Biochemistry - Chapter 15: Metabolism: Basic Concepts and Themes
1/86
Earn XP
Description and Tags
These flashcards cover essential vocabulary and concepts related to metabolism from the lecture notes.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
87 Terms
Metabolism
A highly integrated network of chemical reactions that carry out energy extraction and synthesis of new material.
composed of many interconnected reactions
Catabolism
A set of metabolic pathways that break down complex molecules into simpler ones to capture energy.
Anabolism
A set of metabolic pathways that construct larger molecules from smaller units, consuming energy.
ATP
A nucleotide consisting of adenine, a ribose, and a triphosphate unit, which acts as the primary energy carrier in cells.
is the universal currency of free energy in biological systems
Free energy derived from oxidation of food and from light is transformed into __
Active in complex with Mg2+ or Mn2+
Phototrophs
Organisms that capture energy from sunlight, such as all photosynthetic organisms.
Chemotrophs
Organisms that capture energy through the oxidation of chemicals, such as all animals.
Glycolysis
A 10-step metabolic pathway converting glucose to pyruvate.
Phosphoryl-transfer potential
The tendency of an organic molecule to transfer its terminal phosphoryl group to an acceptor molecule.
Oxidative phosphorylation
A process that uses the energy of a proton gradient to produce ATP.
Activated carriers
Small molecules that carry a functional group or electrons and can donate them to another molecule.
NAD+
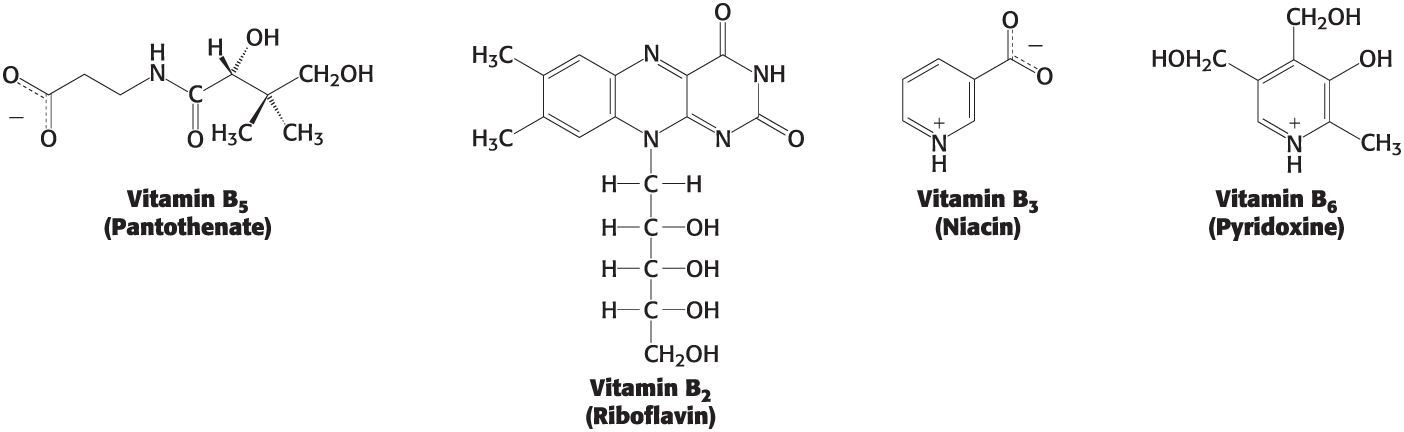
Nicotinamide adenine dinucleotide, an activated carrier that accepts electrons and protons in redox reactions.
FAD
Flavin adenine dinucleotide, an activated carrier that accepts electrons and protons during substrate oxidation.
Coenzyme A
A carrier of acyl groups, derived from vitamin B5, that plays a key role in the transfer of acyl groups and synthesis.
Energy charge
A measure of the energy state of a cell; it reflects the ratio of ATP to ADP and AMP.
Feedback inhibition
A regulatory mechanism in metabolism where the end product of a pathway inhibits an earlier step.
