Exam 3: synapses & vesicles, receptors, neurotransmitters, auditory system, case study
1/127
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
128 Terms
Sensation
The detection of stimuli
Perception
The interpretation of stimuli
Auditory System What is sound?
Pressure waves moving through air or some other physical material. Pressure at a single point in space oscillates as the sound (acoustic) wave travels by
3 Components of sound waves
Frequency, amplitude, timbre
Frequency
How often do the sound waves compress? Highest notes humans can detect is around 20,000 Hz (painful shrill). Low frequency sounds are deep rubles, like from a bass. Humans can detect as low as 20Hz.
Amplitude
How much do the waves displace the medium from baseline? The larger the amplitude of the wave, the greater distance between the peak and trough of the signal, the louder the sound is. Loudness is measured in decibels (dB).
Timbre
How complex is the wave form? The color or character of sound. How sound waves interact.
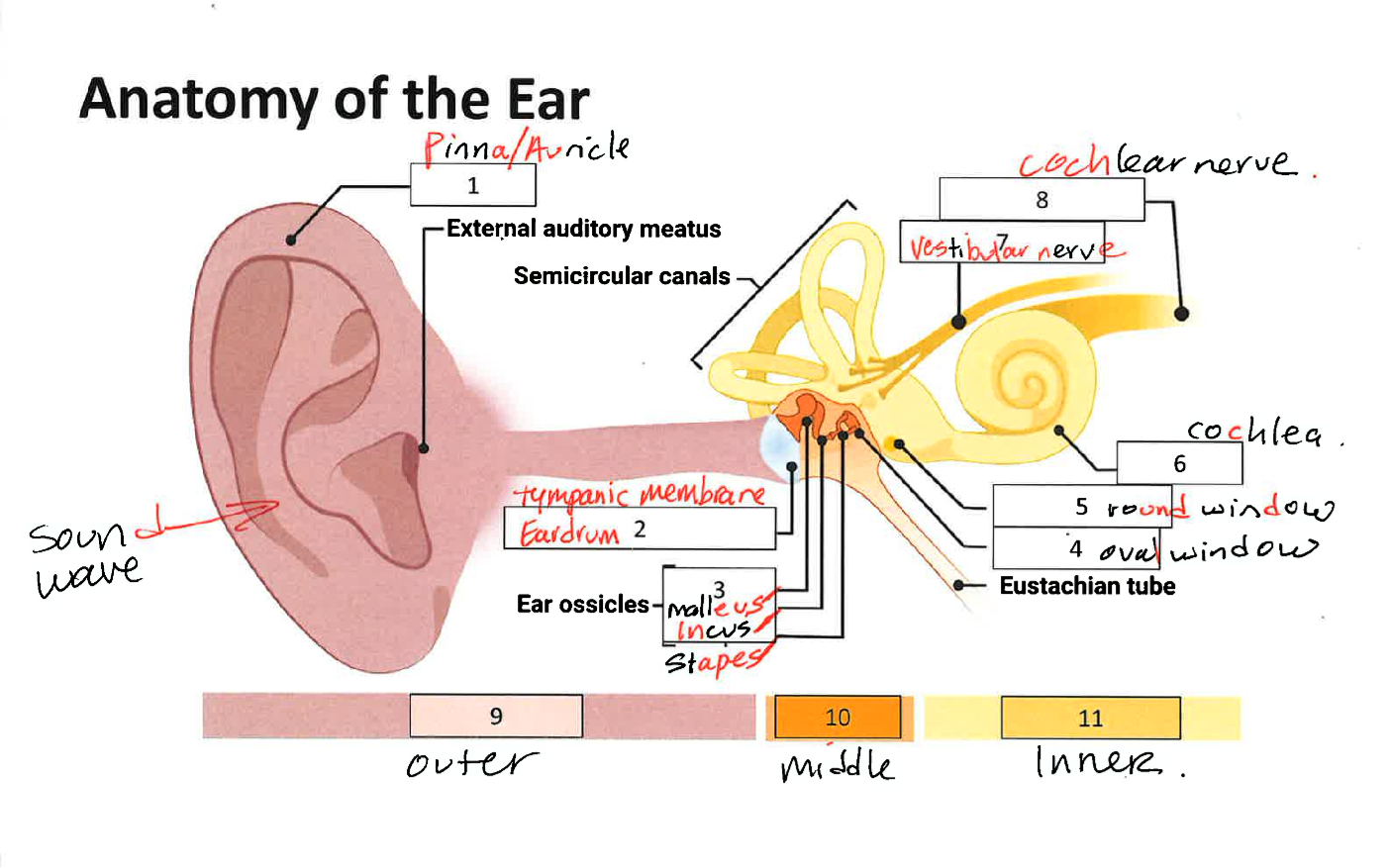
Anatomy of the Ear
Outer Ear, Middle Ear, Inner Ear
Outer Ear
Pinna/Auricle: Functions as a funnel to the auditory canal. Pinna helps us determine where a sound is coming from.
