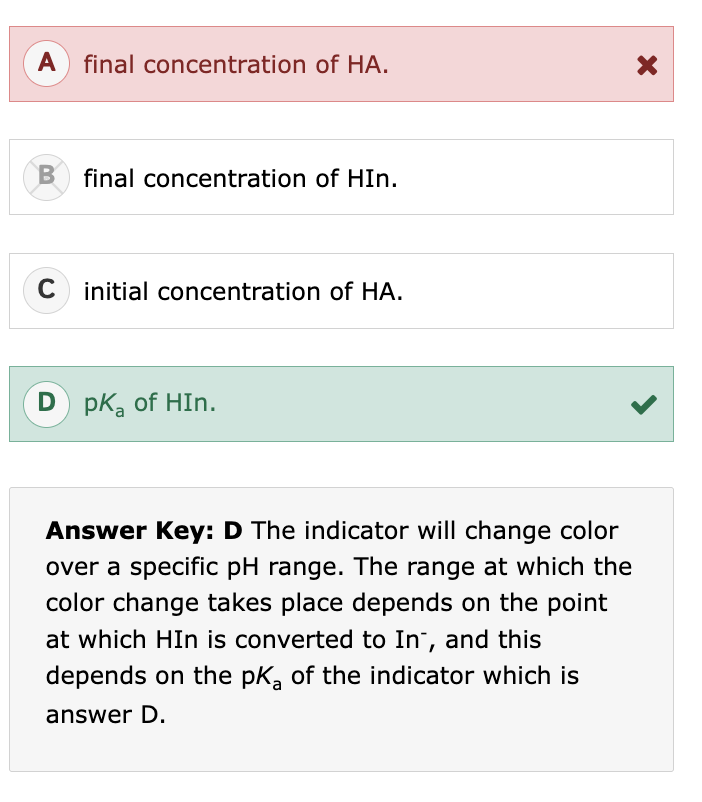
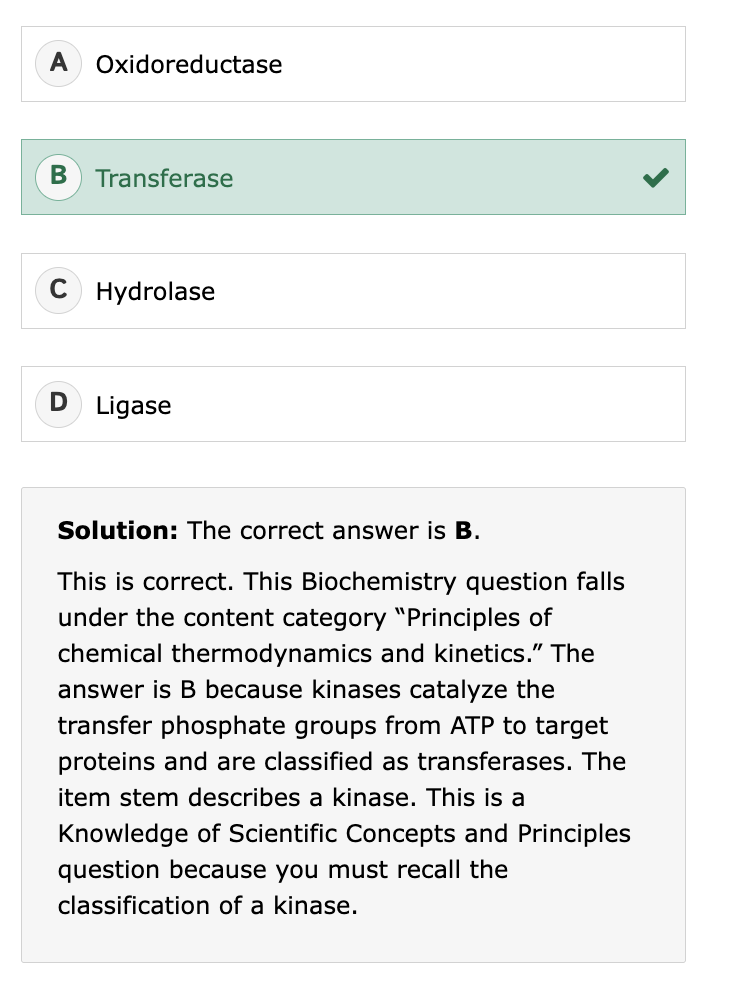
phosphorylation occurs the most efficiently with:
primary alcohols




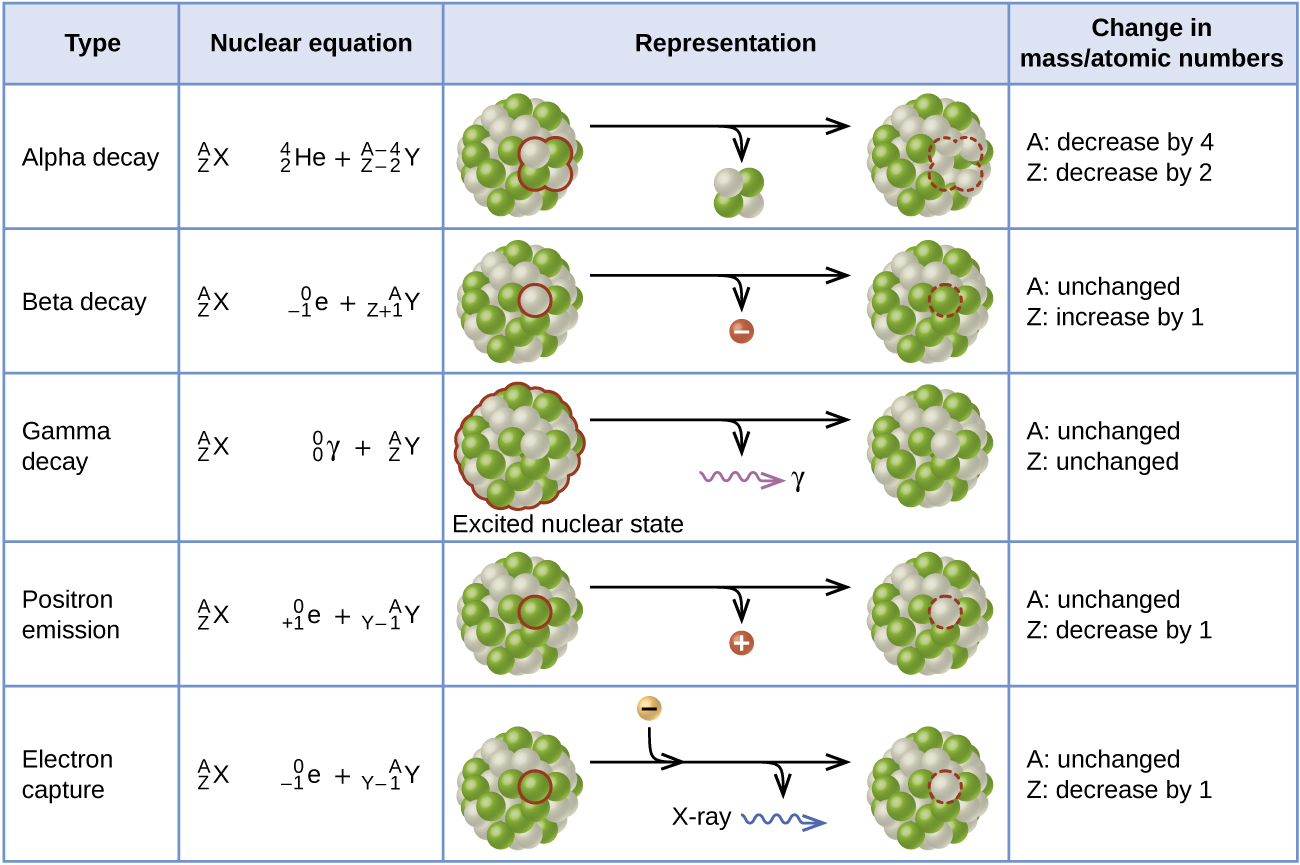
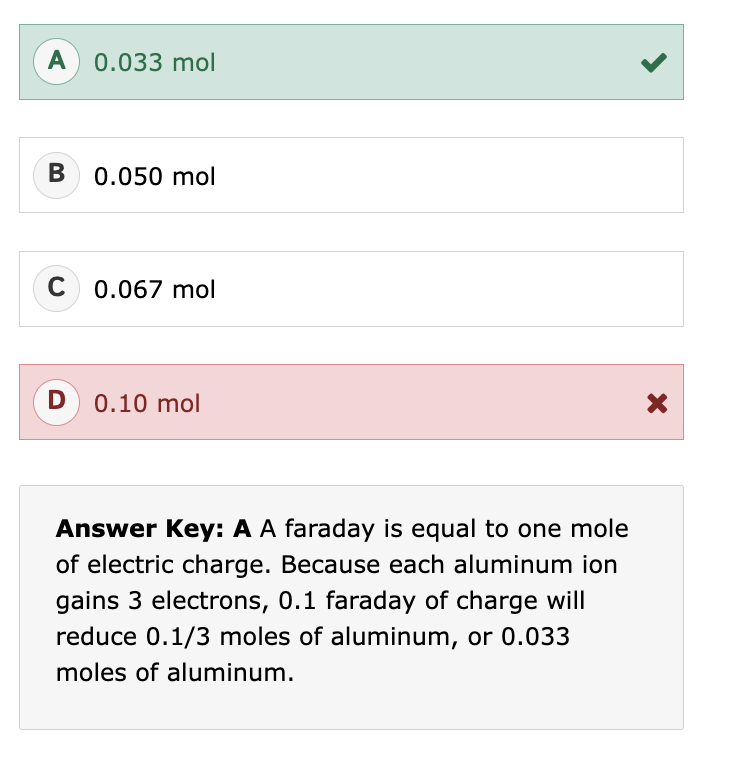