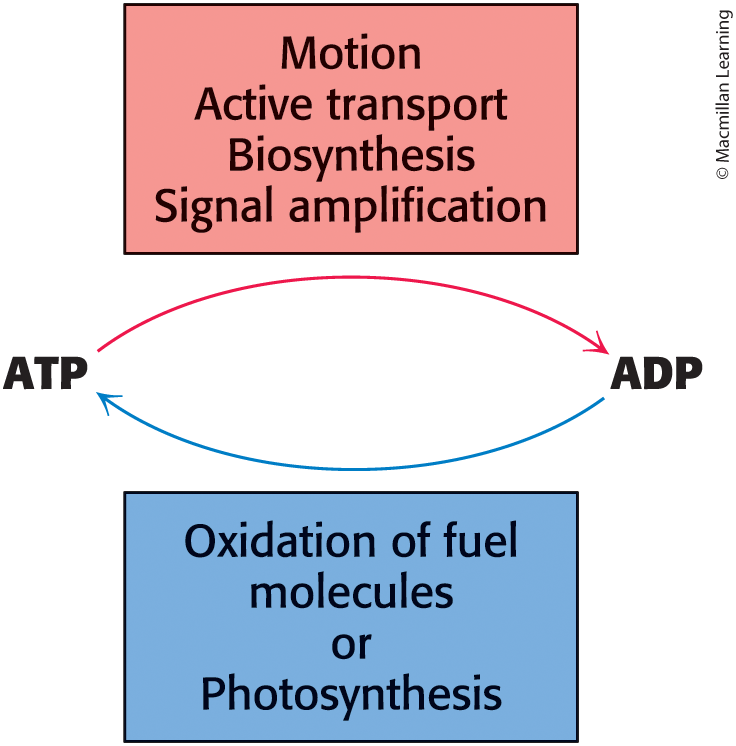
Energy
Is required for mechanical work muscle contraction and cell movement, active transport, and biosynthesis
Metabolic pathway
A series of linked reactions by which fuels are degraded and large molecules are constructed
example: glycolysis is a 10-step metabolic pathway converting glucose to pyruvate
Are interconnected series of enzyme-catalyzed reactions
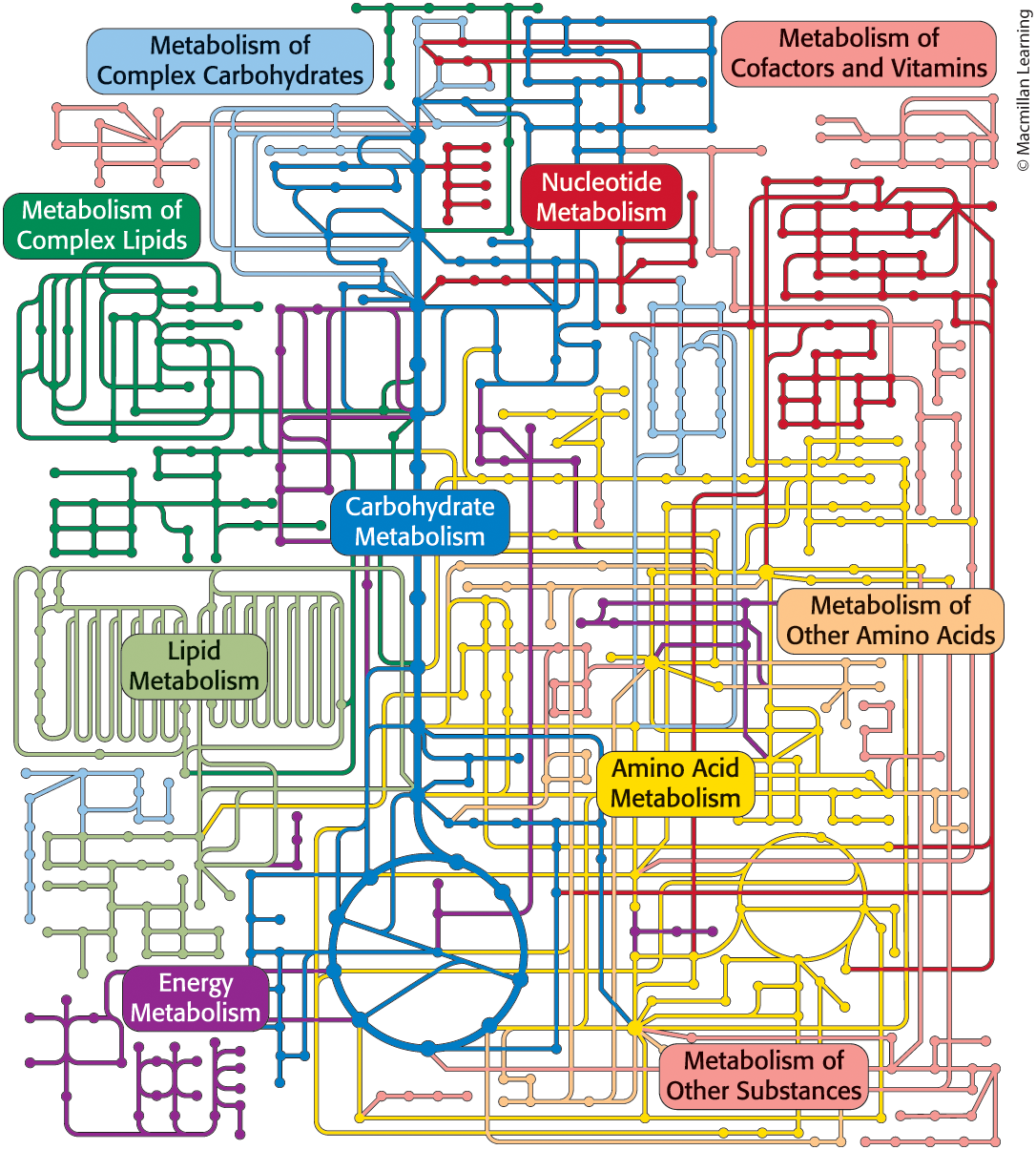
Themes common to all metabolic reactions
Metabolism is a coherent network containing many common motifs
adenosine triphosphate (ATP) is used as an energy currency to link energy-releasing (exergonic) and energy-requiring (endergonic) pathways
Either sunlight or the oxidation of chemical fuels powers ATP formation
~100 molecules serve as activated intermediates
Metabolism uses only a few kinds of mechanisms that are typically simple
Metabolic reactions are highly regulated because metabolic pathways are interdependent
Many of the enzymes involved in metabolism are organized into large complexes
Increases speed and efficiency
Allows efficient processing of unstable or toxic intermediates
Amphibolic pathways
Pathways that can either be anabolic or catabolic depending on cellular energy conditions
A thermodynamically unfavorable reaction
Can be driven by a favorable reaction
A metabolic pathway must meet 2 criteria
individual reactions must be specific
Each of the reactions in the pathway must be thermodynamically favored under real conditions
A reaction can occur spontaneously
Only if ∆G, the change in free energy, is negative.
