Organic Chemistry Test 2 Reaction Summaries
1/71
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
72 Terms
What oxidizes primary OH to aldehydes
PCC, or Dess-Martin/periodinane, or 1. DMSO, (COCl)2/2. Et3N
what oxidizes secondary alcohols to ketones
Cro3/H3O+, or H2CrO4, or Na2Cr2O7/H3O+, or 1. hot KMnO4, NaOH, H2O/2. H3O+ (chromic acid)
Where does a ketone come from in ozonolysis?
zero Hs
where does an aldehyde come from in ozonolysis
one H
where does a formaldehyde come from in ozonolysis?
two Hs
What does friedel crafts acylation do?
adds an acyl group onto a benzene ring, R-carbonyl
what does DIBALH always do?
make an aldehyde
what does LiH(t-BuO)3/H3O+ do?
this is DIBALHs brother so it makes an aldehyde
what does NaBH4/EtOH do in the presence of an aldehyde or ketone
it reduces to a primary alcohol or a secondary alcohol, respectively
what does LiAlH4/ H2O do with an amide or a nitrile?
forms an amine (NH2)
what is the wolff-kishner reduction
completely removes a carbonyl group and turns it into a CH2 group by using H2NNH2/KOH, heat
How does grignards reagent (1. CH3MgBr/2. H3O+) do in the presence of ketone or aldehyde?
usually forms an alcohol and addition of methyl (or whatever it is paired with) to where the carbonyl was.
what occurs with the addition of water to an aldehyde or ketone?
In basic (NaOH,H2O) or acidic (H3O+) conditions to create a gem diol. The carbonyl form is favored in equilibrium (aldehydes are preffered, and least crowded)
What occurs in a cyanohydrin formation?
addition of HCN to an aldehyde or ketone
Nitriles are hydrolyzed using H3O+ and heat into what?
a carboxylic acid
What does an aldehyde/ketone with a primary amine form?
and imine (a double bonded N with a methyl)
What does a ketone or aldehyde form when reacted with a secondary amine (NH)
an enamine which is a N with groups and a double bond one bond away from the nitrogen.
What happens when a ketone or aldehyde is reacted with an alcohol or diol?
Formation of an acetal (from aldehyde because H is still left) or ketal (from ketone because no H) and the Os add where the carbonyl was
How are acetals/ketals removed?
Use Cat. HCl/H2O or use H3O+ to revert back to carbonyl
What is the wittig reaction?
a ketone or aldehyde reacts with the phosphorous ylide to create an alkene.
What would cause the wittig reaction to form a Z alkene?
if the ylide has no EWG (usually smaller groups cause no steric hinderance, and the ylide is unstabilized)
What causes the wittig reaction to form the E alkene?
The ylide is stabilized with an EWG so it needs more room to spread out.
What is tollens reagent and what does it do?
Tollens reagent is Ag(NH3)2OH and it is paired with H3O+ to see if an aldehyde is present as a silver precipitate will form if it is present.
What reagents are used to oxidize aldehydes to carboxylic acids?
1. KMnO4, OH-/2. H3O+ or 1. Ag2O, OH-/2. H3O+
What is the thioacetal formation?
this is just like the acetal formation just with SH reacting as S is replacing the carbonyl.
what occurs when raney Ni is used in the presence of H2?
desulfurization occurs (sulfurs are removed and just the carbons are left)
how is acidity increased?
electronegative atoms in close proximity to a carboxylic acid or if the presence of EWGs
What happens when 1.Hot KMNO4, NaOH, H2O/2. H3O+ reacts in the presence of a primary alcohol?
a carboxylic acid is formed
what is produced when there are 0Hs in the presence of hot KMNO4 of the alkene?
a ketone is produced
what is produced when there is 1 h in the presence of hot KMNO4 of an alkene?
a carboxylic acid is formed
what is produced when there are benzylic hydrogens in the presence of hot kmno4?
benzoic acid is produced
what occurs when there is an alcohol or aldehyde in the presence of chromic acid?
they are oxidized to carboxylic acids (any reagent with Cr will perform this when water is present)
When grignards and carbon dioxide react, what is formed?
carboxylic acids
When a nitrile is reacted with acidic hydrolysis (H2SO4/H2O) or basic hydrolysis (NaOH,H2O/H3O+) what is formed?
a carboxylic acid
what is the reactivity chart of carboxylic acid derivatives?
carboxylate < amide < carboxylic acid = ester < thioester < acid anhydride < acid chloride
What usually happens when there is a reactant in the presence of LiAlH4?
alcohols are produced (there are some exceptions)
what happens when a carboxylic acid is reacted with SOCl2, or PCl3, or PCl5?
an acid chloride is created
What is the fischer esterification?
when a carboxylic acid is reacted with an alcohol an ester is created. (requires acidic conditions)
what is a lactone?
a lactone is essentially a looped ester in a 5 or 6 membered ring form
when a lactone breaks open what does it usually form?
some sort of alcohol is usually formed
what usually happens with an acid anhydride reaction?
the acid anhydrides split into two depending on what it is reacting with
how can acid anhydrides be formed?