Middle Ear
Tympanic membrane/eardrum (0.1mm thick). When sound waves arrive at the tympanic membrane, it vibrates to match the frequency. Ossicles. Both tympanic membrane and ossicles help to amplify sound as well.
Ossicles
Three bones that convey tympanic membrane vibrational sound information to the oval window. Malleus, Incus, Stapes - Connects to oval window. In the middle ear.
Tensor Tympani
Muscle which connects with the malleus
Stapedius
Muscle which connects to the stapes
Acoustic Reflex
When tensor tympani and stapedius contract the ossicles move less which decreases the intensity of loud sounds. This response dampens incoming sound by about 15 dB.
Inner Ear
Semicircular canals. Cochlea (greek for snail shell). Inside cochlea is the Organ of Corti. Vestibular Nerve, Cochlear nerve
Cochlea
(Greek for snail shell) Changes in shape from the base to apex.
Organ of Corti Defined
Inside the cochlea it is responsible for processing physical vibrations and converting them into signals the nervous system can interpret.
The Organ of Corti
Contains components for converting sound waves into action potentials. Next to the Organ of Corti is a liquid called endolymph. It is a high K+ low Na+ solution similar to CSF. Embedded along the interior surface of the Organ of Corti are hair cells the primary sensory neurons that interpret physical movement. Each hair cell contains 30-100s of stereocilia. Movement of inner hair cells detection of sound create neural impulses. Movement of outer hair cells - amplifier to increase the intensity of vibrations (3 rows)
How does acoustic information enter the brain? 1
At rest, ion channels in the cilia are closed. Vesicles are docked at the ribbon synapse.
How does acoustic information enter the brain? 2
When the cilia are displaced tip links pull open the mechanically-gated ion channels. Tip links of sensory heair cells in the inner ear play a critical role in mechanotransduction.
Mechanotransduction defined
The conversion of mechanical stimuli arising from sound and head movements into electrochemical signals.
How does acoustic information enter the brain? 3
The endolymph has a high concentration of K+ and Ca2+ so those ions flow into the cilia when the ion channels open.
How does acoustic information enter the brain? 4
The hair cell depolarizes, opening voltage gated Ca2+ channels. Ca2+ influx causes vesicles to fuse and release neurotransmitter.
Mechanotransduction
Stereocilia on the hair cells vary in length. Shorter hair cells are more responsive to high pitch sounds (closer to the base of the cochlea). Longer hair cells are more responsive to lower pitched sounds (closer to the apex, near the center of the cochlea). Therefore the position of the hair cells along the spiral of the cochlea AND the length of the stereocilia are two tuning mechanisms that make inner hair cells sensitive to specific frequencies. Hair cells form glutamatergic synapses onto bipolar neurons of the spiral ganglion. The axons of these neurons make up the vestibulocochlear nerve (cranial nerve 8).
Pathway leading to processing to the brain Step 1
Some of the nerves from CN 8 synapse an area called the superior olive; some synapse on the cochlear nuclear complex. Superior olive helps us to determine if sound came from the left or right side of our heads.
Pathway leading to processing to the brain Step 2
Auditory information is then passed to the inferior colliculus. Inferior colliculus is important for interaction between multiple sensory inputs and motor response. Particularly responsive to unexpected sounds like a predator
Pathway leading to processing to the brain Step 3
The inferior colliculus passes information to the medial geniculate part of the thalamus.
Pathway leading to processing to the brain Step 4
The thalamus lastly send information ot the primary auditory cortex, A1. Primary auditory cortex also called Herschel’s gyrus. A1 is a the most dorsal portion of the temporal lobe. Once sound information is processed by A1 it then passes through the secondary and tertiary auditory cortices. Earliest studied cortical area in auditory perception, Wernicke’s area.
Wernicke’s area
Speech and language comprehension
Dual Stream cortical processing of auditory information after A!
Dorsal (red) - location of sounds; speech production; and language-related memory. Ventral (green) - sound recognition and sentence comprehension.