Overall free-energy change for a chemically coupled series of reaction
Equals the sum of the free-energy changes of the individual steps
allows for the coupling of thermodynamically unfavorable and favorable reactions in enzyme active sites
In the example, the reactions are coupled by intermediate B
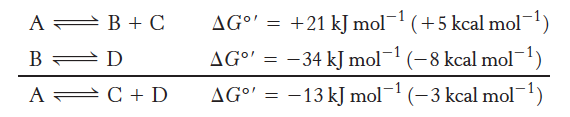
ATP Hydrolysis
Is exergonic
ATP is energy-rich because its triphosphate unit contains two phosphoanhydride linkages
The release of free energy from ATP Hydrolysis
Is by the:
formation of new covalent bonds
Formation of noncovalent interactions with water
Increase in entropy
∆G for ATP hydrolysis
Under typical cellular conditions is approx. -50 kJ mol-1
Structures of ATP, ADP, and AMP
Differ only by the number of phosphates
Enzymes
Catalyze the exchange of phosphoryl groups from one nucleotide to another
some reactions are driven by GTP, UTP, and CTP
Nucleoside monophosphate kinases
Enzymes that phosphorylate nucleoside monophosphates
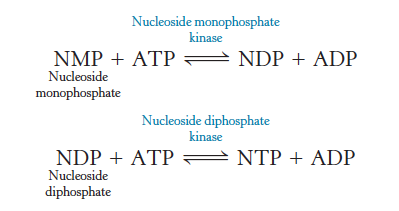
Nucleoside diphosphate kinase
Enzymes that phosphorylate nucleoside diphosphates
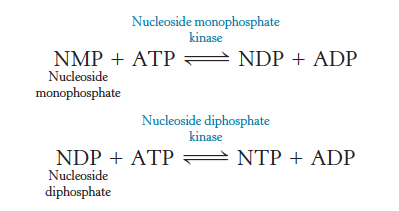
All nucleoside triphosphates
Are energetically equivalent
Are derivatives of ATP
Two important electron carrier (NAD+ and FAD) and the acyl group carrier, coenzymes A
ATP Hydrolysis
Drives metabolism by shifting the equilibrium of coupled reactions
the unfavorable conversion of the compound A into the compound B can be made possible by coupling to ATP Hydrolyis
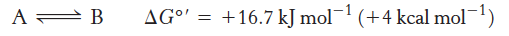
The equilibrium constant K′eq at 25°C
reveals the conversion of A to B cannot take place when the molar ratio of B to A is equal to or greater than 1.15 × 10−3.
K′ = [B]eq / [A]eq = e−∆G°′/2.47 = 1.15 × 10−3
Coupling a Reaction with ATP hydrolysis
Under standard conditions, ∆G°′ of hydrolysis is approximately −30.5 kJ mol−1.
Coupling the conversion of A to B with ATP hydrolysis renders the formation of B exergonic
Coupling these reactions under standard conditions changes the equilibrium ratio of B to A.
The high phosphoryl potential of ATP
Results from structural differences between ATP and its hydrolysis products