by high heating carboxylic acids or dicarboxylic acids
how are lactones usually formed?
from hydroxycarboxylic acids often under acidic conditions
when an amide reacts with H2SO4/H2O (acidic conditions), what is formed?
a carboxylic acid and ammonium
what happens when amides react with NaOH/H2O (basic conditions)
a cabroxylate (a carbonyl with a O-) and amine
when a molecule containes an amine and an alcohol, and it is heated, what happens?
a ring forms usually with an NH on it
what is an amine protecting group?
a benzyl chloride in the presence of base
how are amines deprotected
by using palladium on carbon and hydrogen gas (Pd/C/H2)
what is formed when a primary amide reacts with SOCl2 or P4O10?
dehydration occurs and a nitrile is formed
what happens when a nitrile is reacted with a grignards
a variety of different functional groups can be formed such as a carbonyl
what happens when ketones and aldehydes react with a halogen under acidic conditions?
the alpha carbon is halogenated (ex. Br2/CH3COOH)
a, B carbonyls can be created via E2 elimination using what?
pyridine and heat
what happens when a methyl ketone is reacted with 1. excess NaOH, (halogen)/ 2. H3O+?
a carboxylic acid (or a carboxylate) and a haloform forms (CH3X)
what forms the kinetic enolate?
by using a strong,stericlly hindered base in an aprotic solvent (LDA in THF, or LiHMDS in THF, or LTMP in THF) and this is the less substituted alkene
what forms a thermodynamic enolate
a strong base and a protic solvent (such as NaOMe in MeOH, or NaOEt in EtOH, or NaOH in H2O), forms in the more substituted alkene
what is LDA commonly used to do?