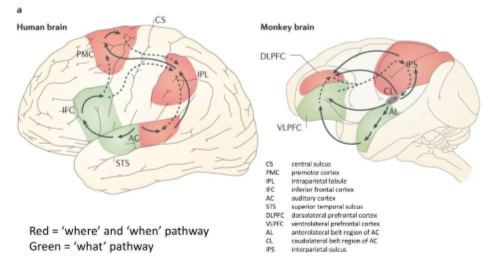
Tonotopically Organized
Organ of Corti, the spiral ganglion, and A1 are tonotopically organized. Adjacent physical areas are responsible for conveying information from adjacent frequencies. hair cells that respond most to 440Hz are right next to cells that respond to 441Hz but far from cells that respond to 14,000Hz.
Clinical Application: Hearing loss Define and types
Hearing loss is defined as a decrease in 25 dB or more. Two categories of hearing loss are conductive hearing loss and sensorineural hearing loss.
Conductive Hearing loss
A result of changes to the auditory system up to the oval window. Example: a tumor in the ear canal, a perforation of the tympanic membrane/eardrum, or changes in middle ear pressure (such as how everything sounds muffled while changing altitudes when an airplane takes off). Also damage or defect of the ossicles.
Sensorineural Hearing loss
Results from changes at the level of the inner ear or further up in the neural pathway. Examples: hair cell damage, a brain tumor, bacterial or viral infections, or exposure to various toxins or drugs, damage to the auditory nerve or cortices.
Causes of hearing loss
Excessive noise exposure. Examples: motorcycles, max volume on headphones or loud concerts/club sound in the 95-110 dB range. Aging - born with about 15,000 hair cells but throughout our life many get damaged. Shorter cells are sensitive to injury so it is common for people to lose sensitivity to high-frequency sounds.
Treating Hearing loss
Hearing aid: a processor that helps to filter out background noise, decrease pitch, and amplify incoming sounds. Cochlear implant: surgically implanted device that receives incoming sound information and directly stimulates the auditory nerve via electrodes. This bypasses the external components of the auditory system. More common in children when diagnosed with bilateral profound sensorineural hearing loss.
Cochlear Implant
Surgically placed within the inner ear, inside the cochlea and essentially takes the place of delicate hair cells. The implant then sends electrical signals directly to the auditory nerve. Two part device the external portion called the processor picks up acoustic sound. The sound processor is held in place over the implant with a magnet. The sound is then processed and sent via radio frequency signal to the internal portion. After receiving the signal the internal portion sends it to the electrodes placed in the cochlea. The electrodes send small pulses to the auditory nerve which then sends the signal to the brain.
Clinical Application: Tinnitus
Condition characterized by the occasional perception of ringing, whistling, buzzing, or clicking sound in the absence of an actual stimulus. Cause: mainly unknown. Main predictor-hearing loss the perception of phantom sound originates in the brain as a result of faulty plasticity the “understimulated” brain produces abnormal signals.
Anatomy of the Ear
What type of synapse allows neurons to share their cytoplasm to exchange things such as ions?
Electrical
The fusion of a synaptic vesicle at the axonal terminal membrane requires.
The opening of voltage-dependent calcium channels.
What proteins make up gap junctions?
Connexins
True or False: All vesicles look the same.
False
What type of synapse has a neurotransmitter released by one neuron, with the neurotransmitter diffusing across the synapse, to bind to a receptor on the next neuron?
Chemical
What protein helps to package vesicles with neurotransmitter?
Vesicular transporters
What type of receptor has a very specific structure that spans the membrane 7 times?
G-protein coupled receptors
Which type of receptor produces a rapid response on the postsynaptic side?
Ligand-gated ion channel receptors
Which type of receptor produces a more modulatory response by activating a series of intracellular events?
G-protein coupled receptors
Which subunit of the GPCRs signals the type of signaling cascade that the receptor will produce?
Alpha
What SNARE protein detects a rise in calcium in the axon terminal?
Synaptotagmin
What is curare? What is the active ingredient in curare?
Toxin, tubocurarine is the active ingredient
Where does d-tubocurarine act at the biochemical level?
Inhibitor of the nAChR
Why did Chucky fall to the ground and show an inability to talk or move the side of his body? What will happen if Chucky does not receive treatment quickly?
Because nAChRs help muscles move; he needs help quickly so that his diaphragm does not stop moving; otherwise he will stop breathing.
Spend some time in your group discussing what characteristics a drug treatment would need to have to reverse the symptoms associated with poisoning by curare. Write down two of your best ideas to share with the class.