phosphoryl-transfer potential is a means of comparing the tendency of organic molecules to transfer a phosphoryl group to an acceptor molecule
Example: ATP has a higher phosphoryl-transfer potential than glycerol 3-phosphate
Is explained by its structure
Phosphoryl-transfer potential
The tendency of an organic molecule to transfer its terminal phosphoryl group to water
example: ATP has a higher phosphoryl-transfer potential than glycerol 3-phosphate
is an important form of cellular energy transformation
ATP has a high phosphoryl-transfer potential because of:
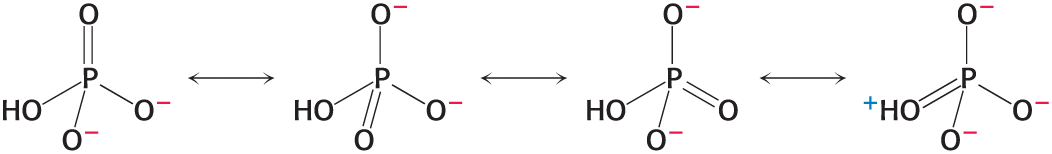
orthophosphate (Pi) has greater resonance stabilization than any of the ATP phosphoryl groups
Electrostatic repulsion of the triphosphate unit
The entropy of the products of ATP hydrolysis is greater
ADP and Pi are stabilized due to hydration
Some compounds
Have higher phosphoryl-transfer potential than ATP
examples: phosphoenolpyruvate (PEP), 1,3-bisphosphoglycerate (1,3-BPG), and creatine phosphate
These compounds can transfer their phosphoryl group to ADP to form ATP
Compounds with High Phosphoryl-Transfer Potential
Can be used to make ATP from ADP
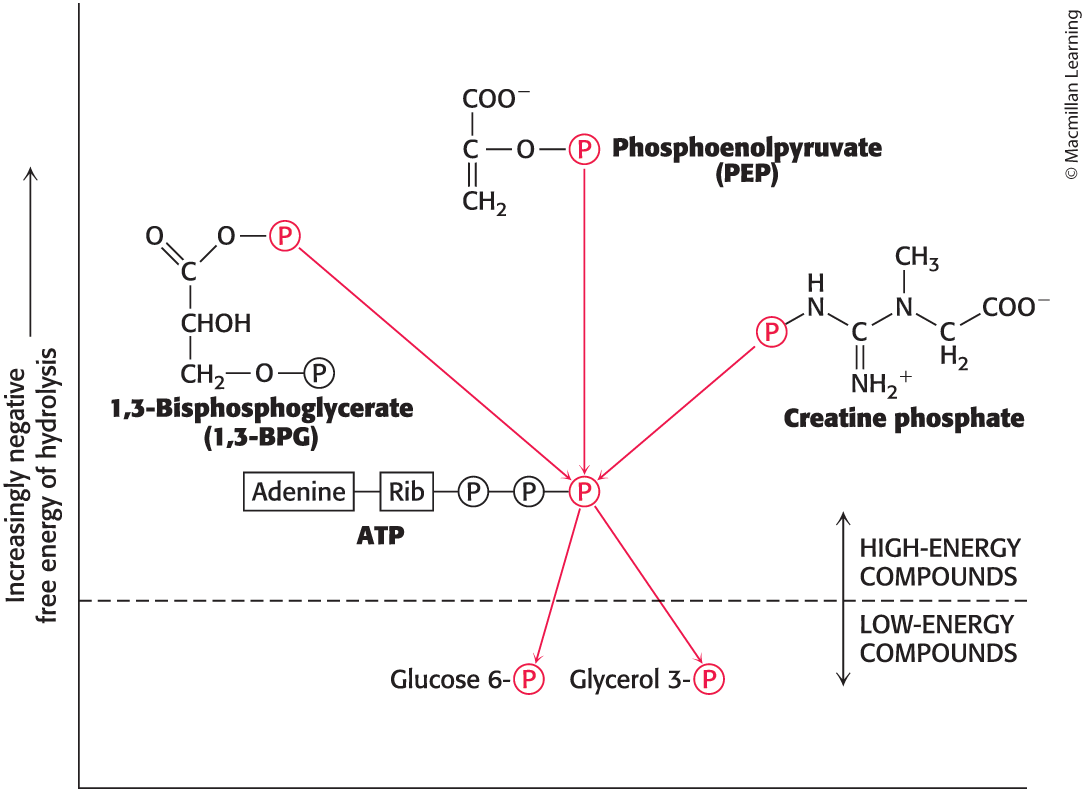
Standard free energies of hydrolysis of some phosphorylated compounds
ATP has an intermediate phosphoryl-transfer potential among biologically important phosphorylated molecules
Enables ATP to function efficiently as a carrier of phosphoryl groups
Creatine Phosphate
Serves as a reservoir of high potential phosphoryl groups
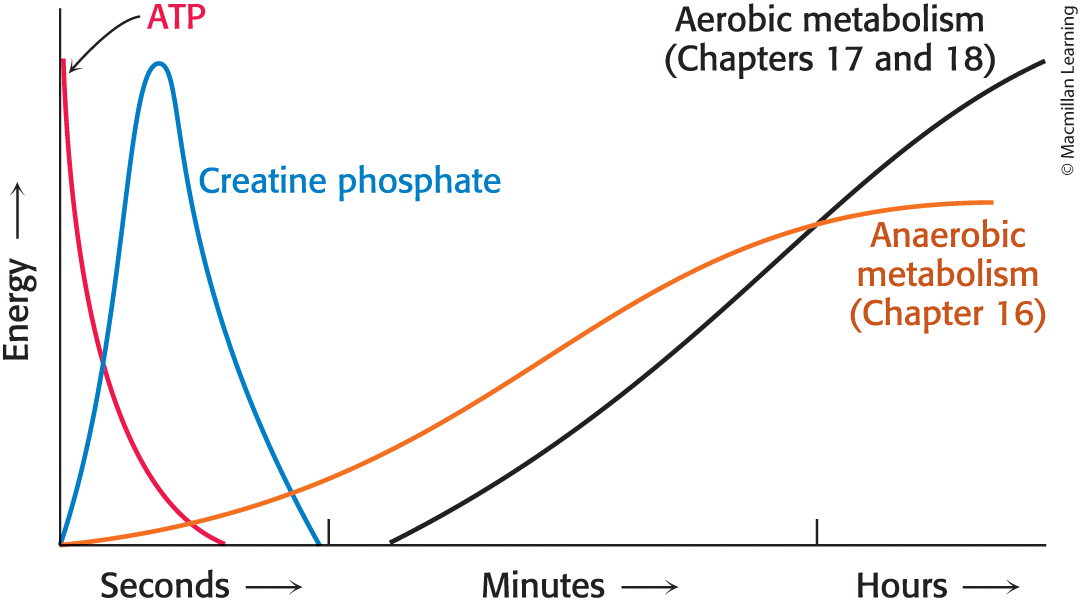
ATP in muscle
Sustains contractile activity for < 1 second
Creatine kinase
Catalyzes the regeneration of ATP from creatine phosphate and ADP
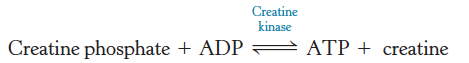
∆G°′ of hydrolysis of creatine phosphate
is −43.1 kJ mol−1
The sources of ATP
Change as Exercise Duration Increases, even within the first few seconds
The oxidation of carbon fuels
Is an important source of cellular energy
ATP is the principal immediate donor of free energy for biological activities, but ATP is limited
ATP must be constantly regenerated from ADP
Oxidation of Fuel Molecules
Takes place one carbon at a time
