DNA
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
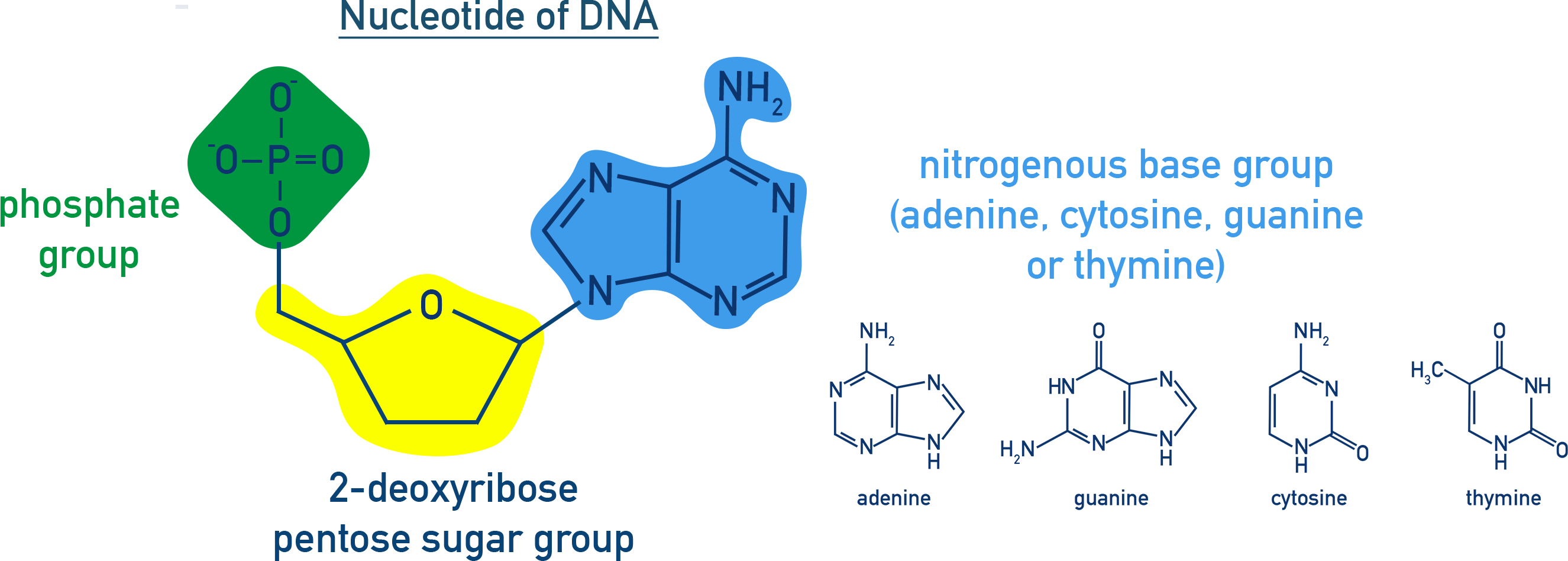
what is a nucleotide? draw the structure of a nucleotide":
monomer made up of PO43- bonded to 2-deoxyribose in turn bonded to 1 of 4 bases
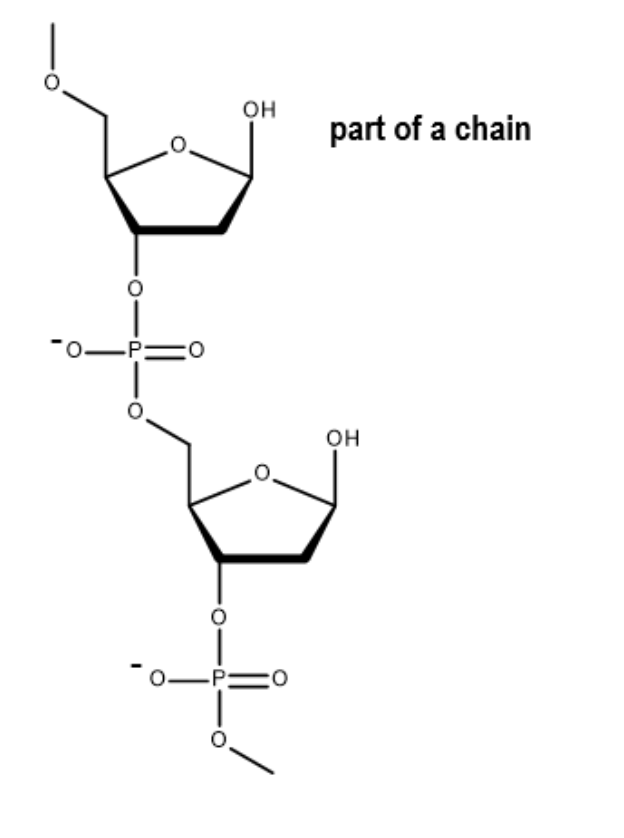
draw 2 nucleotides bonded together by covalent bonds (and trailing bonds):
describe the structure of DNA:
ssDNA = polymer of nucleotides linked by covalent bonds between the PO43- group of one nucleotide and the 2-deoxyribose of another nucleotide