Removing it from the receptor, something to break it down. Maybe dialysis or blood transfusion to get out the curare. Give him oxygen to help him breath.
What is the biochemical target of physostigmine and sarin gas?
AChE (helps break down ACh) (acetylcholine esterase)
What do these chemicals do to the target and how would this affect signaling at the NMJ?
By blocking AChE, there will be more ACh available in the synapse to activate nAChRs. If we increase ACh enough it will hopefully compete for the binding site on the nAChR and knock curare off the receptor.
Why should sarin never be used as a therapeutic to counteract the effects of curare poisoning even though it has the same target as physostigmine?
(Sarin gas is highly toxic and has been used in warfare). The difference between physostigmine and sarin-physostigmine is reversible, while sarin binds irreversibly to the nAChE.
What is Botox?
Poison containing botulinum toxin.
Where does botulinum toxin type-A act at the biochemical level?
Breaks down the SNARE proteins. If the SNARE proteins don’t work correctly ACh vesicles cannot bind and release ACh.
Is Chucky correct about Botox being poisonous? Explain
yes if eaten it is poisonous but can be used to help reduce migraines.
Is Melody also correct about the duration of effects of Botox? Explain
Botox doesn’t last forever because your body breaks it down
Define Neurotransmitters
The substance that are released at chemical synapses: the signaling molecules that allow neurons to communicate with one another. Scientists have identified over 100 neurotransmitters.
1-Glutamate (Glu)
Main excitatory neurotransmitter. Same as the amino acid glutamic acid (E). Role in learning. Glutamatergic neurons are identified by the presence of vGluT - the vesicular glutamate transporter. Glutamate has both ionotropic receptors and metabotropic receptors.
Excitotoxicity
Excess signaling by glutamate can cause neuronal death, called excitotoxicity. This is especially prevalent with NMDA receptors since they also allow the passage of calcium too (that is why calcium is stored in the ER in most cells). Excitotoxicity has been seen in disorders such as Parkinson’s, Alzheimer’s, multiple sclerosis, concussion, and stroke. (1-Glutamate)
Ionotropic Glutamate Receptors (3 classes)
1 & 2) AMPA and Kainate receptors: Na+ flows into the cell and depolarize it. 3) NMDA receptors: Na+ and Ca2+ flow into the cell and depolarize it
Metabotropic Glutamate Receptors (mGluRs)
8 types 3 groups. Some can activate ion channels. All have second messengers and signaling cascades, depending on the alpha subunit. Group 1: excitatory (Galphaq). Group 2 & 3: inhibitory (Galphai)
GABA
Gamma-aminobutyric acid (GABA) is the main inhibitory neurotransmitter in the brain. About 25% of neurons are GABAergic. Synthesized from glutamate by the enzyme glutamic acid decarboxylase (GAD). GAD also used as a marker to label GABA neurons. Many interneurons use GABA as their signaling molecule. Interneurons are neurons that stay within the brain region they are found and help modulate glutamate activity.
Glycine
Also inhibitory but mainly found in the spinal cord and brainstem
GABA Receptors - Ionotropic & Metabotropic
Three classes of GABA receptors A,B, and C. GABA a receptors ligand gated ion channel receptors for Cl-. GABA b and GABA c are metabotropic - Gi coupled; also interact with K+ channels to further hyperpolarize the cell.
3- Dopamine (DA)
While glutamate and GABA are synthesized all over the brain, there are certain regions/nuclei that produce dopamine. VTA and SN. Considered biogenic amine or catecholamine. One of the enzymes important to the synthesis of DA is tyrosine hydroxylase or TH. TH is a marker for DAergic neurons. DA has many roles: reward, motivation, motor control, attention, learning
DA receptors
Only GPCRs. D1 and D2 like receptor
Clinical Application - Loss of DA - Parkinson’s disease (PD)
PD is a neurodegenerative disease that affects about 1% of people 60 and older. By the time motor symptoms appear individuals have already lost 60-80% of DA neurons in the substantia nigra. One way to treat PD is to use L-DOPA. However, beneficial effects do not last forever. Dyskinesias can occur. Another treatment is deep brain stimulation of the subthalamic nucleus.
4 - Serotonin (5HT or 5-HT)
Derived from the amino acid tryptophan (Trp; W). Produced by the raphe nuclei. Latest research shows that the bulk of the 5HT in our bodies is in neurons in the gut. 90% is in neurons in the gut. 8% is in blood (platelets, mast cells). 2% is in the pineal gland. 0.5% in the raphe nuclei in the brain stem. Project widely in brain, control blood flow, activity slows during sleep.