carbon atoms in fuels are oxidized to yield CO2
The more reduced a carbon atom is, the more free energy is released upon oxidation
Electrons are ultimately accepted by oxygen to form H2O
Fats
Are a more efficient fuel source than carbohydrates because the carbon in fats is more reduced
Compounds with High Phosphoryl-Transfer potential
Can couple carbon oxidation to ATP synthesis
Glyceraldehyde 3-phosphate
A metabolite of glucose formed during glucose oxidation
the C-1 carbon is at the aldehyde-oxidation level and is not in its most oxidized state
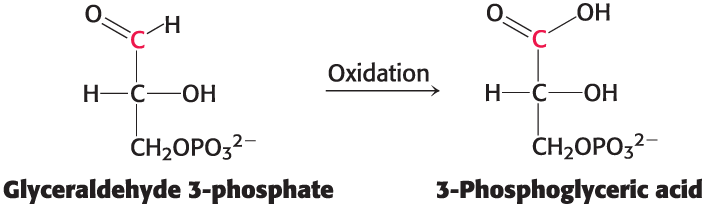
Oxidation of Glyceraldehyde 3-Phosphate
Does not occur directly
carbon oxidation generates , 1-3-bisphosphoglycerate (1,3-BPG), and the electrons released are captured by NAD+ to form NADH
1,3-BPG has high phosphoryl-transfer potential, and its hydrolysis can be coupled to the synthesis of ATP
Ion gradients across membranes
Provide an important form of cellular energy that can be coupled to ATP synthesis
the oxidation of fuel molecules or phototrophy produces electrochemical potentials of ion gradients across membranes
Serves as a versatile means of coupling thermodynamically unfavorable and favorable reactions
In animals, 90% of ATP is generated when the energy of a proton gradient is coupled with ATP synthesis
This process is called oxidative phosphorylation
Proton Gradients Formed using the energy from either sunlight or chemical oxidation
Can power ATP synthesis
Phosphates
Play a prominent role in biochemical processes
__ esters are thermodynamically unstable yet kinetically stable in water
Kinetic stability is due to the negative charges that resist hydrolysis in the absence of enzymes
Their energy release can be manipulated by enzymes
The addition of a __ group changes molecule conformation and behavior
No other ions have the chemical characteristics of __
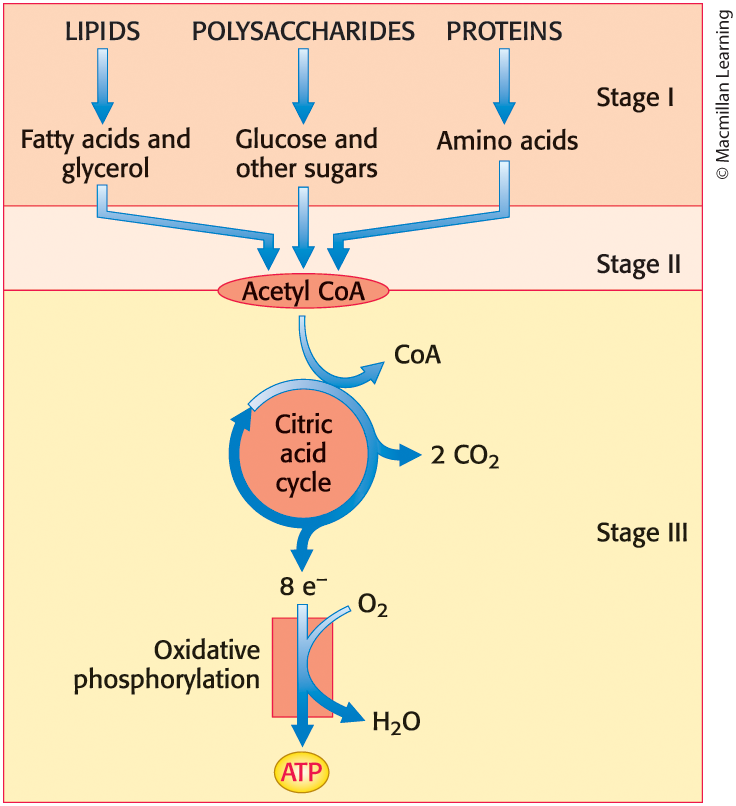
Energy from Food is Extracted in Three Stages
Stage 1: large molecules in food are broken down into smaller units
Stage 2: small molecules are degraded to a few simple units that play a central role in metabolism
Stage 3: ATP is produced from the complete oxidation of the acetyl unit of acetyl CoA