2 complementary strands arranged in the form of a double helix
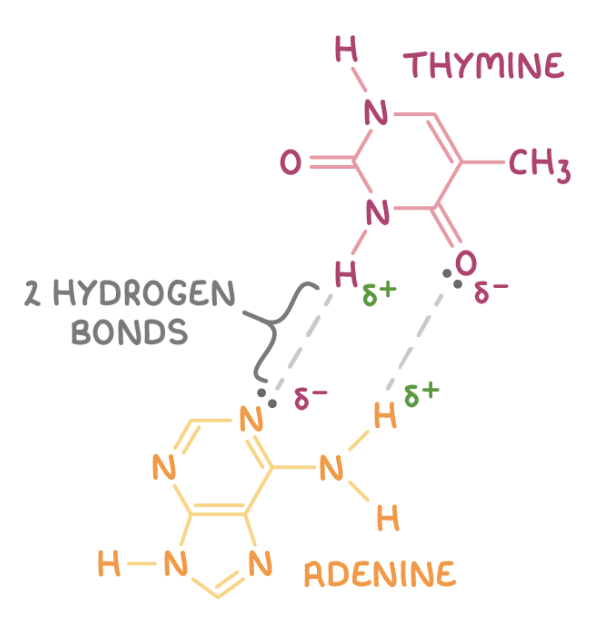
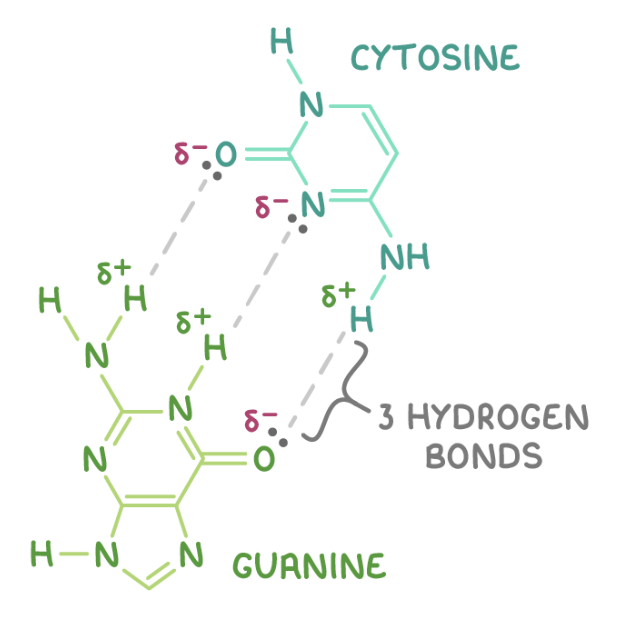
CBP between H bonds - 2 between adenine and thymine, 3 between cytosine and guanine
complementary strands have base sequences that match adenine to thymine and cytosine to guanine
sugar-phosphate-sugar-phosphate polymer chain w/ bases attached to the sugars in the chain
