Biological Molecules
1/49
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
50 Terms
Calcium ions
Muscle contraction and nerve impulse transmission
Sodium ions
Co transport, reabsortoption of water in kidney, and nerve impulse transmission
Potassium ions
Stomata opening and nerve impulse transmission
Hydrogen ions
PH determination and catalyses reaction
Ammonium ions
Nitrogen cycle —> bacteria convert ammonium ions into nitrate ions
Nítrate ions (NO3-)
Mineral ion absorbed by plants to provide a source of nitrogen to make amino acids
Hydrogencarbonate (HCO3-)
Maintain the pH of the blood
Chloride ions CL-
Negative charge to balance positive sodium ion and potassium ions in cells
Phosphate ions (pO43-)
Formation of phospholipids for cell membranes, nuclei acid ad ATP formation
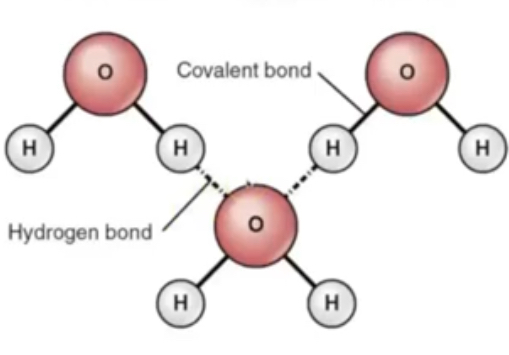
describe the bonds in water
Polar molecule due to uneven charge —> H bonds form between oxygen and hydrogen as positive and negative regions interact with eachother
Individual H bonds weak, but together are strong
The four key properties of water
Solvent in reactions
Transport medium
Coolant
Provides habitats
Describe the role of water as a solvent
Polar (hydrophilic) molecules dissolve in water as water is polar
Slight positive charge on hydrogen atoms attract negative solutes and slight negative charge on oxygen attracts positive solutes
Non polar (hydrophobic) molecules cannot dissolve in water - repelled
Describe the role of water as a transport medium
In blood, or in xylem of plants - polar substances dissolve and are transported
Cohesion = water molecule s stick together via H bonds
Water moves up xylem in plants due to transpiration, it is a continuous stream due to cohesion
Advantage = easier to draw up column than individual
Describe the role of water as a coolant
High specific heat capacity = due to energy required to break hydrogen bonds between water molecules
Advantage = Internal temp or plants / animals stay constant (enzymes dont denature reduce activity levels)
Large latent heat of vaporisation = due to energy required to break H bonds between water molecules to turn into a gas
Advantage = sígnica t cooling effect
Describe the role of water as a habitat
Water buffers temperature, provide a stable environment
Cohesion = surface tension in water, allow small insects to live on surface Away from predators
Ice is less dense than liquid water (due to hydrogen bonds) = so ice floats on top of water, provide a surface habitat for animals, and insulate water below for aquatic organisms
Define monomer and polymer
Monomer = smaller units that create larger molecules
Polymer = made from lots of monomers bonded together
Give examples of three monomers and their polymers
Glucose = starch, cellulose, glycogen
Amino acids = protein
Nucleotide = DNA and RNA
What do carbohydrates split into?
Monomers = single unit (glucose)
Diners = 2 sugars unit
Polymers = many sugar units
Structure of alpha glucose
C6H12O6
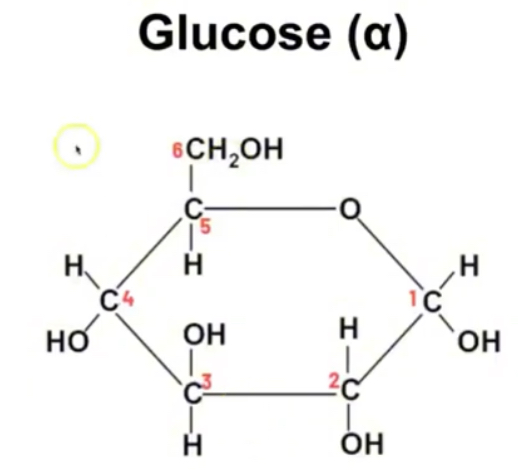
Structure of beta glucose
Carbon 1 has OH group at the top
Isomers of glucose - same molecular formula different sturtal formula
Describe ribose
Monosaccharide
Pentose sugar, has 5 carbons
Describe a disaccharide
Made of two monosaccharides
Joined by a glycosidic bond
Formed via condensation reaction
Name examples of disaccharides forming
Glucose + glucose —> maltose + water
Glucose + galactose —> lactose + water
glucose + fructose —> sucrose + water
Name the isomers to make each disaccharide
Alpha g + alpaca G = maltose
Beta g + Galactose = lactose
Alpha G + fructose = sucrose
What is a condensation reaction
Joining two molecules together, by removing water
Chemical bond is formed
What is a hydrolysis reaction?
Splitting apart of molecules through addition of water molecule
Chemical bond is broken
Descibe how condensation reactions and hydrolysis happens in disaccharides
On carbon 1 and carbon 4, water is eliminated from hydroxyl groups
1-4 glycosidic bond
Water added back too hydroxyl group for hydrolysis to make 2 monosaccharides
Describe the location of starch and its monomers
Plant cells (e.g chloroplast)
Alpha glucose
Bonds = 1,4 glycosidic bonds in amylose
1,4 and 1,6 in amylopectin
What is the function and suture of starch?
