energy
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
Stored energy
Potential energy is what type of energy?
Chemical energy
Stored in the chemical bonds between atoms (in glucose or ATP)
Kinetic energy
Energy type that takes place with objects or particles in motion
The equation for free energy is
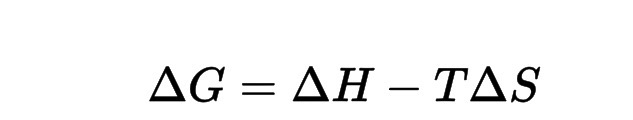
Enthalpy (H)
Total potential energy stored in the chemical bonds of a molecules.
Entropy (S)
Energy that is lost as disorder or heat. It’s energy we cannot use to do work.
Free energy (G)
This is the Gibbs free energy. It is the actual energy left over that the cell can use.
For a cell, the goal of life is
To maximize G (usable energy) and minimize S (lost heat)
Negative Delta G
(Energy OUT/ RELEASED = less free energy
Catabolism
You break a big molecule into smaller ones
Spontaneous
Meaning energetically favorable
Catabolism
Released stored energy by breaking complex molecules
Positive ^G
( energy IN/ABSORBED = More free energy)
Anabolism
You build a complex molecules from simple ones
Non-spontaneous
Endergonic
Anabolism
Using energy from catabolism to synthesize complex molecules
Entropy (S)
Energy that is lost ad disorder/randomness or heat - unusable energy
Transition state
An unstable, high energy state where bonds are being stretched or broken
Activation energy (Ea)
Initial energy necessary for reactions to occur
The higher the activation energy (Ea)
The slower the chemical reaction
The products are at a lower energy state than the reactants
Exergonic
Which of the following is the best way to judge the relative activation energies (Ea) between two given chemical reactions
Compare their reaction rates