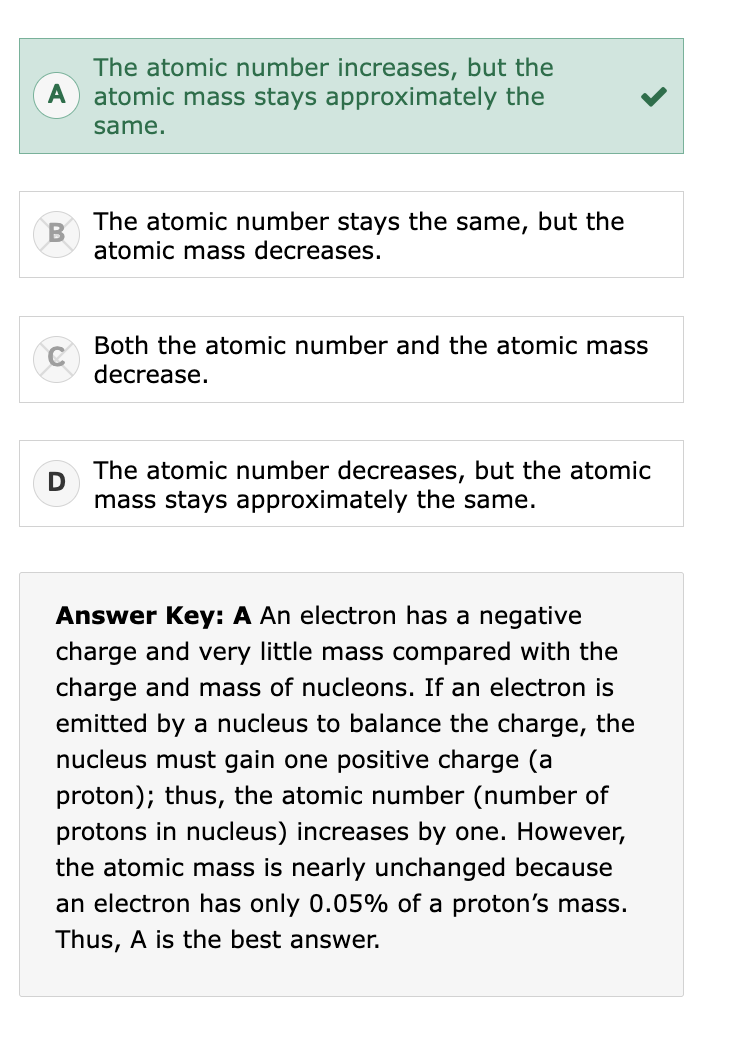
Radioactive Decay






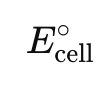
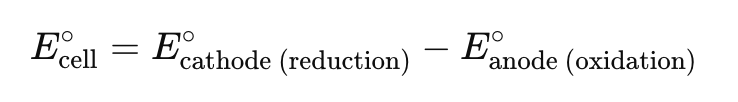
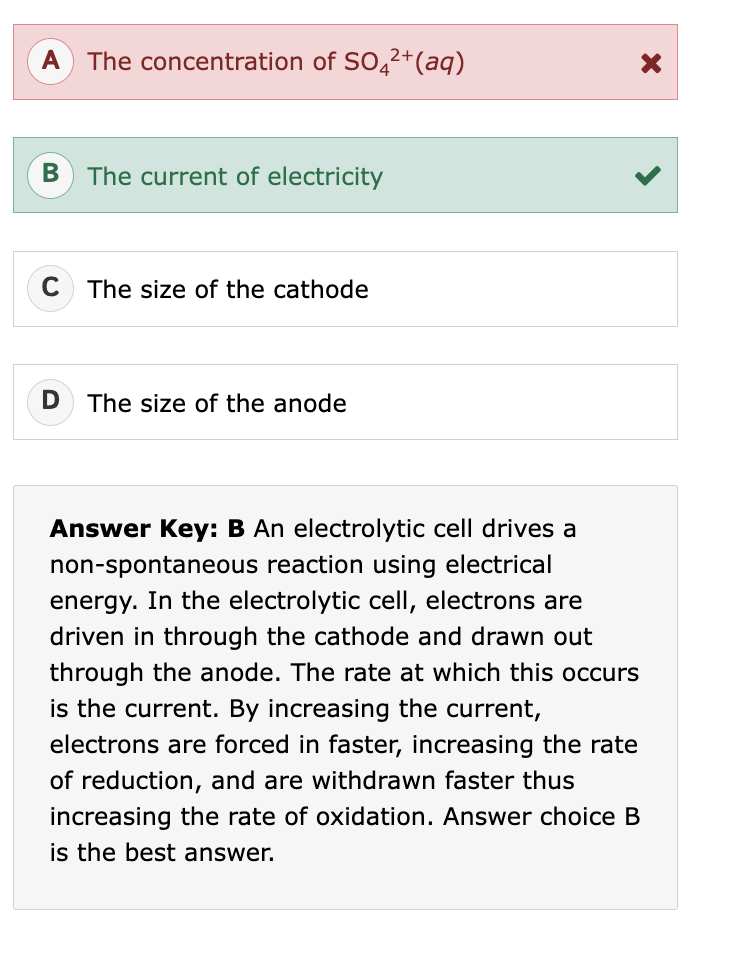
Voltaic Cell where does oxidation/reduction occur?