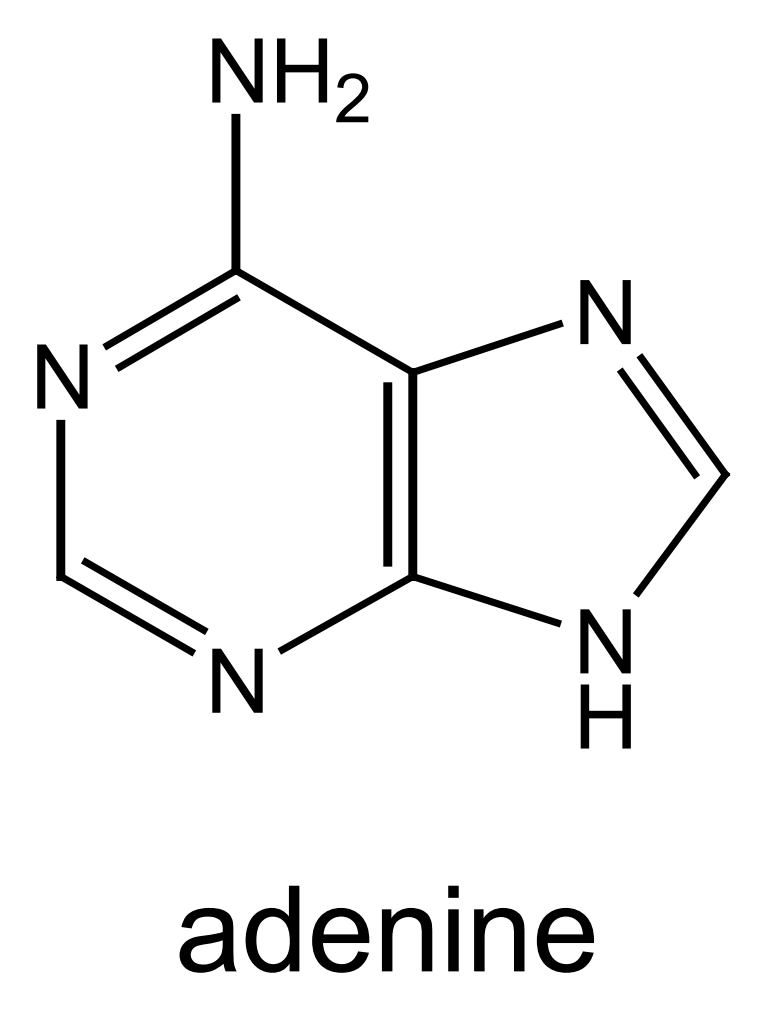
this is the structure of adenine - mark where on the molecule adenine bonds to deoxyribose and its base partner:
(when bonding to deoxyribose - H is lost as part of condensation reaction)
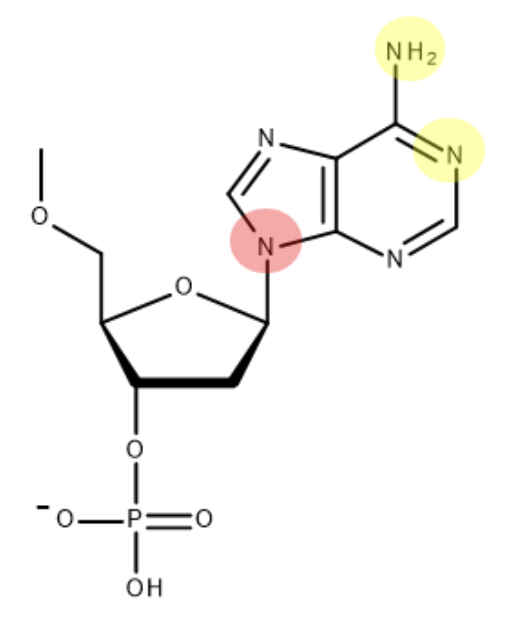
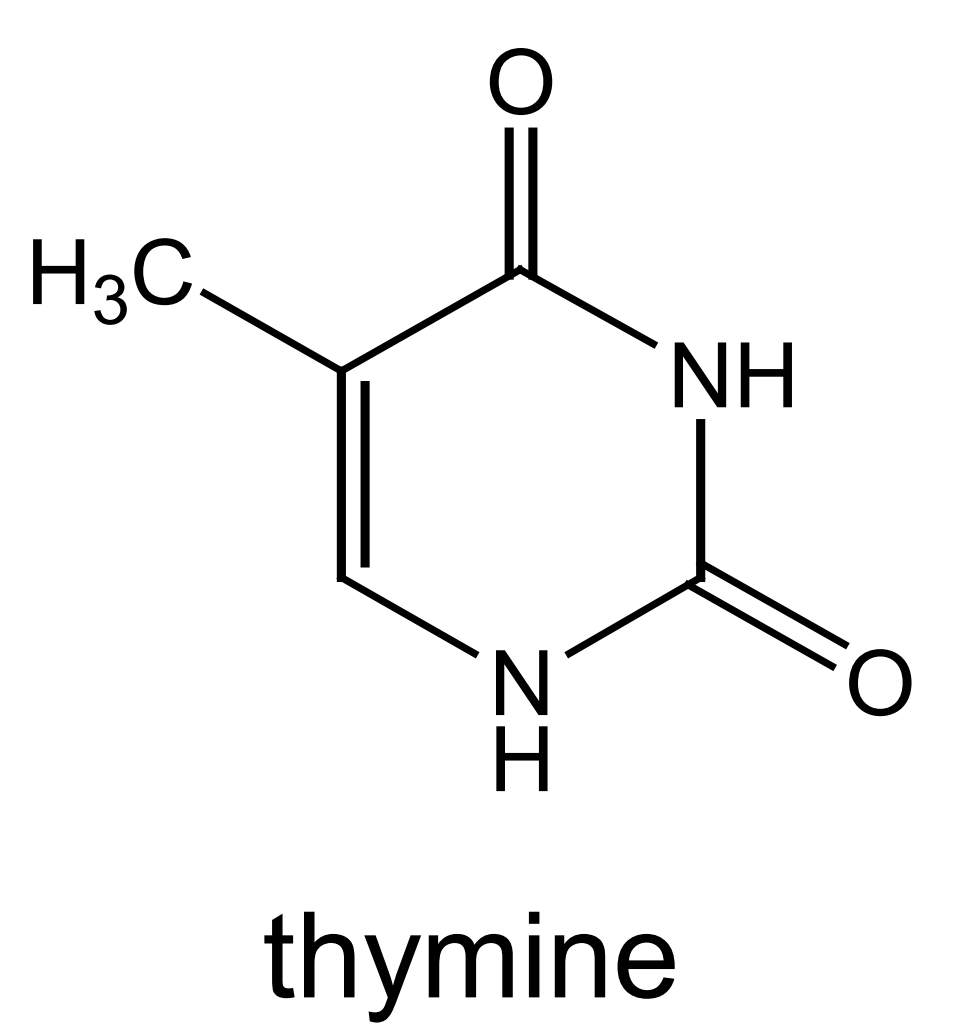
this is the structure of thymine - mark where on the molecule thymine bonds to deoxyribose and its base partner:
