A&P CH2 PG10
1/55
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
56 Terms
What are chemical bonds?
Forces that hold atoms together to form compounds
Why do elements connect by bonding?
So that they become more stable and to form molecules needed for life.
What are two main types of bonds ?
Ionic bonds
Covalent bonds
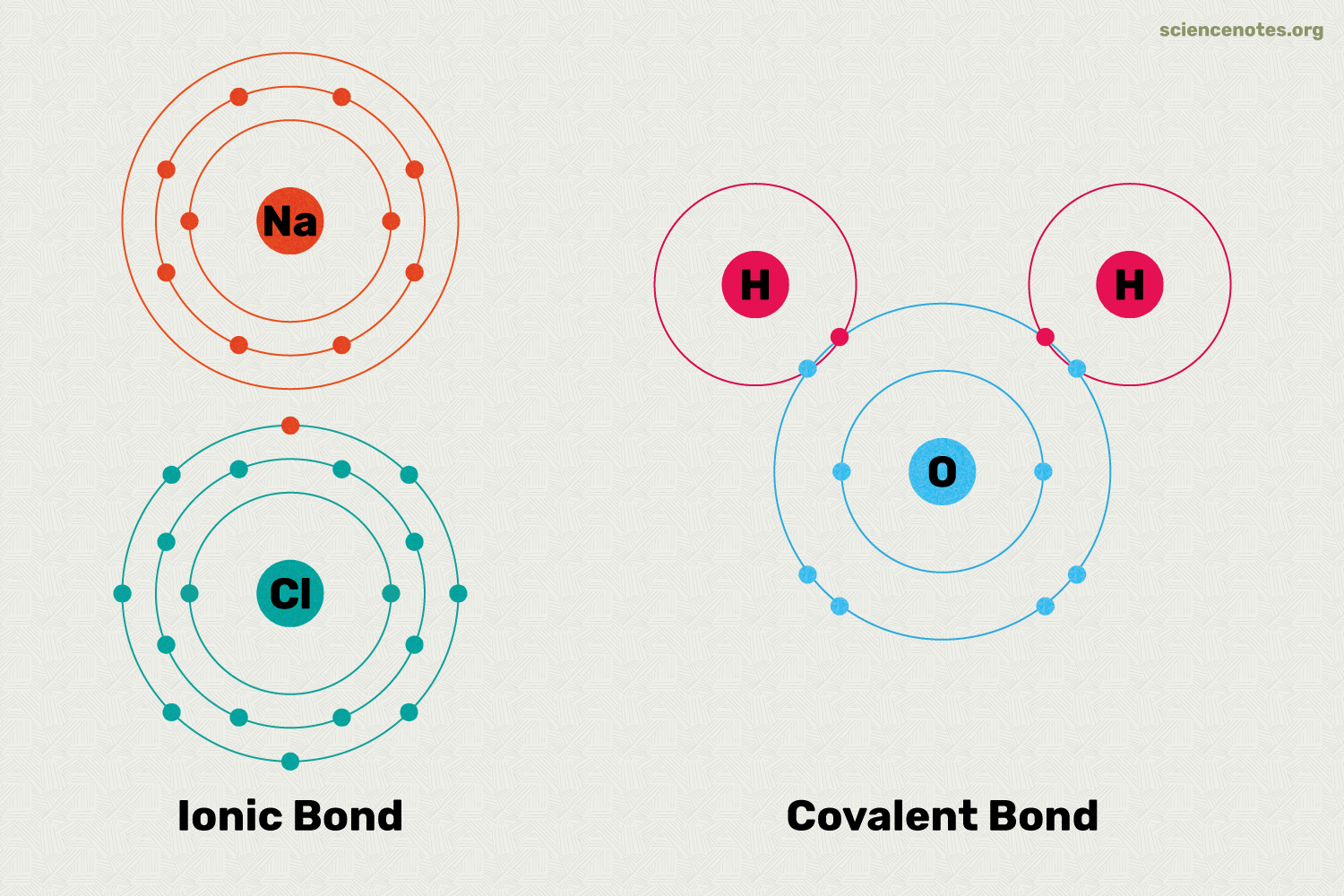
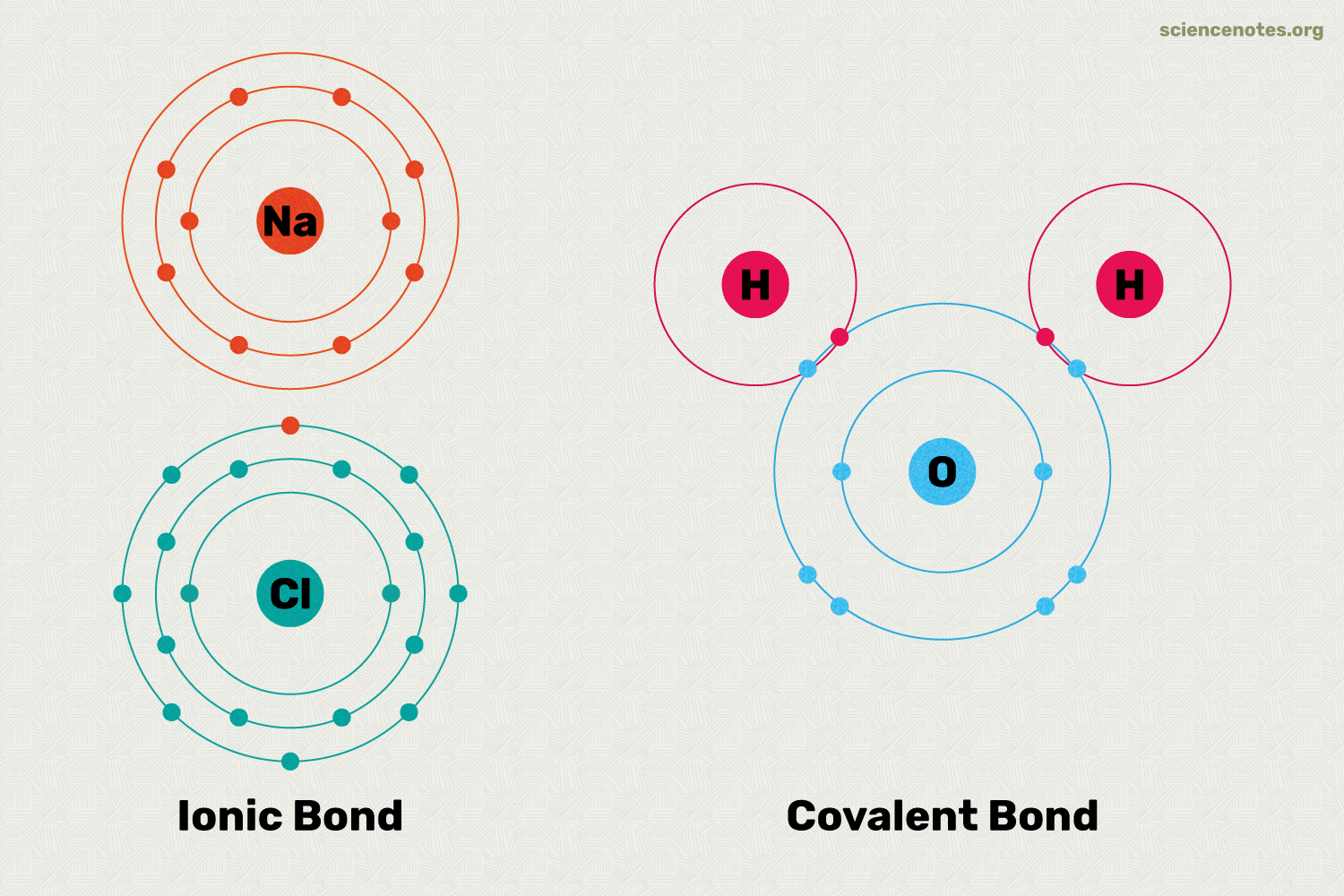
What is an ionic bond?
