Biology 1020 Unit 2
1/84
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
85 Terms
Amphipathic
hydrophilic component and hydrophobic component in same molecule
Cholesterol
small hydrophilic head, steroid rings (more rigid), hydrocarbon tail (amphipathic molecule)
how do phospholipids arrange themselves?
with their hydrophobic tails touching on the inside (away from aqueous environment) and hydrophilic heads at the top and bottom (towards extracellular space or cytoplasm respectively)
liposome
tiny, synthetic, spherical vesicles composed of a lipid bilayer—similar to cell membranes—that can encapsulate drugs. they are built in vitro and can have membrane proteins inserted
endocytosis
an active cellular transport process that engulfs extracellular materials—fluids, nutrients, or pathogens—by merging off portions the plasma membrane to form internal vesicles.
exocytosis
intracellular vesicles fuse with the plasma membrane, releasing their contents (neurotransmitters, hormones, or waste) into the extracellular space
lumen
internal volume enclosed by a membrane
membrane function
1) compartmentalization: forms a physical boundary that divides cellular space for different purposes
2) scaffold for biochemical activities: interacting cellular components can be localized to membrane (embedded or peripherally-attached) for more effective interaction
3) selectively permeable barrier: molecules require gate or channel to enter cell; protects cell
4) signal transduction: membranes have receptors that combine w/ specific molecules (ligands) to regulate processes in cell
5) interaction w/ neighboring cells or ECM
lipid bilayer/plasma membrane composition
1) amphipathic membrane lipids (phosphoglycerides, glycolipids, cholesterol)
2) membrane proteins: integral proteins, peripheral, lipid-anchored —> proteins are all free to move within the plane of the membrane unless they are bound
3) membrane carbohydrates in OUTER leaflet: glycoproteins or glycolipids
4) lipid rafts: membrane domains concentrated in cholesterol and lipids with long saturated tails
integram membrane/transmembrane proteins
penetrate lipid bilayer; have domains that protrude both sides of the membrane
their membrane spanning hydrophobic alpha helices tend to be 20-25 amino acids in length
can be used for signaling: receive ligands on extracellular side and relay info to intracellular side
can also be channels or transporters
are amphipathic (the part that spans membrane is hydrophobic, the rest is hydrophilic)
peripheral membrane proteins
located entirely outside of lipid bilayer, associated by noncovalent bonds
can be extracted w/o disrupting the membrane (using high/low salt or pH)
can be recruited and released as needed
lipid-anchored membrane proteins
located outside lipid bilayer but are covalently linked to a lipid molecule in the bilayer
ie. GPI anchored protein
where are membrane carbohydrates located
on the outer leaflet; they point into extracellular space. cells are “sugar-coated“. sugars may be attached to lipid or protein (glycolipid or glycoprotein)
glycoproteins
oligosaccharides are attached to amino acids of a membrane protein
glycolipids
oligosaccharides are attached to membrane lipids
what is the fluid mosaic model of the plasma membrane made of
membrane lipids (phospholipids, cholesterol, etc), proteins (integral membrane, peripheral, lipid-linked), carbohydrates (glycoproteins, glycolipids)
very flexible and dynamic fluid
cholesterol function
cholesterol fits easily into gaps between phospholipid molecules in a lipid bilayer
physically interrupts the van der Waals interactions between nonpolar tails —> helps make the membrane more fluid in colder temperatures
cholesterol also has rigid steroid rings, so they could also provide structure/rigidity in warmer temperatures
overall, it acts as a buffer to keep the membrane within the optimal fluidity range for a wider range of temperatures and helps the membrane stay stable
lipid rafts
specialized membrane regions where there are certain types of lipids that interact favorable with each other and travel toegther
example of lateral asymmetry since they are stiffer/more rigid regions of cell membrane containing more cholesterol and saturated fatty acid tails —> tighter packing (different from the rest of the surrounding cell membrane environment aka a sea of disordered phosphatidylcholine)
high concentration of signaling proteins and molecules, centers for signal transduction
lateral asymmetry
asymmetry looking at just one leaflet (regions of the same leafet having different properties)
ie. lipid rafts
tight junction-mediated segregation of outer leaflet lipids of the apical and basolateral sides of epithelial cells
sound sensing hair cells of the inner ear
transbilayer asymmetry
refers to difference between inner and outer leaflet. the two different leaflets have distinctly different lipid compositions
how do the outer and inner leaflets differ in composition?