(when bonding to deoxyribose - H is lost as part of condensation reaction)
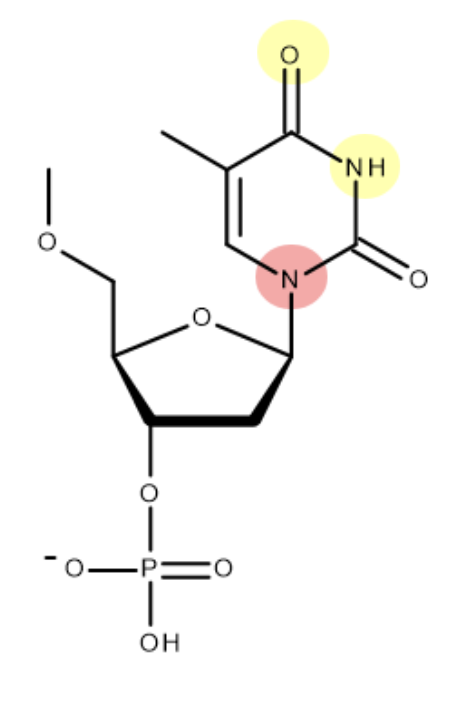
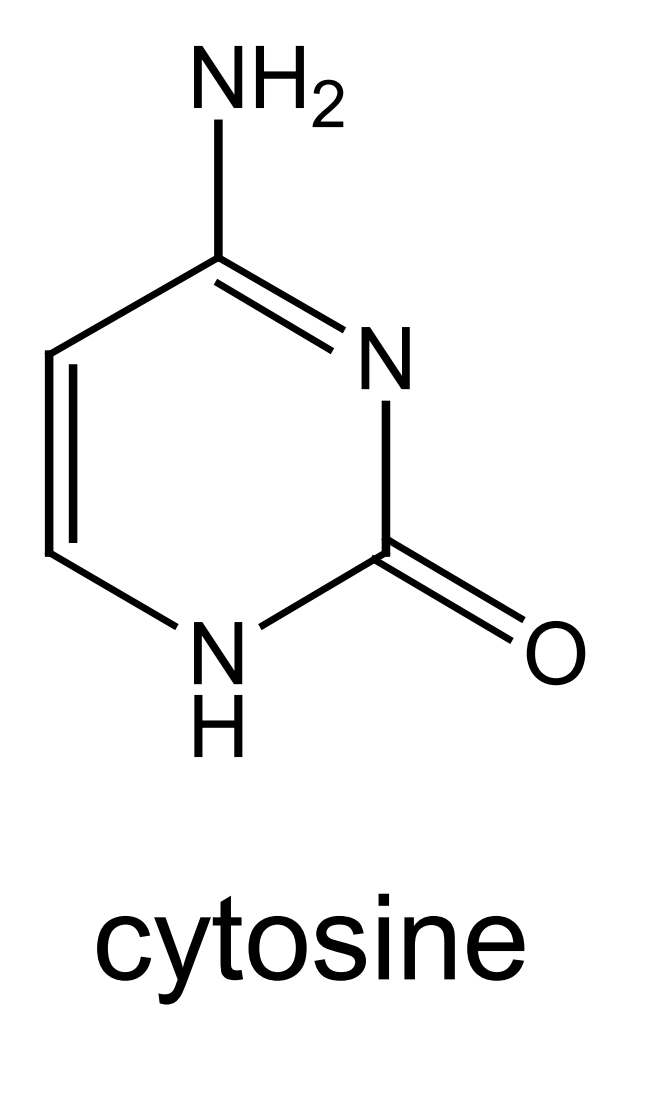
this is the structure of cytosine - mark where on the molecule cytosine bonds to deoxyribose and its base partner:
(when bonding to deoxyribose - H is lost as part of condensation reaction. only one of H atoms in NH2 forms H bond)
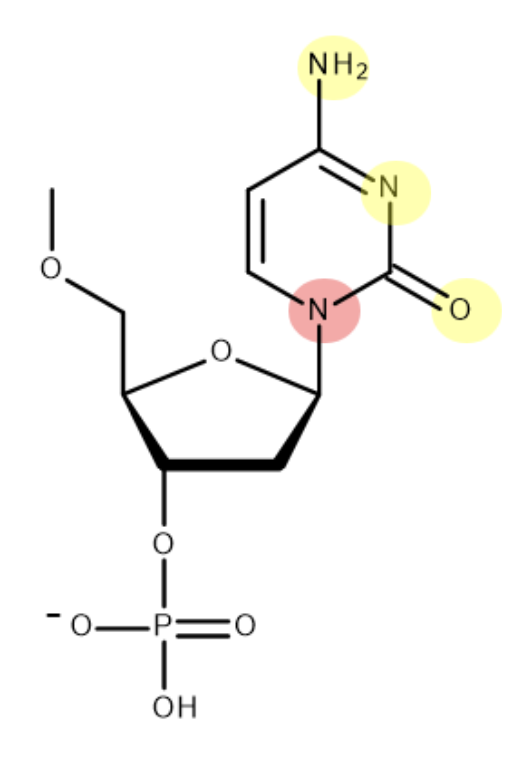
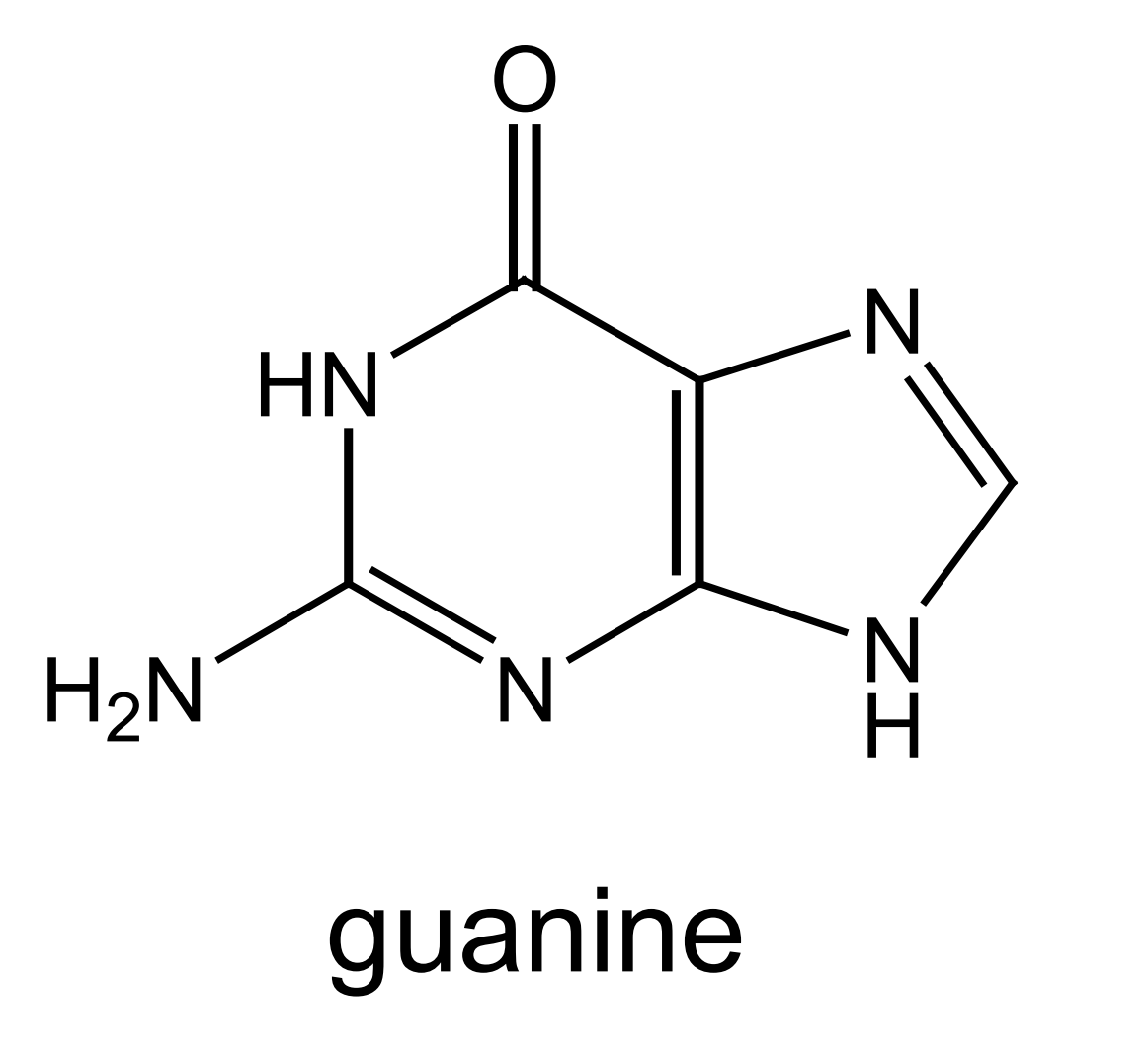