Serotonin Receptors and Functions
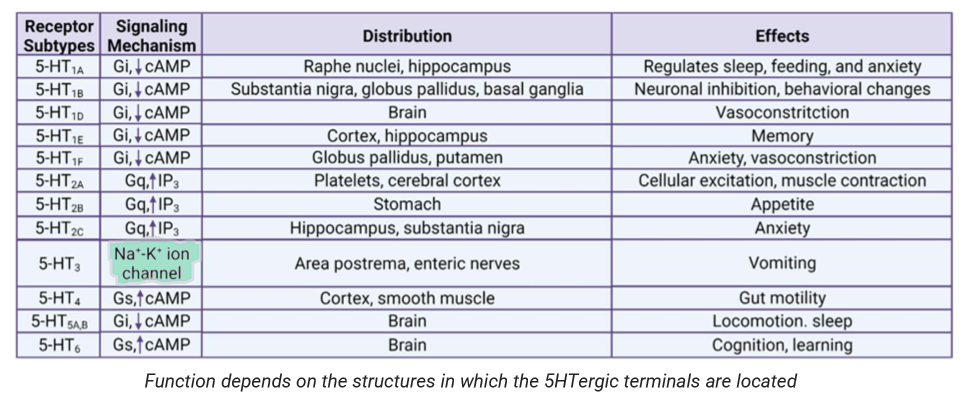
Clinical Application - Mood & anxiety disorders
SSRIs - Selective Serotonin Reuptake inhibitors. First one on the market - prozac (fluoxetine). Major depressive disorder, generalized anxiety disorder, PTSD, OCD.
5 - Acetylcholine (ACh)
First neurotransmitter discovered by Otto Loewi (Nobel Prize in Physiology or medicine with Henry Dale in 1936).
Acetylcholine (ACh)
Functions include: learning and memory, attention, emotions, movement. Sources of ACh: basal forebrain, PPN. ACh in degraded by the enzyme acetylcholinesterase (AChE)
Cholinergic Receptors - ionotropic
Nicotinic ACh receptors: ~10% of ACh receptors. In neuromuscular junction (NMJ). Allow in positive ions, depolarizing. Named due to the fact they bind nicotine as well as ACh.
Cholinergic Receptors - Metabotropic
Muscarinic ACh receptors. ~90% of ACh receptors, G-Protein linked receptors that activate other ionic channels via a second messenger cascade. Depolarizing or hyperpolarizing can do both.
Clinical Application - ACh in Alzheimer’s disease
Neurodegenerative brain disorder. Loss of ACh inputs to hippocampus, memory problems. Loss of ACh inputs to cortex, attention problems. Treatment is acetylcholinesterase inhibitors (AChE), only minimally effective.
6 - Norepinephrine (NE)
Neurotransmitter synthesized from DA. Source: Locus coeruleus (LC) (goes to kidney. Functions: arousal (attention), wakefulness, learning, stress (relapse)
Define Neuropeptides
Class of large signaling molecules that some neurons synthesize.
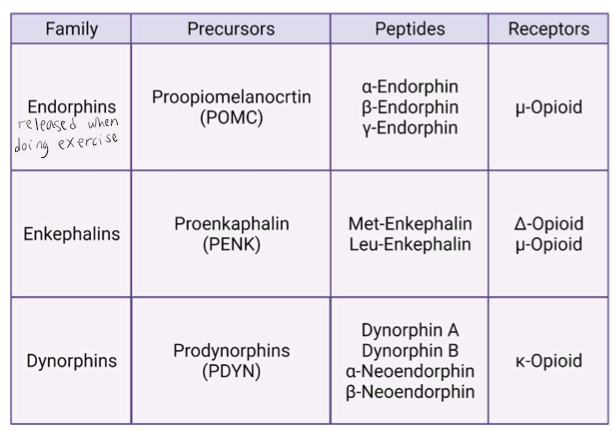
Neuropeptides: Endorphins
Endogenous opioids (3 families): modulate pain, neuroendocrine regulation, neuroimmune regulation, pleasure or euphoria. Expression of receptors may areas, but especially the periaqueductal gray area pain.
Endocannabinoids (eCBs) - Retrograde Transmission
Lipid-based neurotransmitter. Active ingredient in cannabis (Δ9-THC) acts on cannabinoid receptors throughout the brain and body. Endogenous ligand for these receptors are endocannabinoids. Endocannabinoids are retrograde neurotransmitters - they are synthesized in the postsynaptic neuron. Retrograde neurotransmitters are not packaged in vesicles. They are released by the postsynaptic cell and signal on receptors on the presynaptic cell.