outer leaflet is rich in phosphatidylcholine (PC) and sphingomyein (localizes to lipid rafts). Glycolipids are only found in this leaflet for cell signaling purposes.
phosphatidylserine (PS) which is negatively charged is only on inner leaflet —> charge asymmetry
phosphatydlinositide is also confined to the inner leaflet, which is a key signaling lipid and protein docking lipids
Flippase enzymes move inner membrane lipids inside if they accidentally flip over to maintain membrane asymmetry
Overall, cholesterol is evenly distributed between leaflets, but the phosphoglycerides (PC, PS, PE, and PI) and sphingomyelin (SM) are asymmetrically distributed
key experiments that proven existence of fluid mosaic model and lipid bilayer
Gorter and Grendel: measured the lipid content/cell surface area of red blood cells —> supports lipid bilayer
FRAP: shows recovery after photobleaching (showed membrane fluidity)
Freeze fracture TEM: showed that proteins penetrate through membranes —> supports fluid mosaic model
Frye and Edidin: demonstrates rapid intermixing of human + mouse antigens —> shows membrane fluidity
Gorter and Grendel experiment (1925)
isolate RBCs from whole blood by using low speed centrifugation to pellet RBCs and leave white blood cells as a layer on top of the RBC pellet
calculated surface area of red blood cell (good choice because it has no internal organelles —> no extra intracellular membranes)
extract lipids from known number of RBCs using organic solvent
forms a lipid monolayer on water trough (phospholipids spontaneously arrange themselves so that their hydrophobic heads are in the water)
measure surface area of monolayer
the surface area of monolayer was double the surface area of the RBC determined by light microscopy —> showed that there was a bilayer
Iateral diffusion
membrane phospholipids move rather quickly within respective leaflets spontaneously
transverse flip flop
requires flippase enzyme… phospholipids do not spontaneously move from one monolayer to another
what does membrane fluidity depend on?
length of hydrocarbon tails: longer tails —> more VDWs —> tighter packing —> less mobility
shorter tail membranes are more fluid at cooler temperature
temperature: higher temperatures —> more fluid bc phospholipids can make and break VDWs more easily
cholesterol: has rigid ring structure, so it helps maintain fluidity @ optimal level at high and low temperatures; acts as a stabilizing buffer
ratio of unsaturated fatty acid tails to saturated fatty acid tails:
unsaturated phospholipids have kinks & more double bonds —> not able to pack as closely —> less VDWs —> more fluid (lower melting point)
saturated phospholipids are straight chains —> tight packing —> more VDWs —> less fluid
all cell membranes contain mix of unsat and sat fatty acids… its the ratio that determines fluidity
transition temperature
the specific temperature at which a biological membrane changes its physical state from a rigid, ordered "gel" phase to a fluid, disordered "liquid-crystalline" phase. as temperature increases, there is a sharp transition between rigid and fluid states. the transition temperature is this temperature
what is the importance of fatty acid unsaturation for membrane fluidity? of enzymes that desaturate fatty acids?
unsaturated fatty acids allow for more membrane fluidity. more double bonds —> more kinks —> less lateral packing —> less VDWs —> more fluidity
what is meant by the membrane’s transition temp, and how is it affected by the degree of saturation or length of fatty acyl chain? how are these properties important in the formation of lipid rafts.
the membrane’s transition temp is the temp at which it goes from solid/rigid to fluid state.
If there is a greater proportion of unsat, fluidity will be higher since less tight packing —> less VDWs.
The shorter the fatty acid tails are, the more fluid the membranes since there is less space for VDWs and interactions.