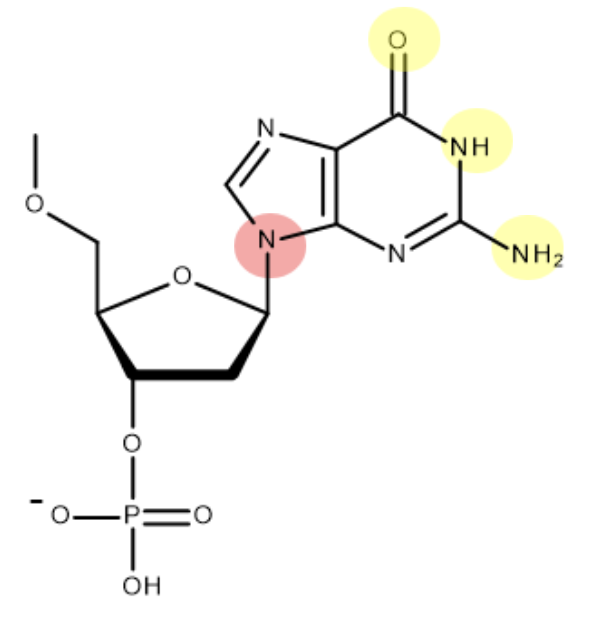
this is the structure of guanine - mark where on the molecule guanine bonds to deoxyribose and its base partner:
(when bonding to deoxyribose - H is lost as part of condensation reaction. only one of H atoms in NH2 forms H bond)
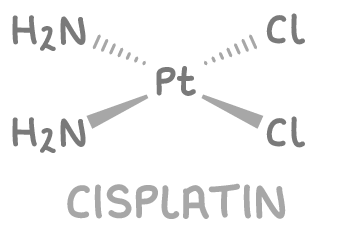
give the structure of cisplatin and explain why transplatin would be unsuitable as an anticancer drug:
not transplatin - Cl ligands must be on same side to substitute so that Pt can bind to guanine bases