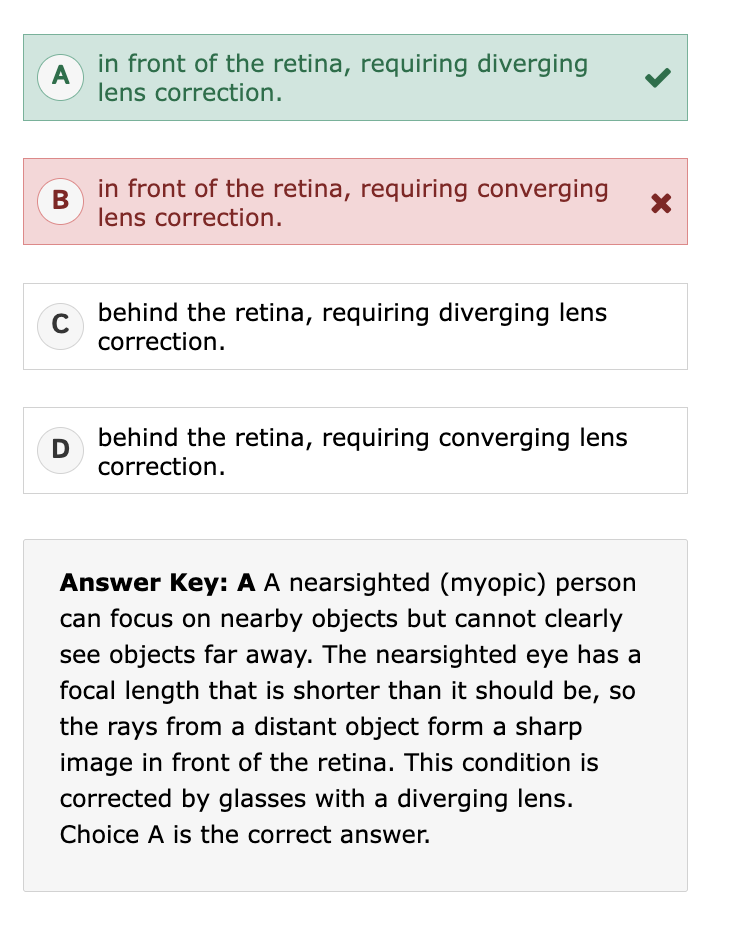
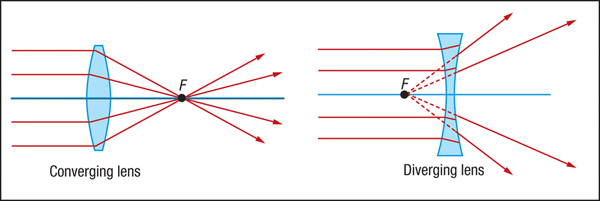
converging vs diverging lens

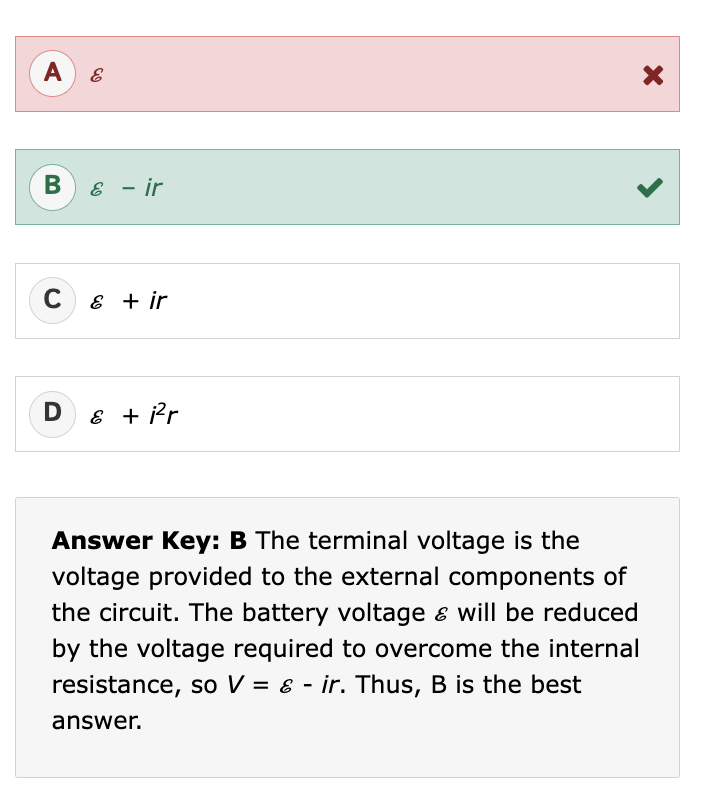





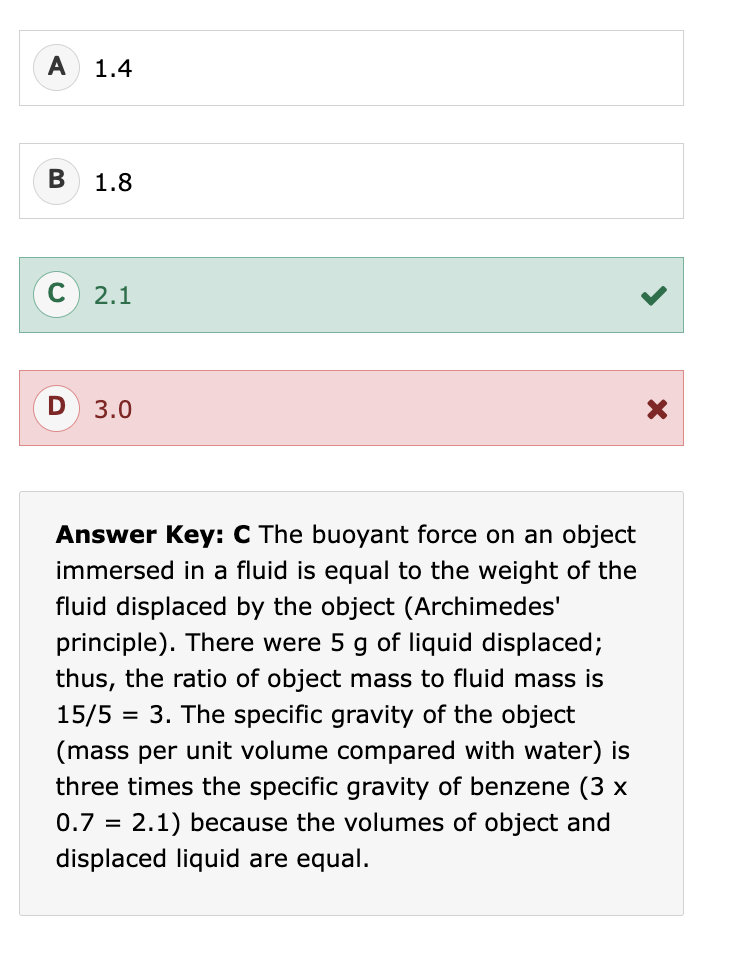