Lipid rafts would have longer fatty acid tails and more saturated phospholipids since lipid rafts are stiffer and thicker
how can the two sides of a lipid bilayer have different ionic charges? what is the functional significance of this?
different ionic charges occur bc the two sides are composed of different phospholipids with different properties. Phosphotidylserine (negatively charged) is located solely on the inner leaflet by flippase. This is important since many signaling proteins in cytosol are positively charged, so they attract. Additionally, difference in charges creates an electrical gradient across the membrane to store energy
Fluorescence recovery after photobleaching (FRAP)
method for measuring fluidity of membrane or mobility of membrane protein (lateral diffusion)
FRAP process
label membrane lipids or proteins w/ fluorescence (tag w/ antibodies and very small fluorophores)
photobleach spot with laser beam
measure rate of recovery in that region over time… membrane lipid or protein should spontaneously diffuse back into “blank“ area if they are mobile
rate of recovery can tell you how fluid it is
does the cell need to be fixed for labeling a membrane lipid
no, we’re labeling something on the membrane and don’t need to get inside cell. we don’t have to poke holes in the membrane.
RE: we always have to fix cells for immunofluorescence since we’re tagging a protein/molecule inside the cell
photobleaching
high energy dose of blue light (for fluoroscein) or green light (for rhodamine) induces covalent changes in ring structure, destroying capacity to emit additional photons of green/red light
rhodamine
fluorophore that is excited by green light and emits red light
fluorescein
fluorophore that is excited by blue light and emits green light
FRAP graph for a fully mobile lipid/protein
graph shows full recovery at the same level as before
FRAP graph for bound lipid/protein
not full recovery, graph plateaus and does not reach original height
how do we label integral membrane proteins for FRAP?
direct immunofluoresence: use a primary antibody that has a fluorophore conjugated directly to it (no secondary antibody bc we want the tag to be as small as possible)
transfect cells with construct (DNA plasmid) encoding GFP-tagged membrane protein (fusion protein)
why would fluorescence not recover after FRAP?
protein’s lateral mobility may be restricted or anchored down in some way
modes of restricting integral membrane protein lateral mobility
lateral clustering of IMPs (more closely packed)
ECM binding/clustering of IMP: anchored to something in ECM
cytoskeletal restraint of IMPs: anchored to something in cytoplasm (interior)
usually a scaffold protein (ankyrin or spectrin)
cell adhesion mediated clustering of receptors (cell-to-cell interaction)
ie. tight junctions
gap junction
communication portal/tunnel between two neighboring cells; made up of connexions (transmembrane protein). not mobile at all because they are physically bound together (in FRAP, the photobleached gap does not close). allows small ions, sugars, amino acids, etc to flow directly from one cell to another
lateral aggregation
occurs within the plane of a membrane; proteins cluster together and move slower through the membrane (ie. lipid raft). for a gap junction, the connexons huddle together in a dense patch in the membrane
extracellular aggregation
they are completely stuck and immobile bc they are physically bound to connexons of the other cell in a gap junction. occurs when proteins are forced into a cluster by something outside the cell.
freeze fracture TEM
freeze tissue in liquid nitrogen chilled solvent
specimen is split open w/ sharp blade (chooses path of least resistance and splits down the bilayer)
peel membrane layer back and deposit heavy metal stain on newly exposed surfaces
TEM. Lots of bumps will be visible, while opposite leaflet has pit where the protein was displaced
P face: cytoplasm side, E face: extracellular side
Frye and Edidin experiment (1970)
fused a mouse cell and human cell into a heterokaryon
labeled mouse antigens (fluorescein) on one side and human antigens (rhodamine) on the other side (secondary antigens)
they saw rapid intermixing of the antigens within 40 minutes, demonstrating that the membrane is fluid
also showed that mosaic formation is dependent on temperature: warmer temps = more mosaic
lateral asymmetry in sound sensing hair cells of inner ear
FRAP demonstrates that the apical side has a higher recovery than lateral and basal sides —> apical side is more fluid. tight junctions act as physical barricade and prevents lipids and proteins from apical area from drifing down into basal domain. need to maintain lateral asymmetry
epithelial cells and lateral asymmetry
apical side is tougher & made to face harsh conditions of lumen
more saturated fatty acids, cholesterol, and lipid rafts
basal lateral side is less tough
maintained by tight junctions
tight junctions
composed of transmembrane proteins that reach out from the cell and bind directly to some proteins on neighboring cell
fuses two outer leaflets of two membranes together
can serve as a zipper to prevent leakage (ie. prevents acid from gut from leaking into bloodstream)
can serve as a dam to prevent contents from apical side and basolateral side from mixing
restricts lateral mobility of outer but not inner leaflet
active transport
energy is required to move this solute up or against its concentration gradient from low to high concentrations. can occur directly with ATP hydrolysis or indirectly (cotransport) without ATP
what type of molecules can cross the membrane through simple diffusion?