The extraction of energy from food molecules
By aerobic organisms
Metabolic pathways
Contain many recurring motifs
Activated carriers
Small molecules to which a chemical group or electrons have been added, which can then be donated to another molecule
frequently act as coenzymes or cosubstrates
Example: ATP is an activated carrier of phosphoryl groups
NADH
An activated carrier of electrons for fuel oxidation
fuel molecules transfer electrons to carriers, which then transfer their high-potential electrons to O2
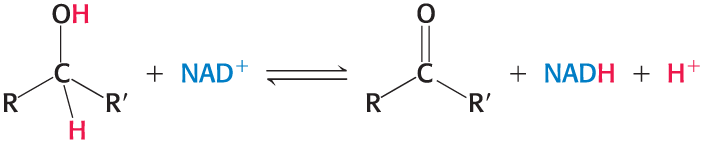
Nicotinamide adenine dinucleotide (NAD+)
Accepts a proton and two electrons in the oxidation of a substrate to form NADH
the reactive part is its nicotinamide ring
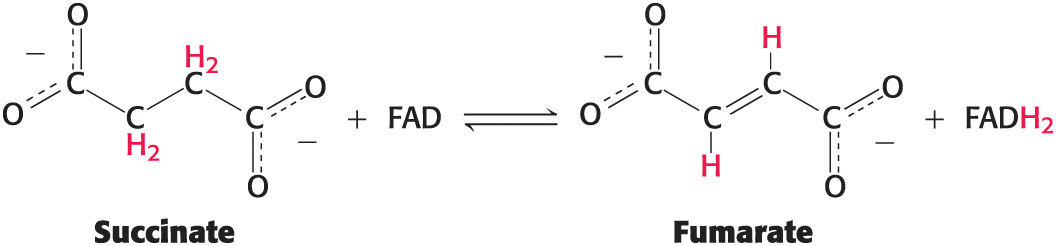
FADH2
An activated carrier of electrons for fuel oxidation
Flavin adenine dinucleotide (FAD)
Accepts two protons and two electrons in the oxidation of a substrate to form FADH2
the reactive part is its isoalloxazine ring
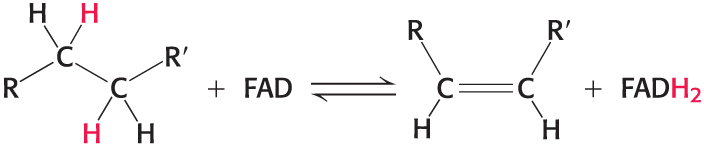
Structures of the Reactive Compounds of FAD and FADH2
Reveal that electrons and protons are carried by the reactive isoalloxazine ring component
the isoalloxazine ring is a derivative of the vitamin riboflavin
NADPH
Activated carrier of electrons for reductive biosynthesis
in most biosynthesis, precursors are more oxidized than the products
ATP and reducing power are needed
Example: four electrons are needed to reduce a keto group to a methylene group
__ is the electron donor in most reductive biosyntheses
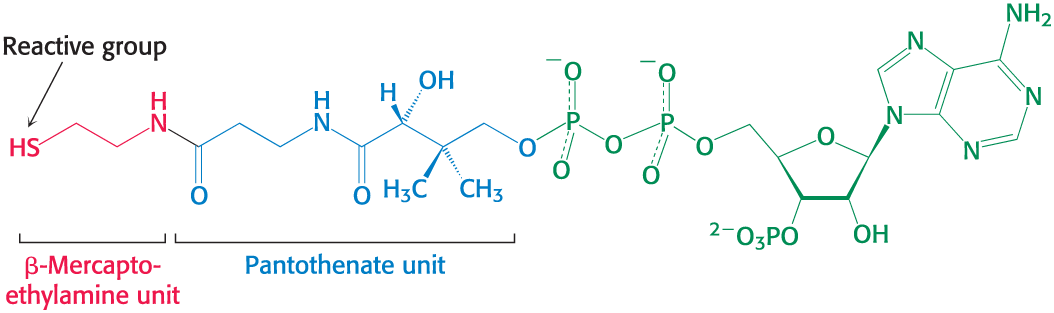
Coenzyme A (CoA)
Activated carrier of two carbon fragments
a carrier of acyl groups that is derived from vitamin B5 (pantothenate)
The reactive part is its terminal sulfhydryl group
Acyl groups are linked to __ by thioester bonds to form an acyl __
Acetyl linked to is called acetyl __.
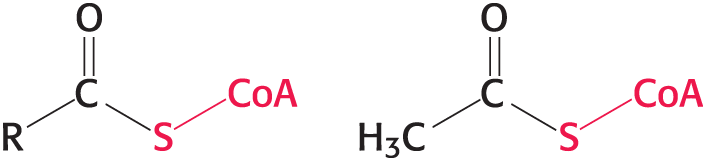
Structure of Coenzyme A
CoA-SH
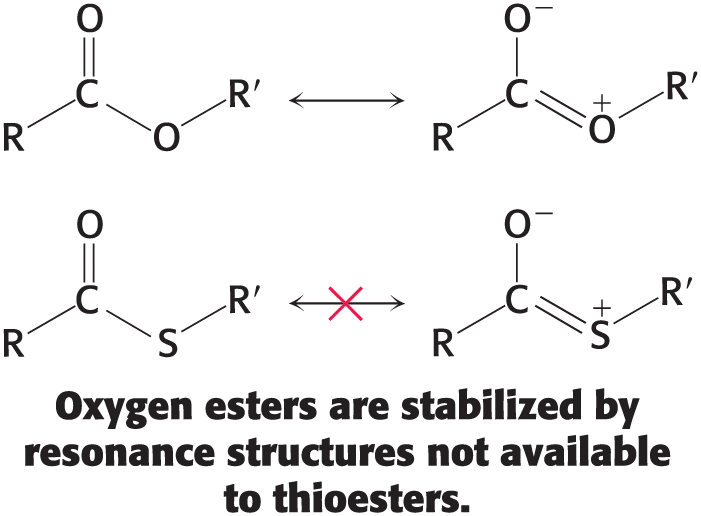
The transfer of the acyl group
Is exergonic because the thioester is thermodynamically unstable
The ∆G°′ for the hydrolysis of acetyl CoA has a large negative value.