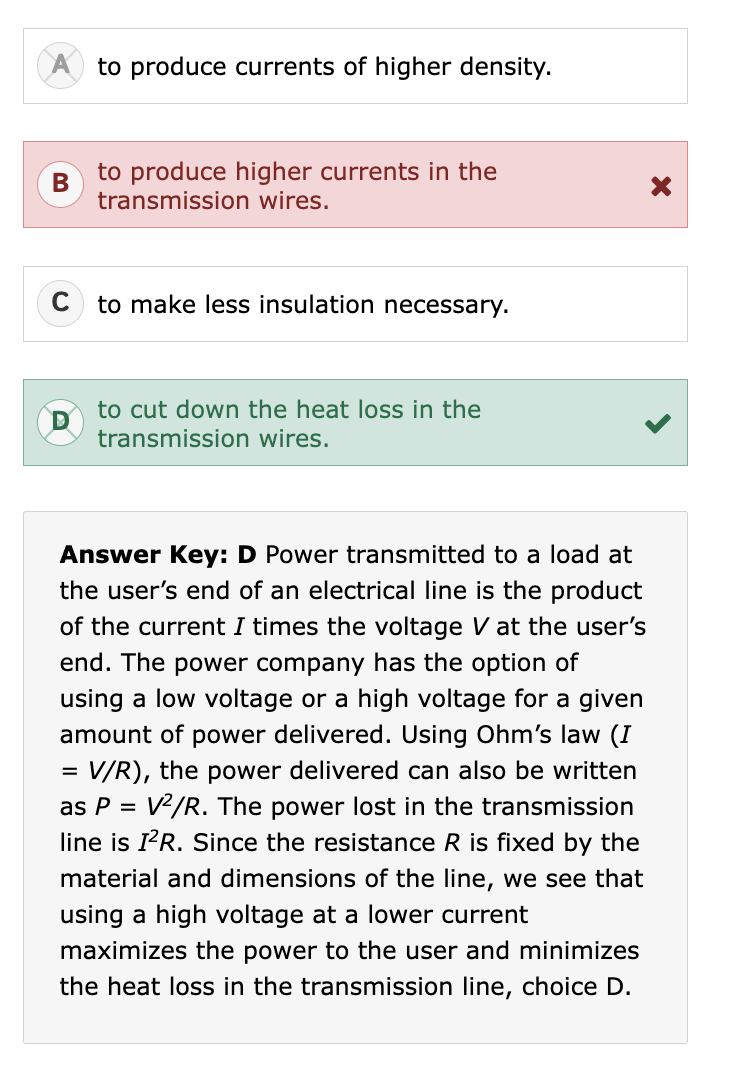

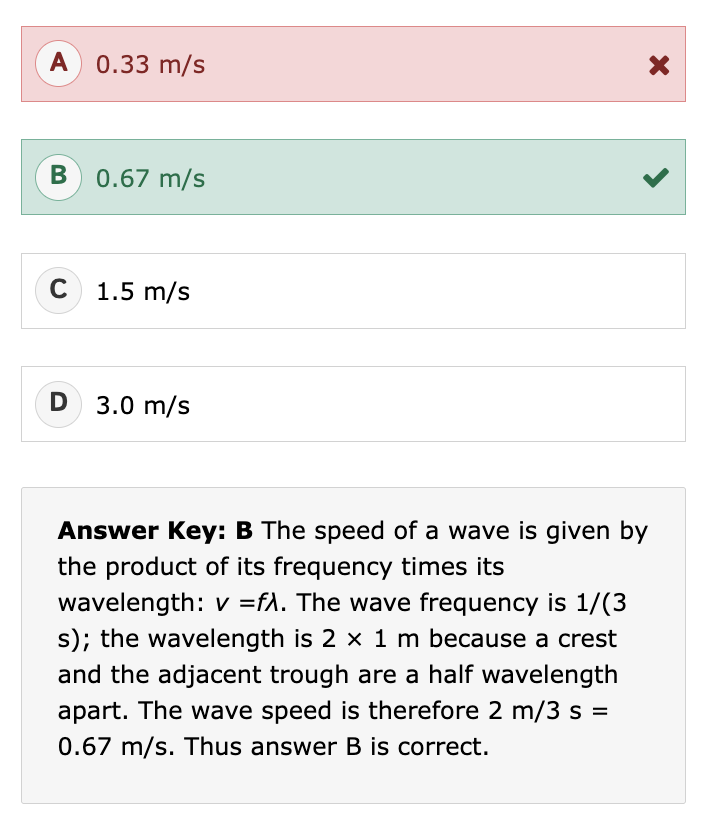

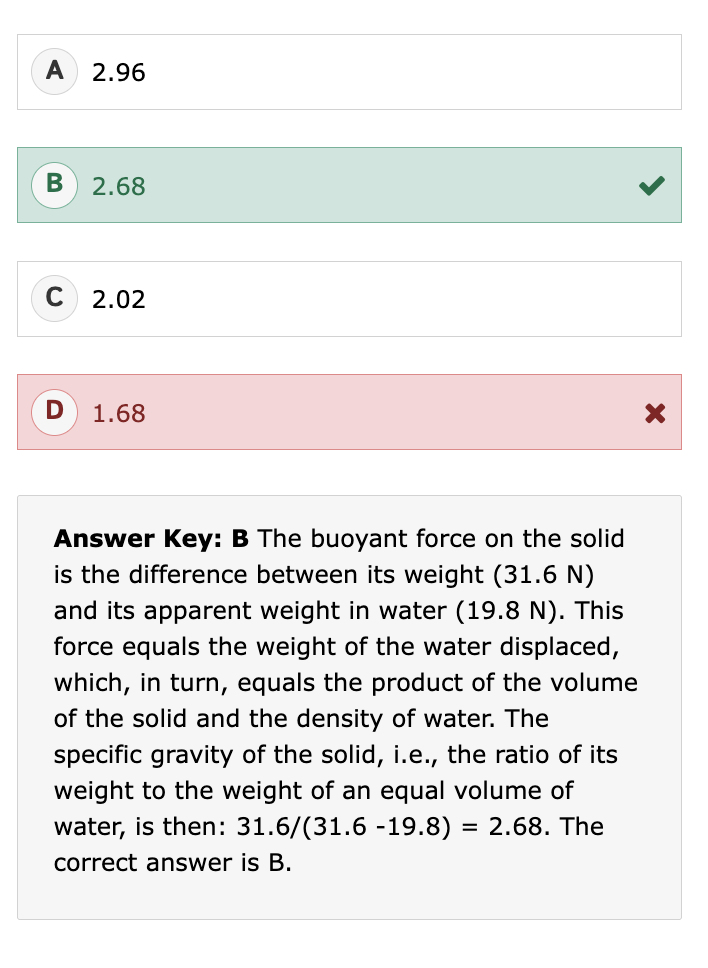
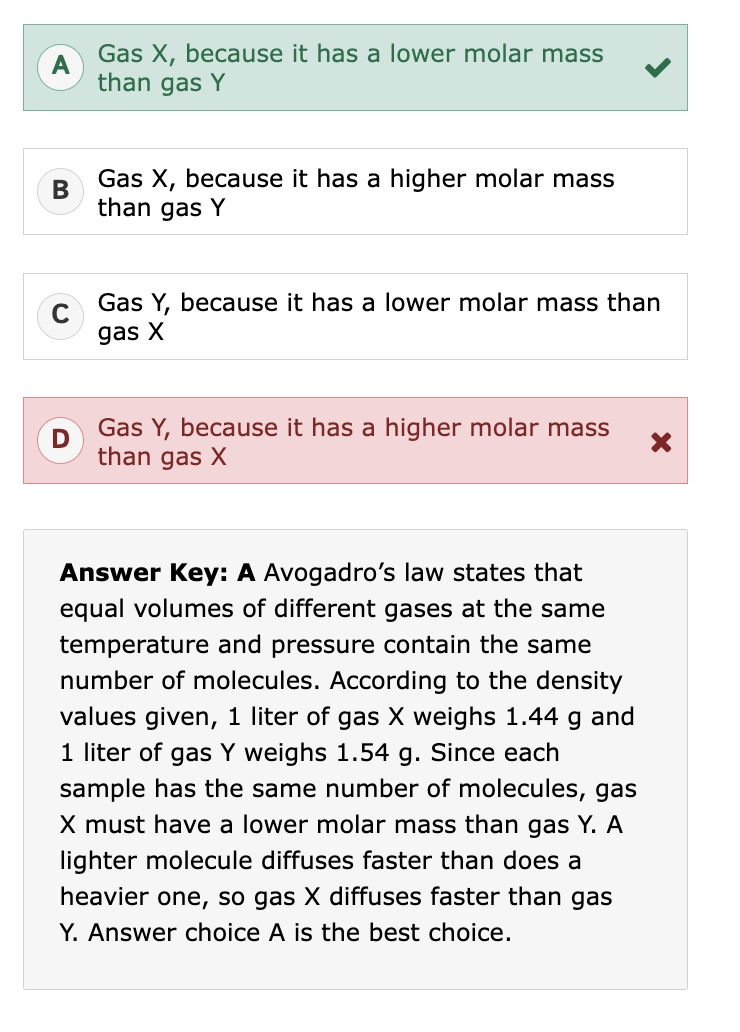
What is specific gravity (SG)?