Stores glucose
Amylose = un branched helix, can compact to fit a lot of glucose in small space
Amylopectin = branched molecule, increases surface area for rapid hydrolysis back to glucose
Insoluble = doesn’t affect water potential
Descibe the location and monomers of cellulose
Location = Plants, cell wall
Monomers = beta glucose, 1-4 glycosidic bonds
Descibe the structure and function of cellulose
Structure strength for cell wall
Polymer forms long straight chains that are held in parallel by hydrogen bonds —> make fibrils
Macrofirbirls combine to make cellulose fibre
Insoluble - doesn’t affect water potential
Hydrogen bonds provide collective strength
Describe the location and monomers of glycogen
Alpha glucose
1-4 and 1-6 glycosidic bonds
Animal = muscle / liver cells
Descibe the structure and function of glycogen
Store of glucose
Highly branched molecule - increases surface area for rapid hydrolysis back to glucose
Insoluble
Define features of lipids
Macromolecules, dont make polymers
Non polar
Insoluble in water
Dissolve in organic solvents (ethanol)
Hydrophobic
Made up of fatty acids and glycerol
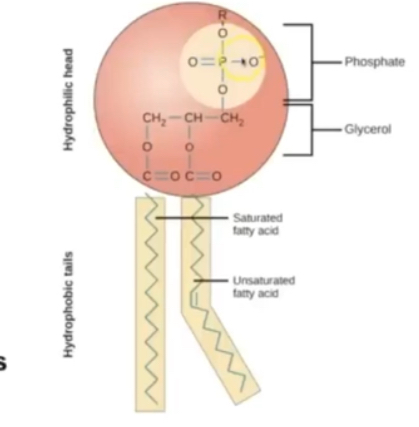
Describe the structure oftwo lipids: triglycerides and phospholipids
T = glycerol molecule and 3 fatty acids
P = glycerol backbone + phosphate group + 2 fatty acids
How are triglycerides made?
Formed via condensation reactions between one molecule of glycerol and three fatty acids
Ester bond formed from this (3, one for each fatty acid that bonded )
R group = fatty acids, what does it mean if they are saturated / unsaturated?
Saturated fatty acids = hydrocarbon chain has only single bonds between carbons
Unsaturated fatty acids = hydrocarbon chain has at least one double bond between carbons
Describe the properties of triglycerides
Can transfer energy = large ratio of energy storing carbon hydrogen bonds to carbon atoms
Metabolic water source = high ratio of hydrogen to oxygen atoms, because they release water if they are oxidised
Insoluble = wont affect water potential and osmosis
Low in mass = a lot can be stored in animal without preventing movement
Describe properties of phospholipids
Hydrophilic head = attract water as it is charged
Phosphate is charged, so repels other fts
Hydrophobic tail = repels with water, mixes with fats as the fatty acid chain is not charged
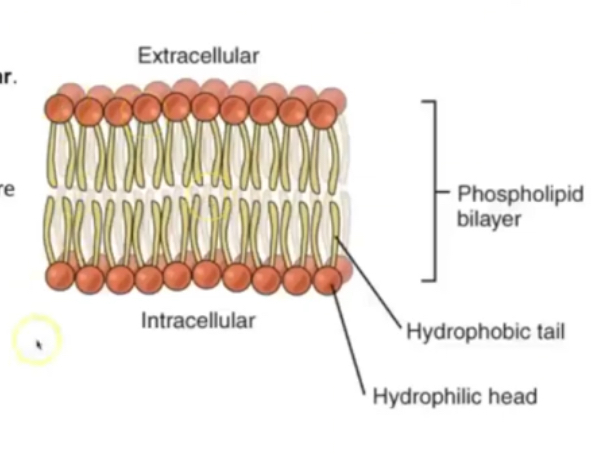
Descibe the phospholipid bilayer
Have 2 charged regions, so are polar
\in water they position so heads are exposed to water and tails are not
Makes up plasma membrane in cells
Describe cholesterol structure and function
Sterol = 4 carbon rings and hydroxyl group at one end, have hydrophobic and hydrophilic regions
Cholesterol embedded within cell membranes to reduce fluidity of membrane at high temperature, and increase fluidity at low temps
Draw amino acid general structure
Amine group = H N H
Carboxyl group = COOH
Hydrogen
R group - variable group
Define the four levels of structure in a protein
Primary
Secondary
Tertiary
Quaternary
Primary structure:
Order of amino acids in polypeptide chain (polymer)
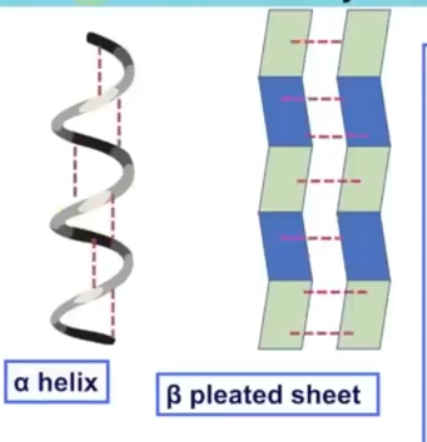
Secondary structure
Sequence of amino acids cause parts of protein molecule to behind into alpha helix or beta pleated sheets
These are held by hydrogen bonds (that form between the C=O of carboxyl and H of amine group)
Tertiary structure
The secondary structure is bent and folded to form a precise 3D shape, held by:
Hydrophobic and hydrophilic interactions (weak)
Hydrogen bonds (weak)
Ionic bonds (stronger, form between R groups)
Disulfide bonds = (strong covalent bonds between R groups of amino acids that contain sulcus)
Quaternary structure + example of conjugated protein
Protein made up of more than one polypeptide chain
Example:
Haemoglobin made of four polypeptide chains
Has prosthetic group (containing iron) attached to polypeptide chain = conjugated protein, which men’s has a non protein group added rto it