Electrons of the C=O bond cannot form resonance structures with the C—S bond that are as stable as those that they can form with the C—O bond
Activated carriers
Illustrate key aspects of metabolism
kinetic stability allows enzymatic control over the flow of energy
NADH, NADPH, and FADH2 react slowly with O2 in the absence of a catalyst
ATP and acetyl CoA are hydrolyzed slowly in the absence of a catalyst
A small set of carriers accomplishes the majority of the exchanges of activated groups in metabolic pathways
Many activated carriers
Are derived from vitamins
Vitamins
Organic molecules that are needed in small amounts in the diets of some higher animals
must be modified to serve its function
Most activated carriers that act as coenzymes are derived from __
The B vitamins
Are a diverse group of small, water soluble molecules
Key reactions
Are reiterated throughout metabolism
the thousands of metabolic reactions can be subdivided into 6 types
Oxidation-reduction reactions
useful energy is often derived from the oxidation of carbon compounds
Examples: oxidation-reduction reactions of the citric acid cycle
Group-Transfer Reactions
Used to synthesize ATP and in signaling pathways, among others
example: phosphoryl group transfer
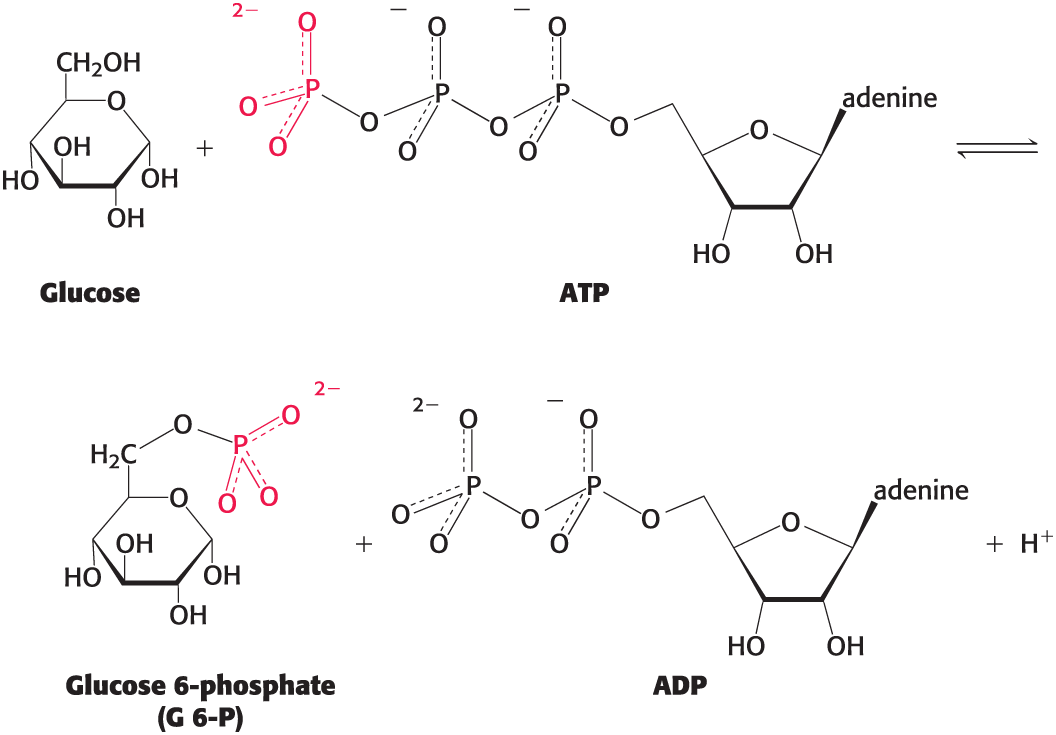
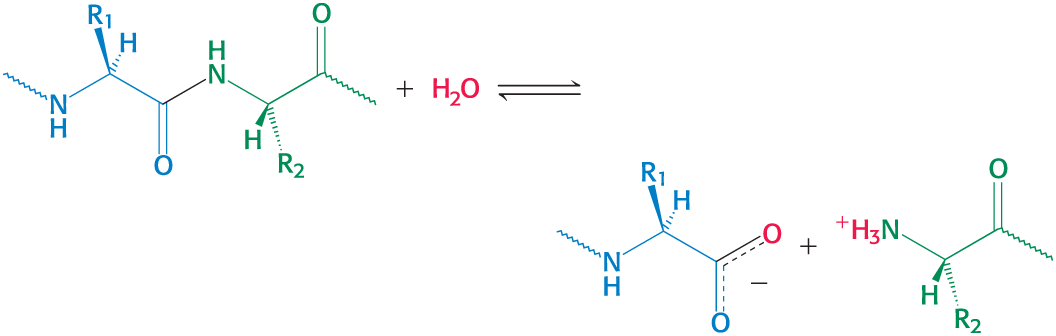
Hydrolytic Reactions
Hydrolysis cleaves bonds by the addition of water
commonly used to degrade large molecules
Example: hydrolytic cleave of proteins
Carbon Bond Cleavage Pt 1
Can occur by means other than hydrolysis and oxidation
example: the conversion of the six-carbon molecule fructose 1,6-bisphosphate into two three-carbon fragments during glycolysis
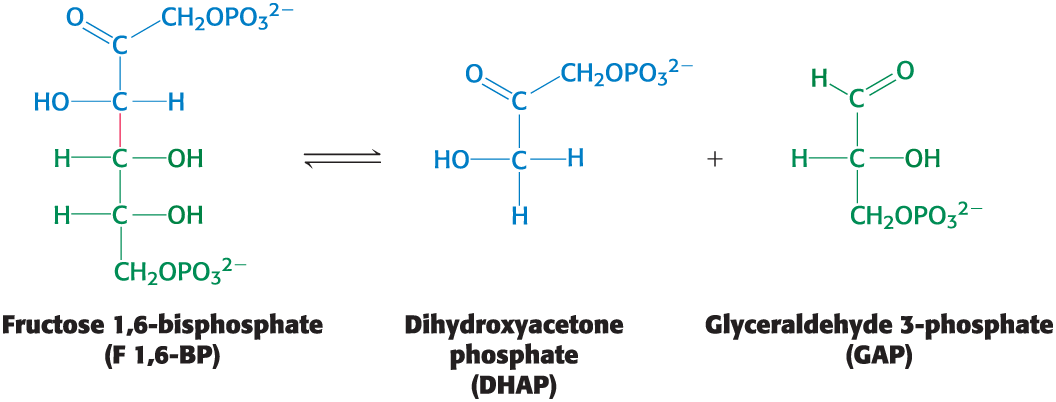
Carbon Bond Cleavage Pt 2
Dehydration is an important subclass
example: the generation of phosphoenolpyruvate from 2-phosphoglycerate
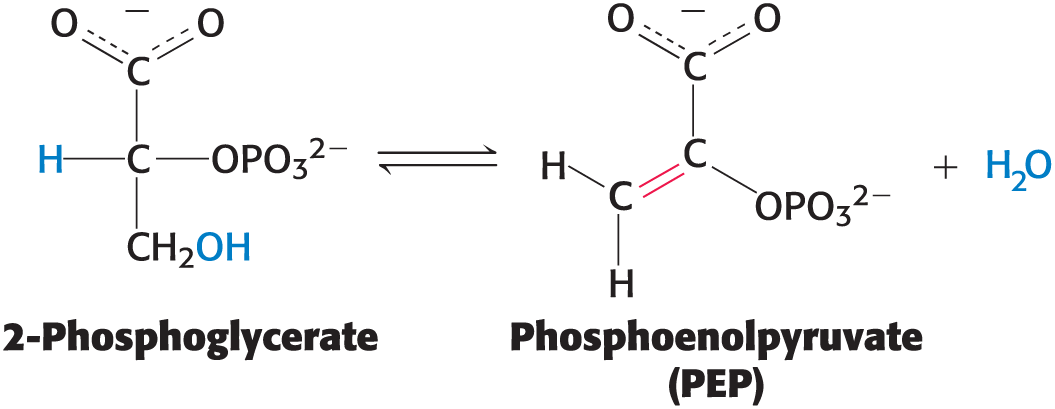
Isomerization reactions