small nonpolar molecules (ie. O2)
small uncharged polar molecules (H2O) (can also use aquaporin channels for bulk movement)
what types of molecules cannot cross the membrane by itself?
ions
large polar molecules
structural anatomy of the bacterial potassium (K+) ion channel
contains selectivity filter that only allows dehydrated K+ ions to pass through channel (no larger ions or smaller ions (giving up hydration shell is unfavorable))
K+ is positively charged, and the selectivity filter contains electronegative oxygens which are partially negative —> draws K+ in
contains gating alpha helices, which decide when the ions can pass (must go through conformation change)
types of ion channel gates and how they open
voltage gated: responds to an initial stimulus (slight voltage change and depolarization) —> triggers opening of voltage gated ion channel once at threshold —> more depolarization
ligand gated (can be extracellular or intracellular) —> produces conformational change
mechanically gated: responds to tension and pressure, usually attached to cytoplasm and is physically pulled open
ligand
any molecule that binds to a receptor
agonist
specific type of ligand that activates the receptor and triggers its normal repsonse. all agonists are ligands, but not all ligands are agonists
voltage gated sodium ion channel opening process
resting membrane potential is -70mV (inside is 70mV more negative than outside)
small depolarization occurs —> membrane becomes less negative & reaches threshold ~55mV
voltage gated Na+ channels open in response, causing a spike and driving action potential as Na+ moves into cell
repolarization and return to resting state as K+ channel opens and K+ moves out of cell
patch clamp technique
key experiment that allowed scientists to record an ion channel opening and closing
tiny pipette isolates and seals off one ion gated channel. the only way ions can get through is if channel opens
what is the mechanism that snaps voltage gated channels closed quickly (even before membrane repolarizes)
inactivation peptide blocks flow of K+ through membrane like a tampon… this is the phase when repolarization has not happened yet but channel has closed
transporter mediated transport/facilitated transport
mediated by protein transporters that undergo a conformational change when bound by solute
uniporter
type of facilitated transporter; transport one thing at a time
symporter
type of facilitated transporter, coupled transport (2 things at once in the same direction)
antiporter
type of facilitated transporter, coupled transport (2 things at once in opposite directions)
sodium potassium pump
example of active transport pump:
3 Na+ ions bind —> ATP binds —> close cytoplasmic side —> open to outside —> 2K+ bind —> eventual release of K+ with ATP hydrolysis
each ATP hydrolysis results in 3 NA+ moving outside and 2K+ moving inside
types of transport by integral membrane proteins (summary)