Nitric Oxide (NO)
Gasotransmitter. Synthesized as needed, not stored in vesicles. NO is formed when the amino acid arginine is degraded by the enzyme NO synthase (NOS). Receptors for NO are intracellular - soluble guanylate cyclase (sGC)
What are receptors?
Proteins that are capable of sending a signal to change the function or activity of a neuron. They are large transmembrane proteins. Ligands (aka signaling molecule, aka neurotransmitter, or drug) bind to the part of the receptor on the extracellular side. Receptors are specific for their ligand or neurotransmitter
Types of postsynaptic receptors
Ligand-gated ion channel (ionotropic). G-protein coupled receptors (metabotropic)
Ionotropic Receptors
Also known as ligand-gated ion channel receptors (that we have seen before when discussing ion channels). In the diagram the neurotransmitter stays on the extracellular side, ONLY ions pass through. Induce a change in membrane potential (EPSP or IPSP)
Metabotropic Receptors-GPCRs
Also known as GPCRs - G-protein coupled receptors. They act SLOWER than ligand-gated ion channel receptors. They produce a modulatory signal. Instead of allowing ions though they begin a signaling cascade inside the neuron. They are also transmembrane proteins with a very specific structure - they span the membrane 7 times. On the intracellular side, the receptor is linked to a G-protein. There are different types of G-protein alpha subunits
GPCRs
GPCRs need energy to move around the different molecules. Instead of ATP it uses GTP. When the neurotransmitter binds, GDP is exchanged for GTP. Then the alpha subunits will detach with GTP attached and will start a signaling cascade. The beta and gamma subunits usually stick together and either stay attached to the membrane or activate a different signaling cascade.
Metabotropic Receptors - GPCRs - Alpha Subunits
Galphas: s is for stimulatory. These receptors increase activity of an enzyme called adenylyl cyclase, cyclic AMP, and PKA. Galphai: i is for inhibitory. These receptors decrease activity of adenylyl cyclase, cAMP, and PKA. Galphaq: generally excitatory activates different enzymes compared to Galphas
Autoreceptors
On occasion a neurotransmitter may have receptors on the presynaptic side (on the membrane of the axon terminal). These receptors are often inhibitory and serve a self-regulatory function. They can often decrease or increase neurotransmitter release from that axon terminal
Electrical Synapses
Cells share their cytoplasm and there is an exchange of ions and other small molecules (ATP). Related to the reticular theory that was proposed by Golgi. Channel connects the two neurons - made of proteins called connexis.
Electrical Synapses or Gap Junctions
Can pass information bidirectionally. Sometimes referred to as gap junction-mediated connections. The most thoroughly studied electrical synapses occur between excitatory projection neurons of the inferior olivary nucleus and between inhibitory interneurons of the neocortex, hippocampus, and thalamus.
Clinical Application: Charcot- Marie- Tooth (CMT) disease
Rare genetic disorder that damages part of the PNS. Results in muscle weakness, difficulty walking, experience abnormal sensations (tingling and pain in extremities). A connexin protein - Cx32, is heavily expressed in Schwann cells. Mutations in Cx32 are associated with the X-linked form of CMT.
Chemical Synapses
Signaling molecule (neurotransmitter) is released from the presynaptic neuron, diffuses across the synapse, and binds to its corresponding receptors. Chemical synapse about 20-40nm wide. Depending on the neurotransmitter released and the corresponding receptors, a variety of responses can be produced. A specific synapse between neurons and muscles are called the NMJ or neuromuscular junction
Small vesicles (often clear)
About 40nm wide. Although small, 1000-10’s of thousands of molecules of NT stored in each one. Found exclusively in axon terminals. Classical NTs like glutamate, GABA, dopamine, norepinephrine.
Large Dense Core Vesicles
About 100-250nm wide. Store peptide NTs like dynorphin or enkephalin. Packaged near the nucleus so can be found throughout neuron.
How are vesicles packaged with NT?
Transmembrane proteins called vesicular transporters help fill the vesicles with neurotransmitters. Tranporter is specific for the neurotransmitter it lets in. vAChT=vesicular acetylcholine transporter. The transport of NT into the vesicle is aided by vesicular ATPases and antiporters.
Where are vesicles found in the axon terminal?
Readily releasable pool (RRP), Recycling pool, Reserve pool