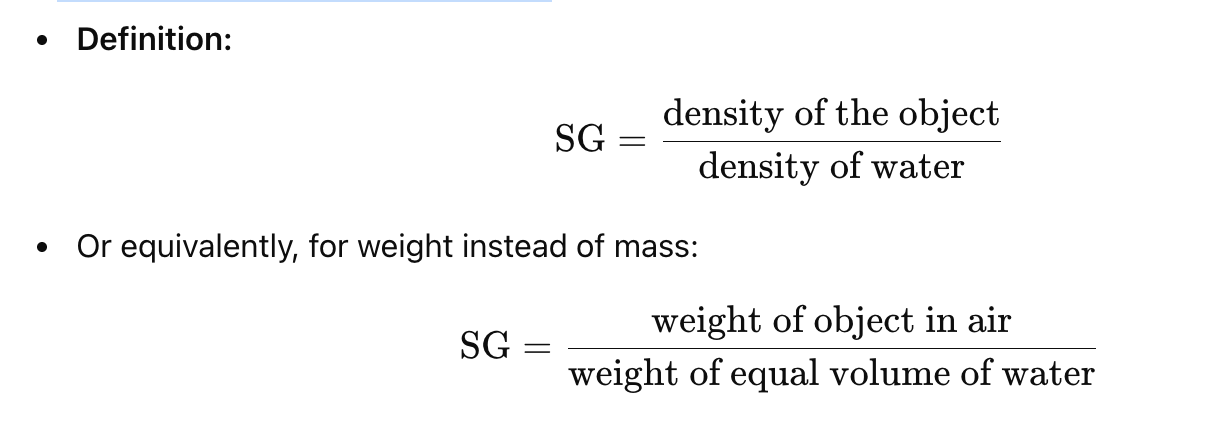
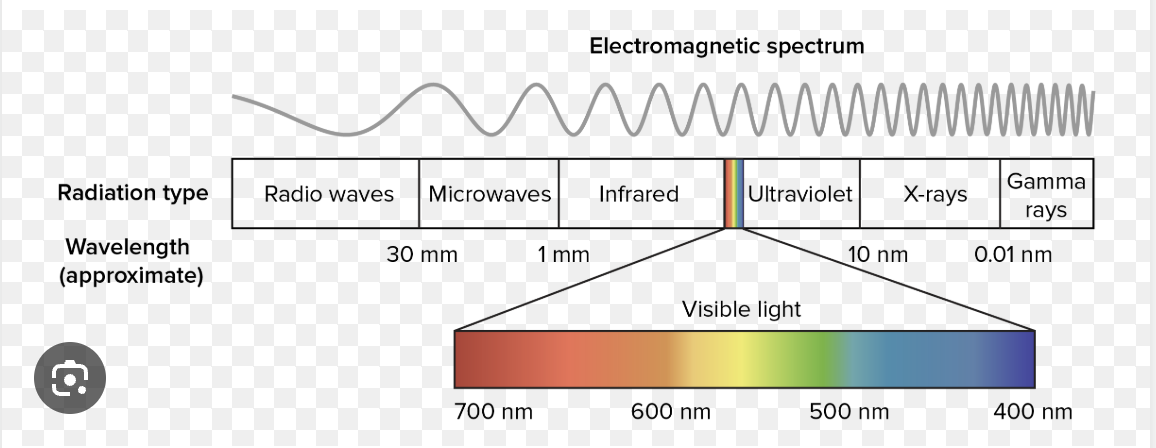
Light wavelengths





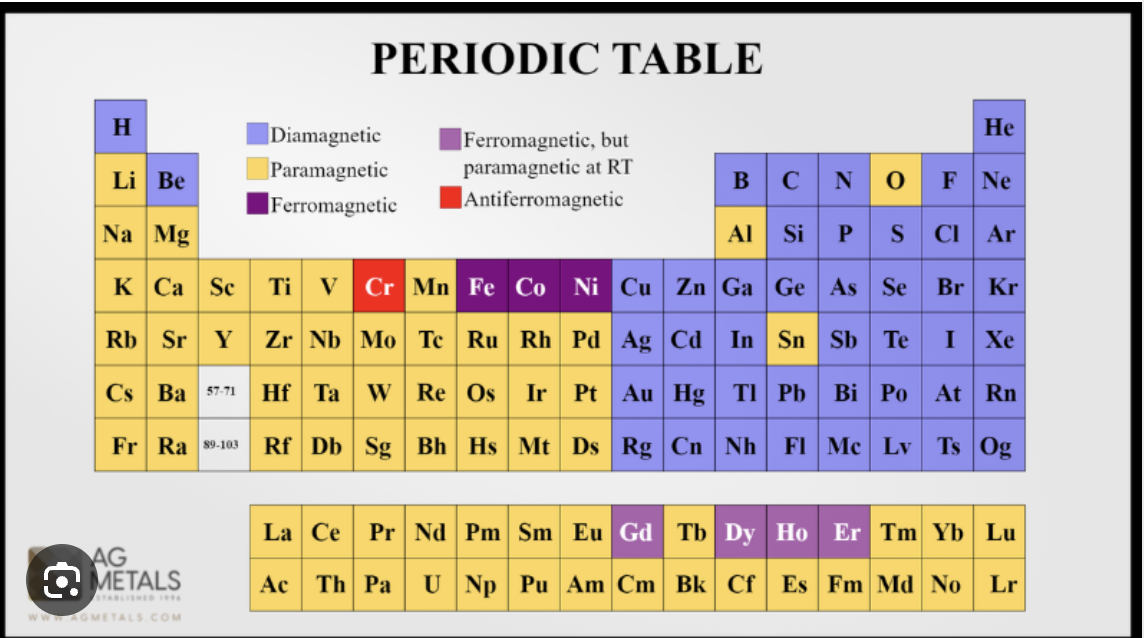
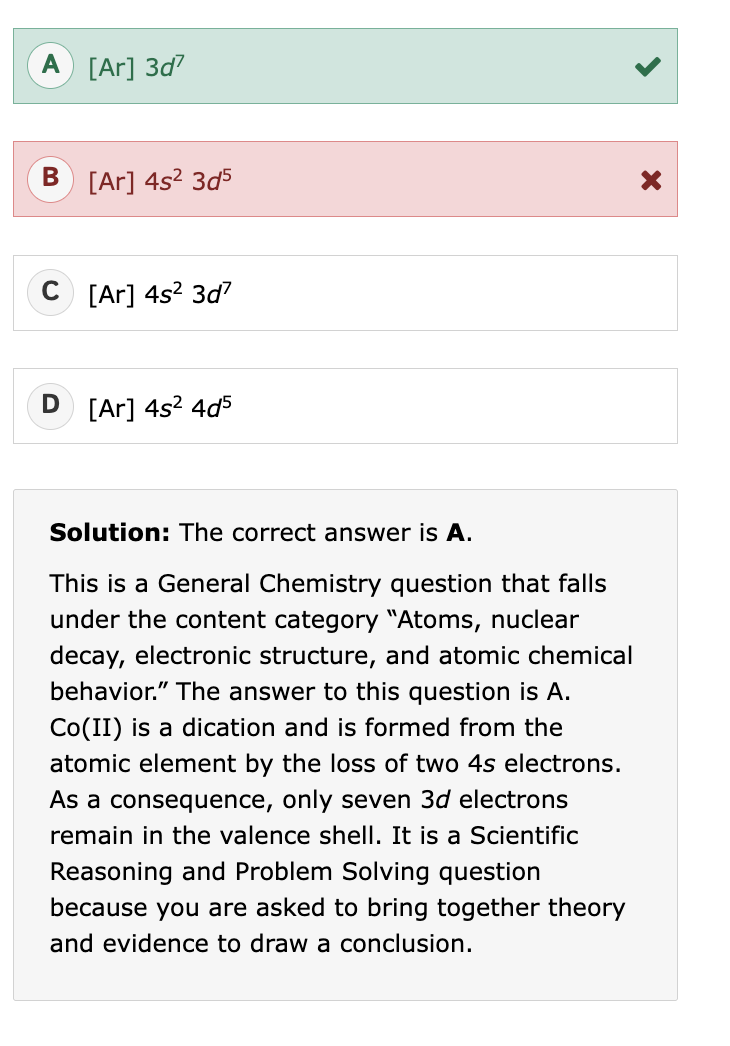
Periodic table :)




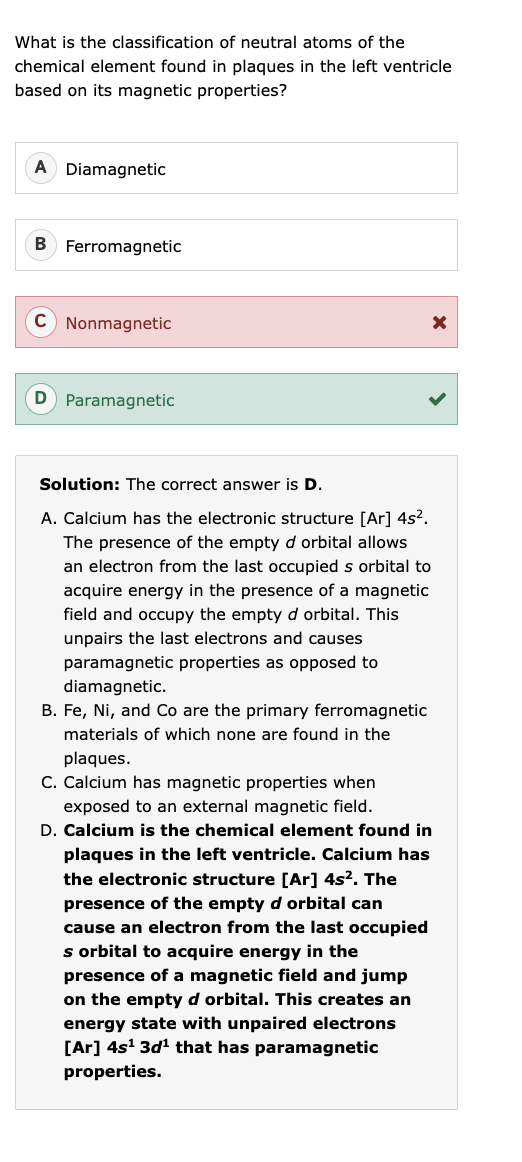