often deprotonates the alpha carbon of a ketone or alpha carbon of an ester or aldehyde or nitrile
acetoacetic esters are used to make
ketone derivatives once decarboxylated
what are malonic esters used to make
carboxylic acid derivatives once decarboxylated
what can enamines be used to make?
alkylation or acylation because H2O or H3O+ to remove the iminium ion so it can leave
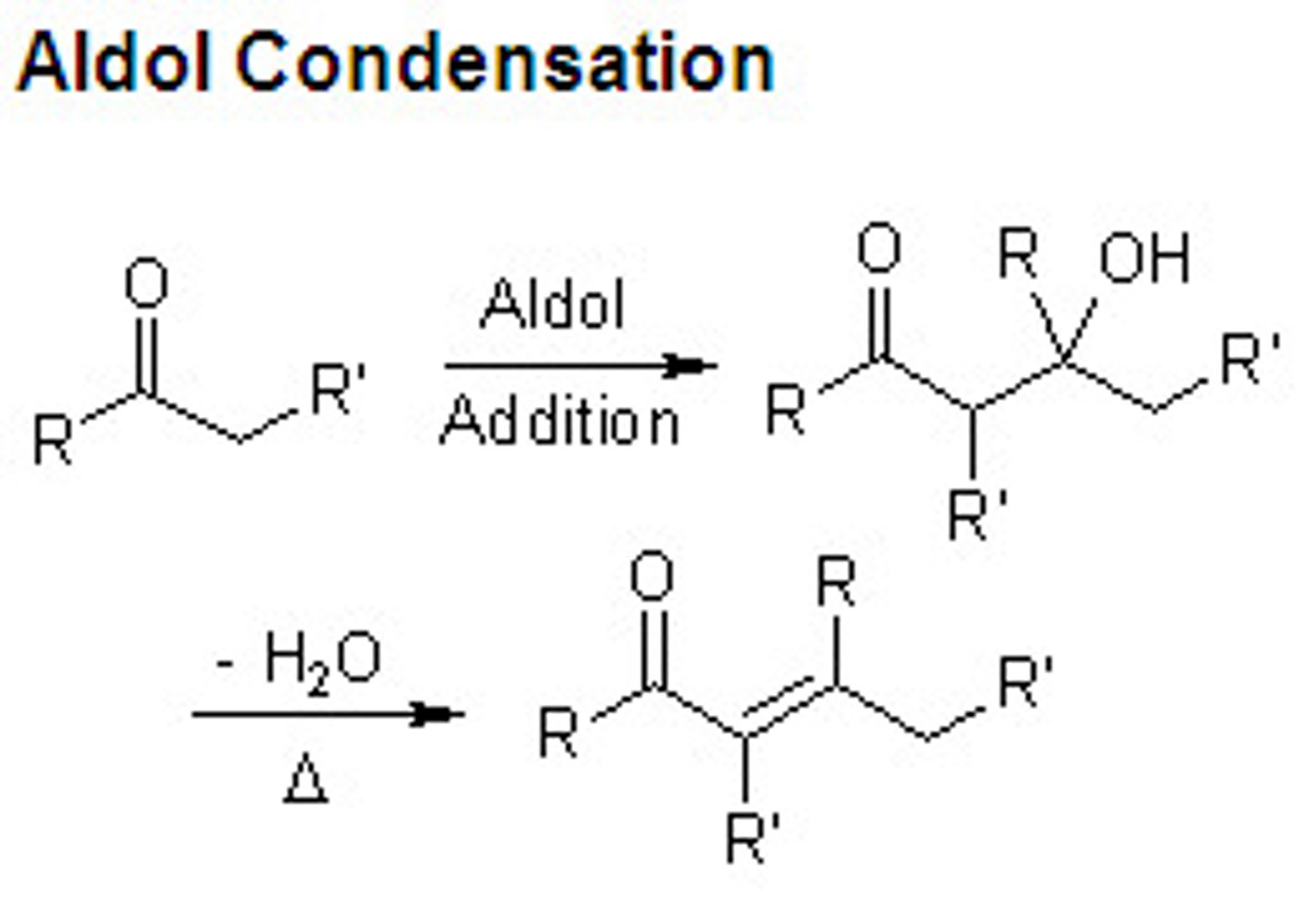
self aldol reaction
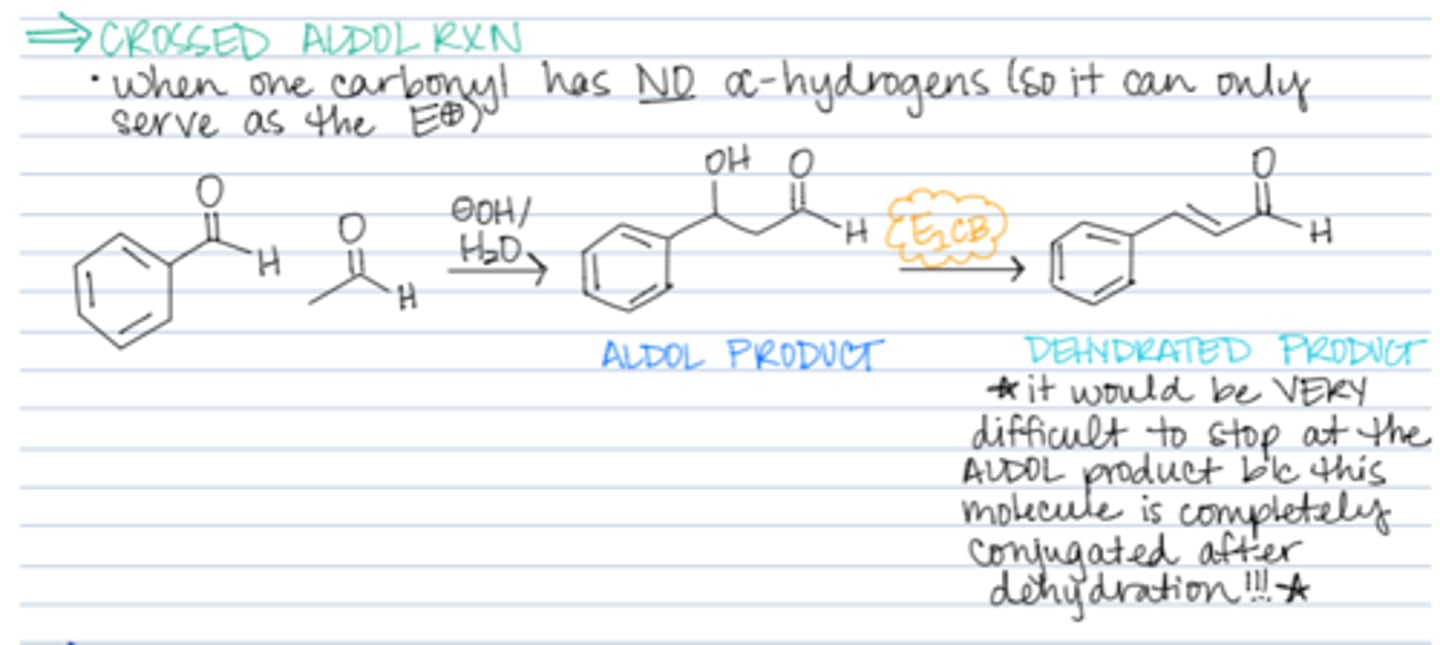
crossed aldol reaction
intramolecular aldol reaction
the ketone will usually form the enolate and attack the aldehyde
what bases and solvents are typically used for the aldol reaction
NaOEt/EtOH, or NaOH/H2O, or NaOMe/MeOH and sometimes LDA but it is selective towards the kinetic enolate
claisen schmidt aldol consendation
a ketone reacts with a carbonyl compound that lacks alpha protons and there is no aldol addition product intermediate
what is the points of an aldol reactions
to make an aldehyde and alkene
What prefers direct addition (1,2) (kinetic)
Grignards, LiAlH4, Wittig, organolithium and these attack directly at the carbonyl.
What prefers conjugate addition (1,4) (thermodynamic)
CN, Gilman (CuLi), enolates, amines, attack at the beta carbon
What does the gilman reagent do?
(CuLi) substitutes halides and leaves the carbonyls alone
what does an amide in h3O+ create?
a carboxylic acid and an amine
grignards reagent and organolithium (Ch3Li) does what?
multiple attacks and makes OH with H3O+ present
what is the difference between an aldol and a claisen reaction?
The aldol reaction starts with a ketone or aldehyde and a claisen ester.
What is the Micheal addition?
forms a product that contain alpha, beta, alpha trend