passive:
direct diffusion
channels
facilitated diffusion by transporters
active:
direct w/ ATP; pumps
indirect (ie. coordinated movement of one solute up its concentration coupled to another moving down)
Na+/glucose co transporter/symporter
indirect active transport driven by Na+ gradient across plasma membrane
movement of glucose from low extracellular concentration to high cytosolic concentration (against gradient) driven by energy stored in Na+ gradient
sodium moves down concentration gradient while glucose moves against concentration gradient; both moving into membrane
polarized cell
apical and basolateral parts of cell have distinct protein/lipid compositions (ie. epithelial cells)
glucose transport across epithelial cell
intestinal lumen —> epithelial cell via active transport (low to high concentration)
epithelial cell —> blood space via diffusion (high to low concentration)
mediated by selective localization of Na+/glucose cotransporter in apical domain and facilitated diffusion of glucose transporter/uniporter to basolateral domain
channels
passive
pumps
active
transporters
uniporters: passive
cotransporters: active
transmembrane domain of proteins must consist of ______ to have favorable interactions w/ hydrophobic tails
nonpolar amino acids
hydrophobicity plots
hydrophobic regions of ~20 amino acids indicate a transmembrane domain (can count how many there are based on graph)
what is amino acid length of hydrophobic segments
20 amino acids
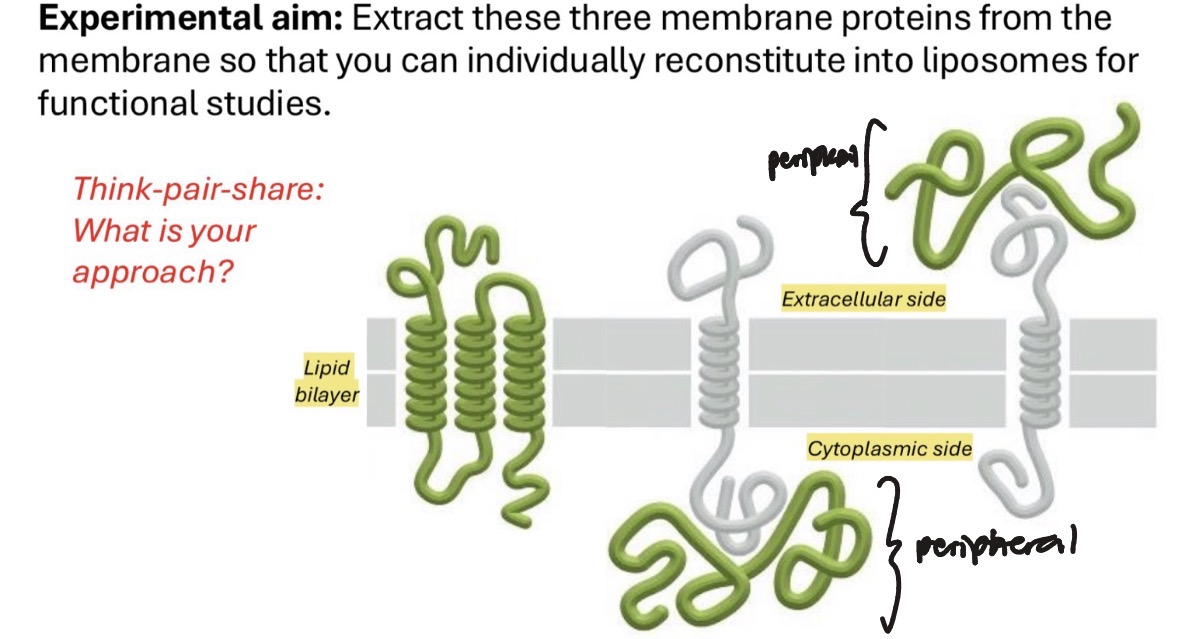
how do we extract integral membrane proteins
extract using an amphipathic detergent that will dissolve bilayer
how do we extract peripheral membrane proteins
extract w/o dissolving bilayer, use high/low salt extraction
ionic detergents
solubilize and denature integral membrane proteins (ie. SDS). use if doing immunoblot after
nonionic detergent
more gentle, preserves protein structure and function while solubilizing/extracting it (ie. Triton X-100). use if want to study function (structure determines function)
*after poking holes, use differential centrifugation to remove cytosol (supernatant)

reconstitution
taking parts from a cell to study in a cell-free environment in liposomes or in vitro environment to eliminate noise and get more control
how can SDS ionic detergent work well with immunoblot
SDS is one of the early steps in preparing samples for a western/immunoblot.
SDS denatures protein into linear form and gives it a positive charge so it can flow towards positive end of gel
allows protein to move according to its molecular weight instead of being affected by charge
hydropathy plots
visualize amphipathic nature of integral membrane proteins
positive peaks on hydropathy plot
stretches of hydrophobic amino acids, often indicating transmembrane alpha helices
low/negative regions
hydrophilic segments exposed to cytosol or extracellular space