magnetic properties periodic table
ATP phosphates

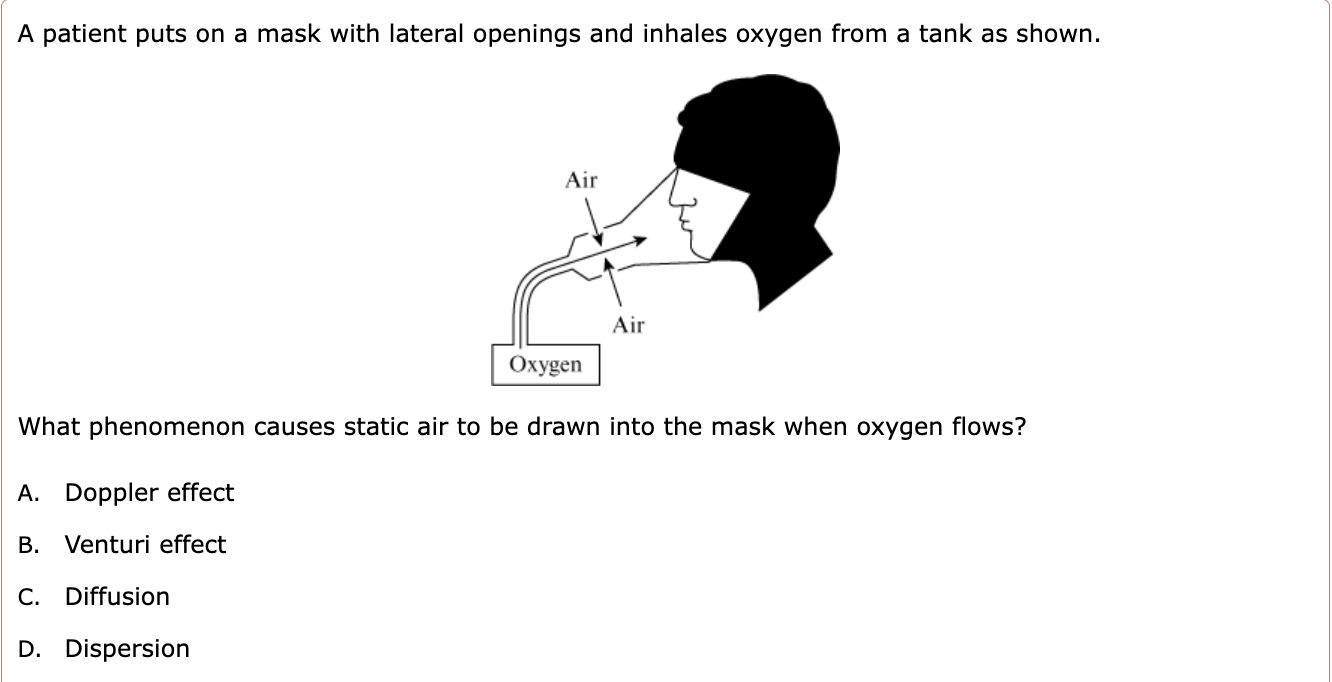

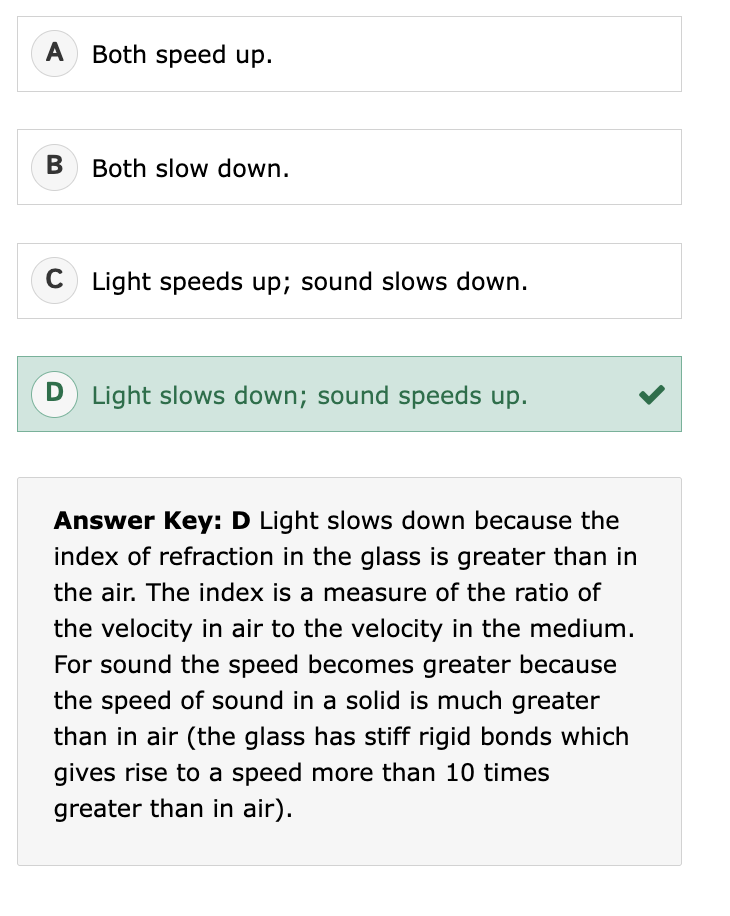
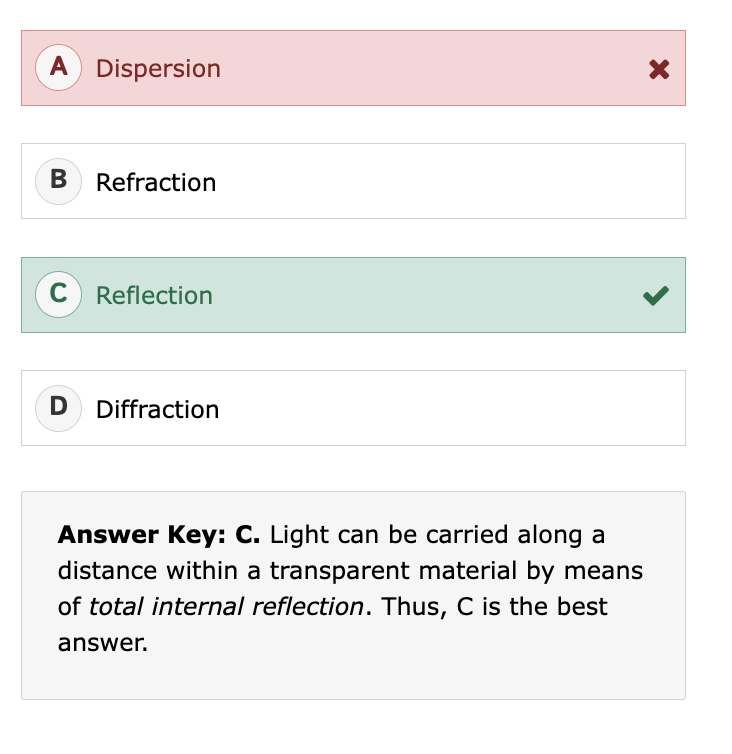
What is the difference between diffusion and dispersion?