Rearranges atoms within a molecule
typically to prepare the molecule for a subsequent reaction
Example: the conversion of citrate to isocitrate
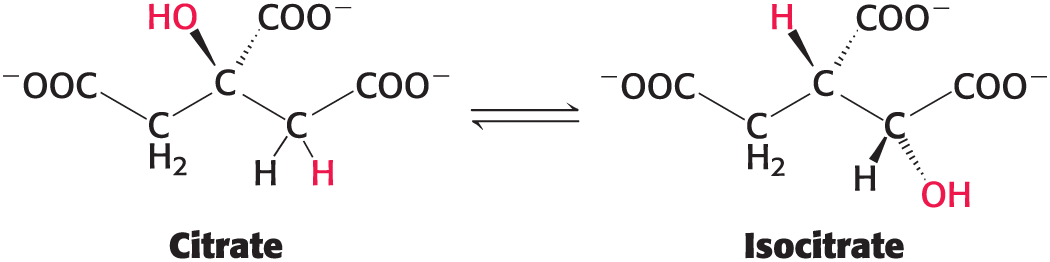
Ligation Reactions
Forms bonds using free energy from ATP hydrolysis
example: the formation of oxaloacetate from pyruvate and CO2
Metabolic processes
Are regulated in three principal ways
Metabolic pathways
Must be regulated to create homeostasis (a stable biochemical environment)
Metabolism is regulated by three mechanisms
altering the amount of enzymes
Restricting the accessibility of substrates
Regulating the catalytic activity of enzymes internally or externally
Controlling the amounts of enzymes
the amount of a particular enzyme depends on both its rate of synthesis and its rate of degradation
The level of many enzymes is adjusted by a change in the rate of transcription of the genes encoding them
Controlling the accessibility of substrates
Compartmentalization often segregates opposed reactions
example: fatty acid oxidation occurs in the mitochondrial matrix, while fatty acid synthesis occurs in the cytoplasm
Controlling catalytic activity
Catalytic activity is regulated allosterically or by covalent modification
feedback inhibition is an example of allosteric regulation
Concentrations of allosteric activators and inhibitors can be changed
Reversible covalent modification can control catalytic rates of enzymes
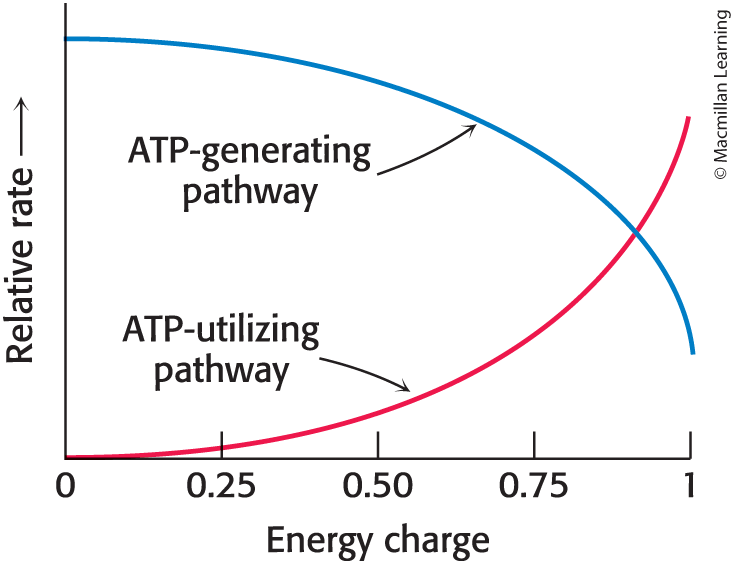
Energy charge
Proportional to the mole fraction of ATP plus half the mole fraction of ADP
ranges from 0 (all AMP) to 1 (all ATP)
ATP-generating pathways are inhibited, and ATP-utilizing pathways are stimulated under conditions of high-energy charge
Energy charge is maintained within narrow limits (0.90 to 0.95)
Enzymes regulating these pathways are allosterically inhibited or activated by binding to ATP or AMP
Is a useful concept for understanding how metabolism is regulated