describe how cisplatin is used as an anti-cancer drug:
the bonding of Pt to N atom in guanine displaces the Cl- ligands, resulting in crosslinking which causing kinks in the DNA structure
so altered DNA cannot unwind or replicate as it normally would
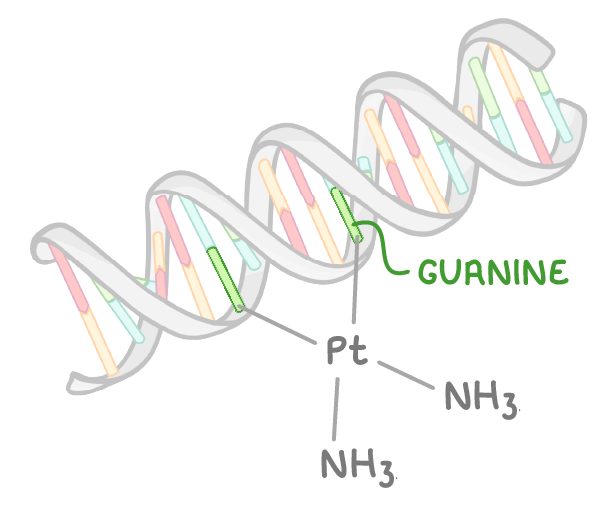
explain why drugs such as cisplatin can have adverse effects - suggest how these can be controlled and why cisplatin is still used for chemotherapy:
can bind to DNA in normal cells as well as cancer cells
can cause side effects especially in rapidly /ing cells e.g. hair loss due to damage to hair follicle cells
can be controlled by giving small doses/targeting cisplatin to the tumour
still used - balance of long-term +ve effects (curing cancer) outweigh -ve short term effects
draw how a cytosine unit H bonds to a guanine unit:
draw how a thymine unit H bonds to an adenine unit: