8.1 & 8.2 Review (Physical & Chemical Properties and Changes and Classifying Matter)
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
Physical Property
A characteristic of a pure substance that can be observed without changing it into another substance

Physical Change
A change in a substance that does not involve a change in the identity of the substance

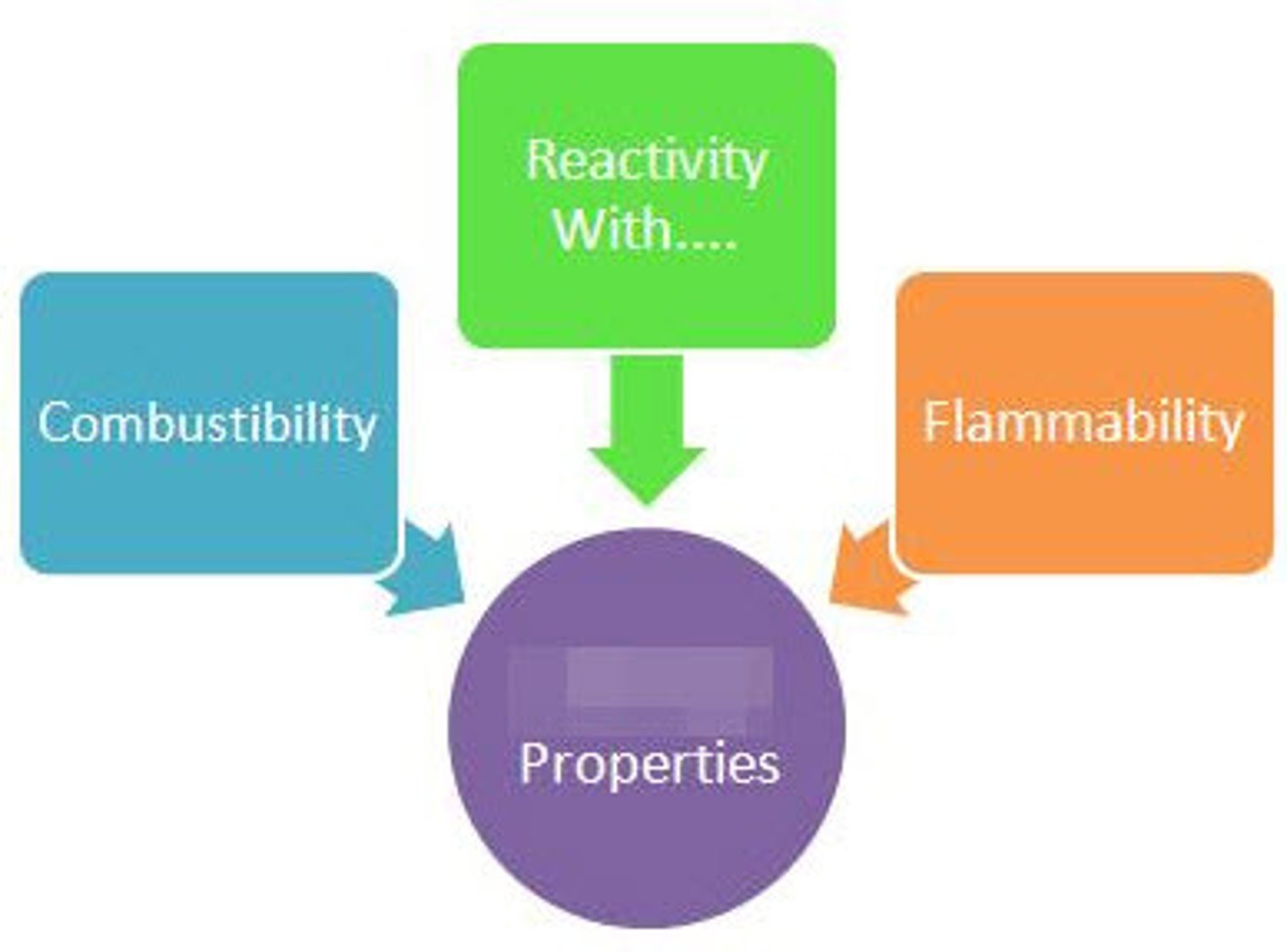
Chemical Property
a property of matter that describes a substance's ability to participate in chemical reactions
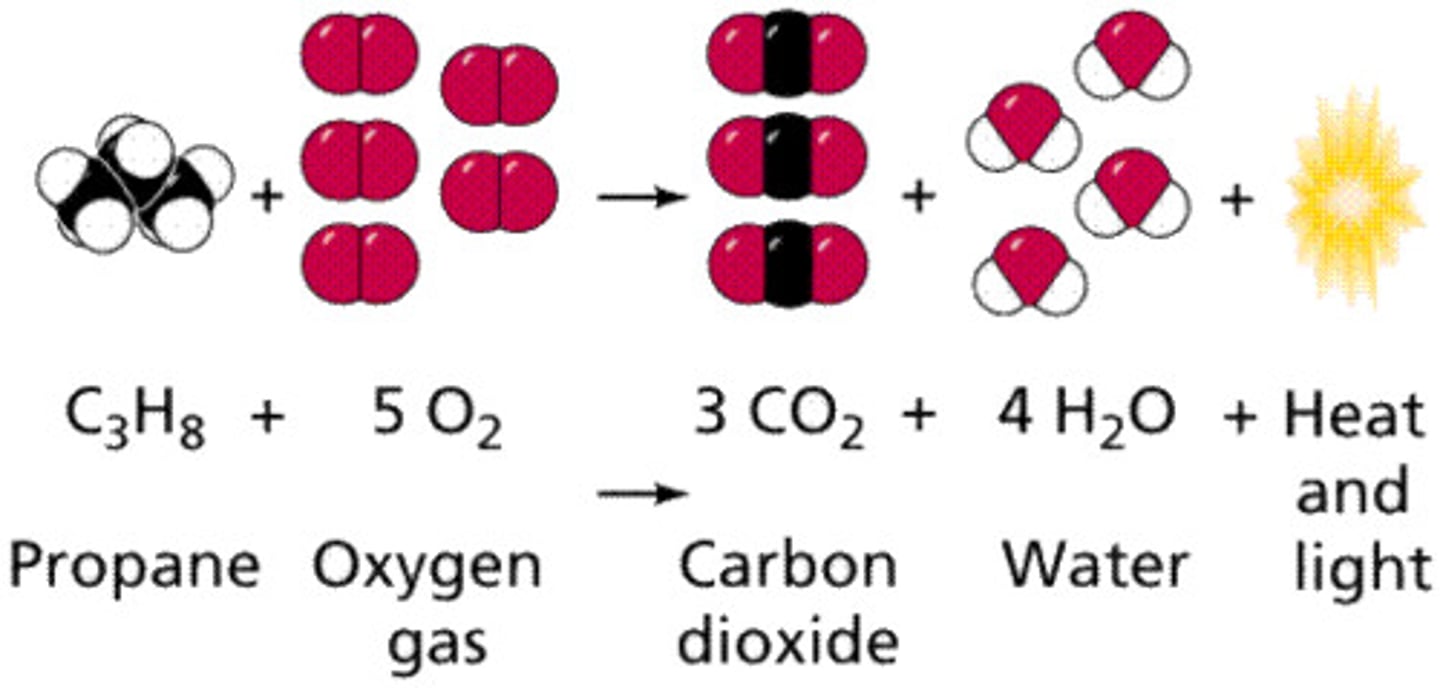
Chemical Change
a change that produces matter with a different composition than the original matter when bond break or new bond form.
Texture
The feel, appearance, or consistency of a surface, substance, or fabric. Physical Property
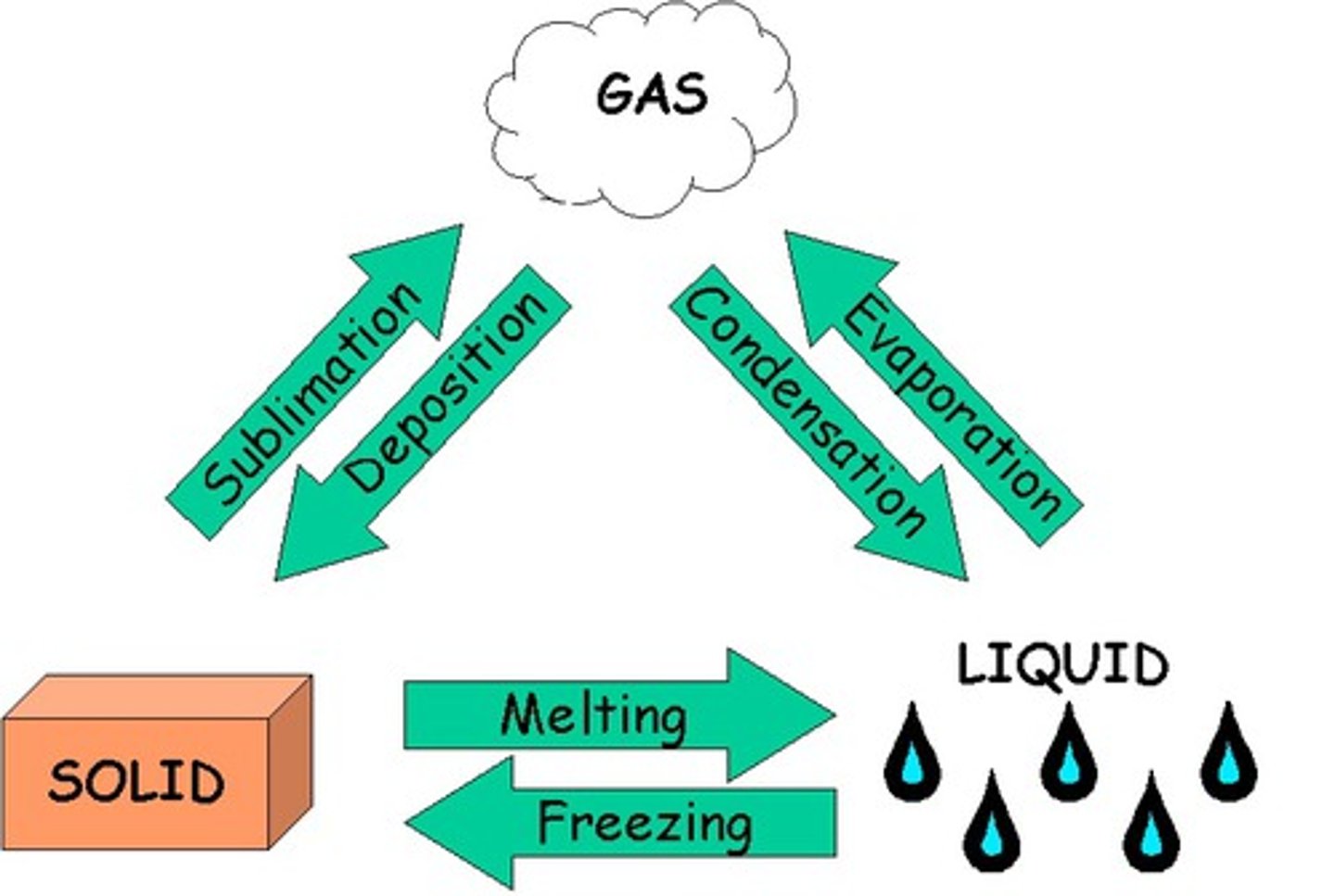
change of state
a physical change of a substance from one state to another by boiling, freezing, melting, condensation, vaporization. Physical Change
boiling point
The temperature at which a liquid changes to a gas. Physical Property

Reactivity
The ease and speed with which an element combines, or reacts, with other elements and compounds. Chemical Property

precipitate
A solid that forms from a solution during a chemical reaction.

evidence of a chemical change
gives off or production of a gas (bubbles), changes color (not always), temperature change, forms a precipitate
Flammability
A material's ability to burn in the presence of oxygen. Chemical Property

corrosiveness
The gradual breaking down a substance by chemical means like tarnishing or rusting metal; chemical property
rust
the formation of reddish-brown ferric oxides on iron by low-temperature oxidation in the presence of water (chemical process)

crumpling, breaking, tearing, chopping, crushing
physical change examples
burning, photosynthesis, souring, digesting, rotting, rusting
chemical change examples
What are the three main classifications of matter?
Element, compound, and mixture.
What are the two types of elements?
Monatomic and diatomic.
What type of mixture is a solution?
A homogeneous mixture.
What distinguishes a compound from a mixture?
Compounds are chemically bonded and cannot be separated by physical means, while mixtures are physically combined and can be separated.
How are diatomic elements classified?
They are classified as elements, although they consist of two atoms.
What is the difference between homogeneous and heterogeneous mixtures?
Homogeneous mixtures have a uniform composition, while heterogeneous mixtures have a non-uniform composition.
What is a molecule?
A group of two or more atoms bonded together.
Cobalt
Monatomic element
Chlorine
Diatomic Element
Silicon Dioxide
Compound
Air
Homogeneous Mixture
Oil & Water
Heterogenous Mixture
A Chemical Equation
Mixture