Diffusion:
Movement of particles from high → low concentration
Driven by random motion
Goal: even distribution
Example: perfume spreading in a room
Dispersion:
Separation of waves (usually light) by wavelength
Caused by different speeds of components
Goal: separate components
Example: rainbow from a prism
MCAT memory trick:
Diffusion = “DIFFerence in concentration”
Dispersion = “DISPersing light into colors”
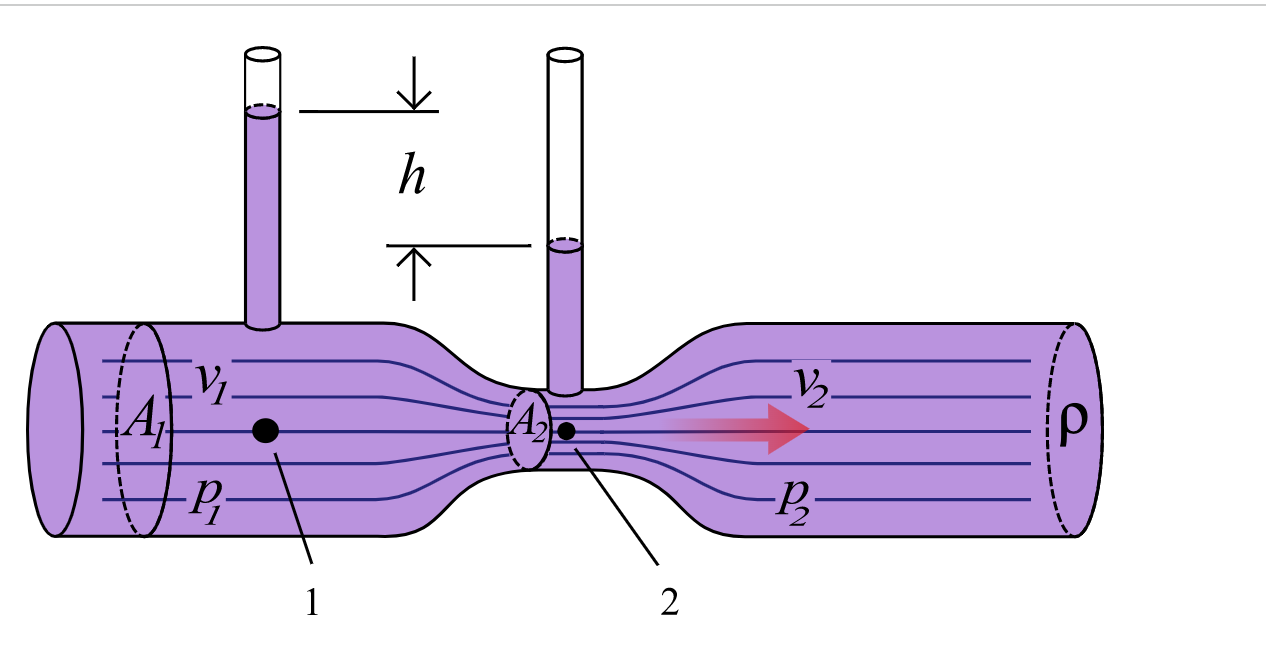
Venturi Effect




How can you tell if a bond is a coordinate (dative) covalent bond?
**Coordinate covalent bond = covalent bond where BOTH shared electrons come from one atom.
How to spot it:
Look for an atom with a lone pair (common: N, O, S, halogens)
Look for an electron-deficient atom (common: H⁺, metal cation)
If the lone pair forms a bond without the other atom contributing electrons, it’s coordinate covalent
Visual trick: Draw an arrow → from the donating atom to the acceptor
Examples:
NH₃ + H⁺ → NH₄⁺ (N donates lone pair)
CO → Fe²⁺ (C donates lone pair to Fe²⁺)
Memory trick:
“Someone gives both their electrons → coordinate covalent”






















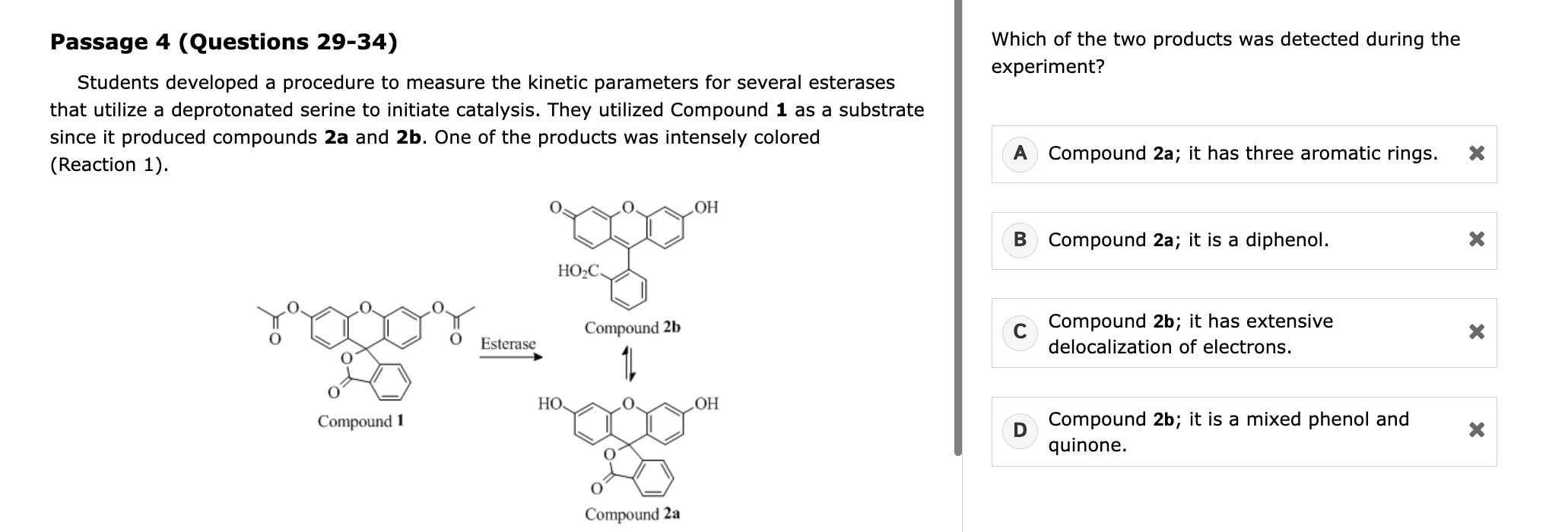
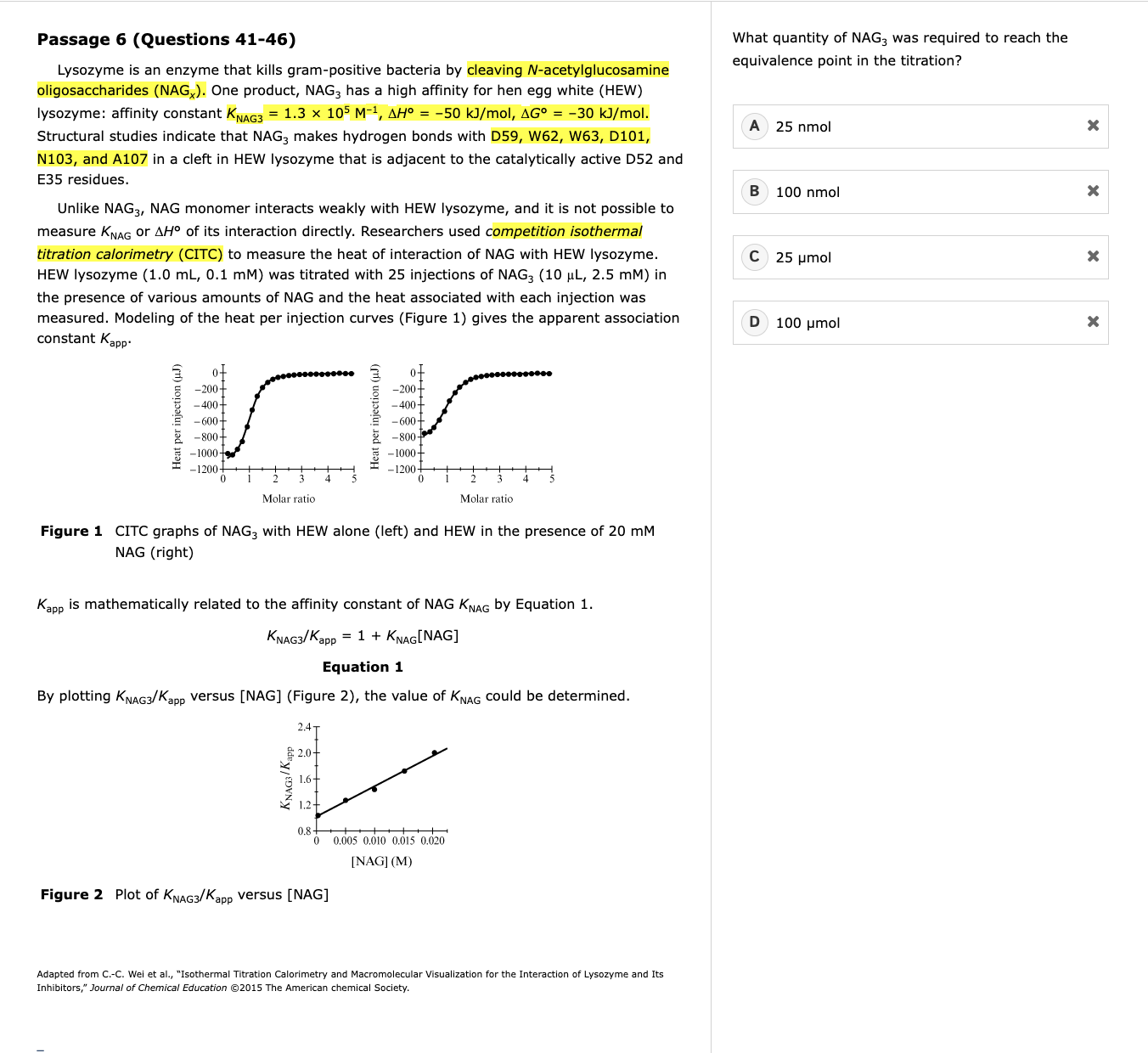
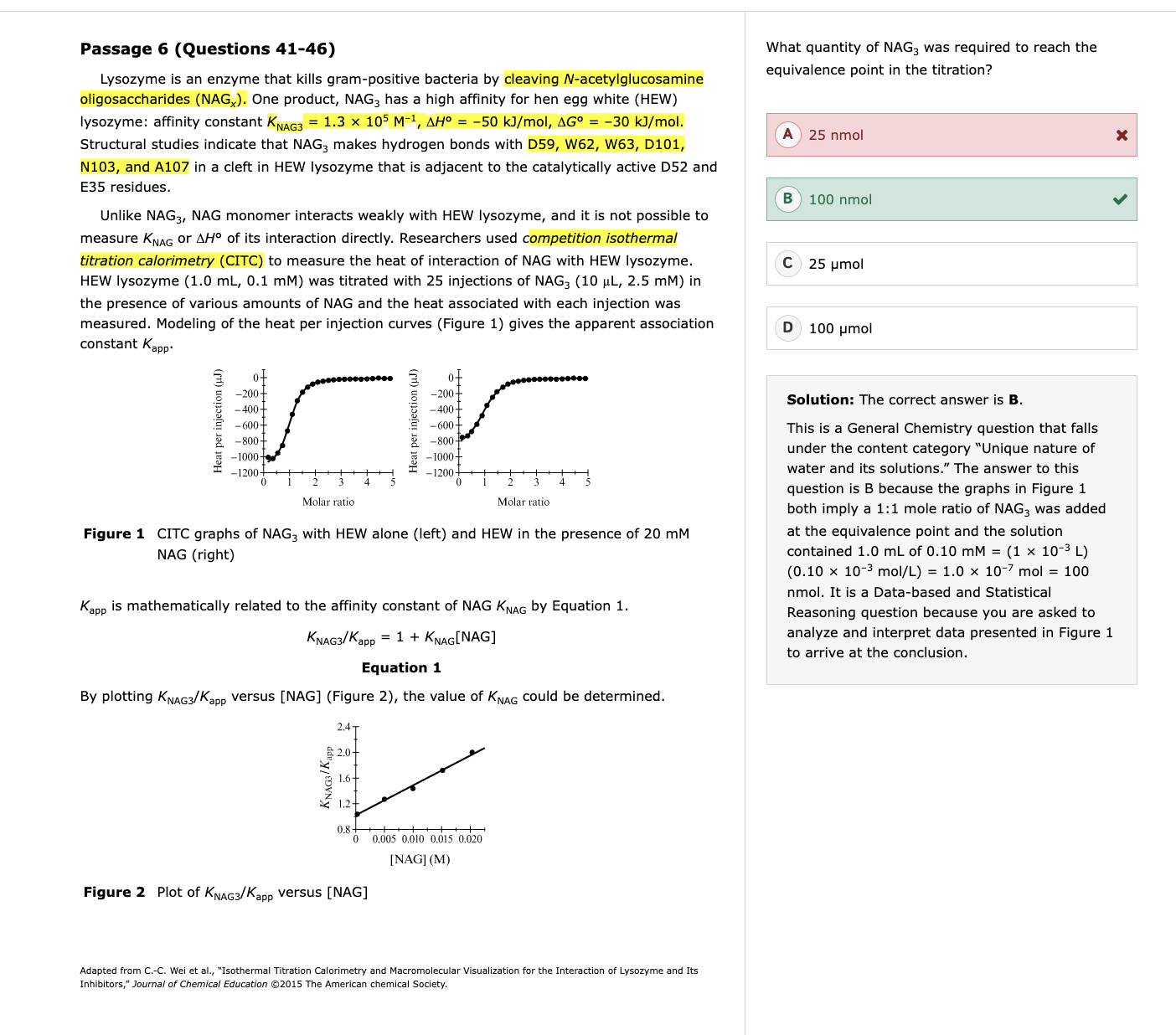
ITC and CITC
Understand what ITC measures – it detects the heat released or absorbed when a ligand binds a protein.
Recognize how competition affects it (CITC) – a competitor reduces available binding sites, so injections of your ligand produce less heat.
Translate experimental manipulations into outcomes – e.g., more competitor → fewer free sites → smaller measured ΔH → lower apparent binding constant Kapp.