The attraction between positive and negative ions.
T/F: ionic compounds are called salts.
True
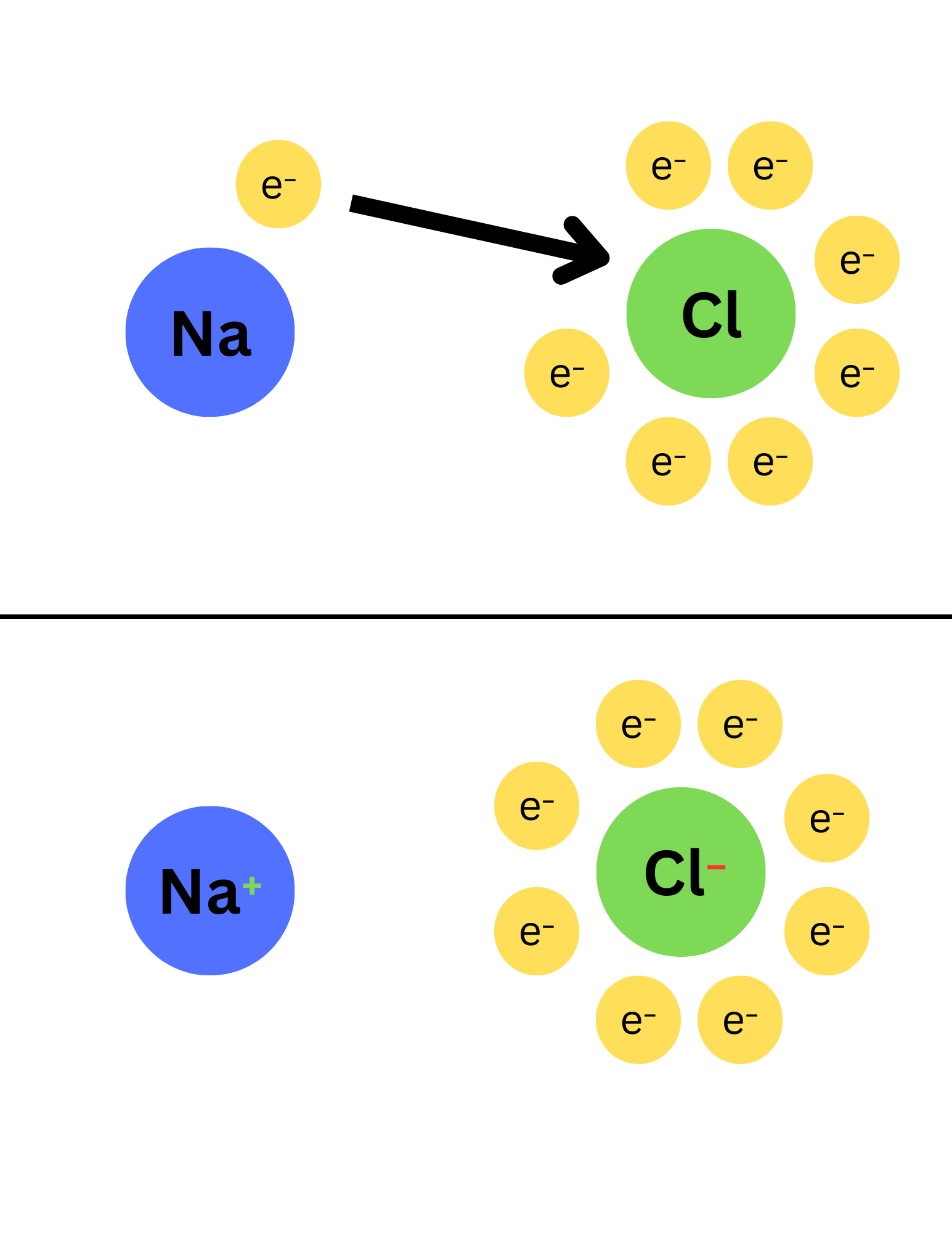
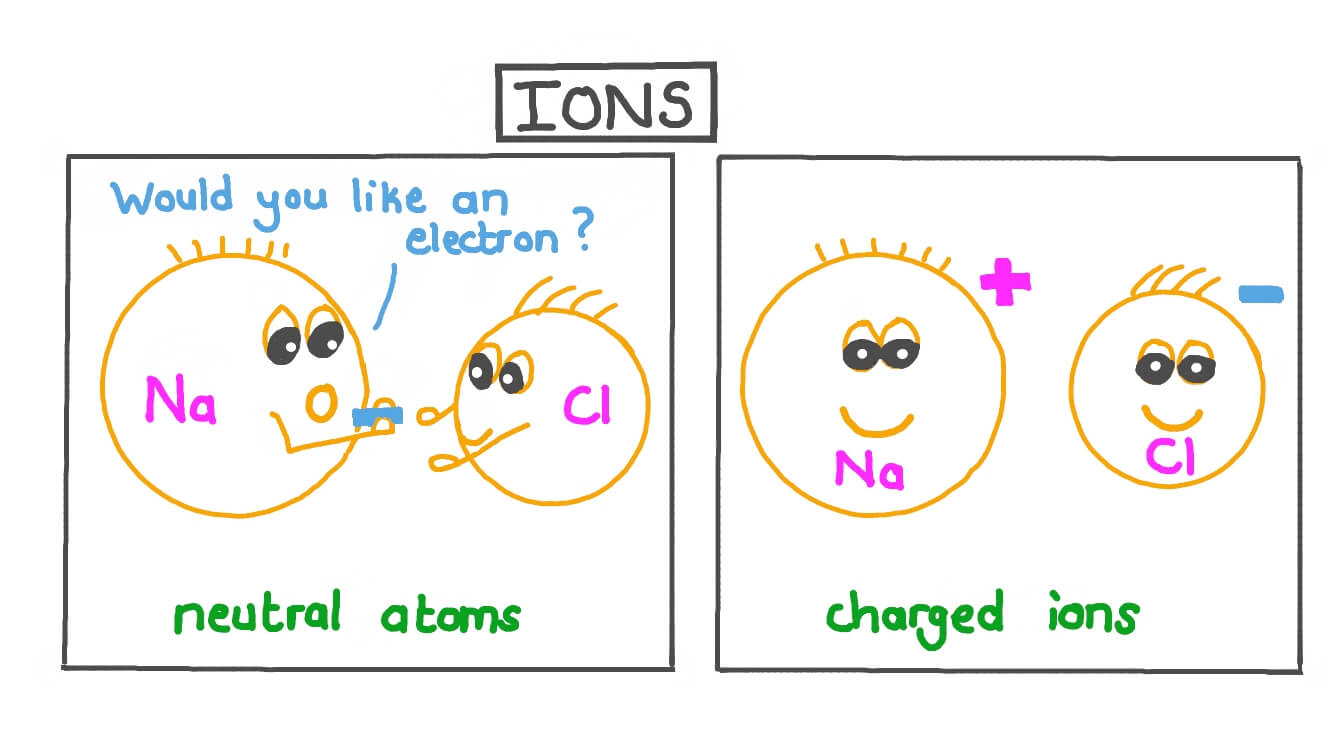
What is an ion ?
An atom that has gained or lost electrons.
What are electrons?
Negatively charged subatomic particles
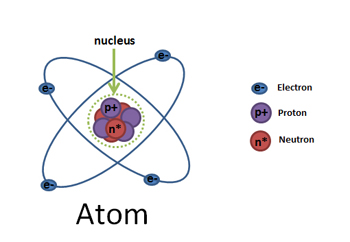
T/F: atoms become ions by gaining or losing electrons.
True
What are the two types of ions?
Cation
Anion
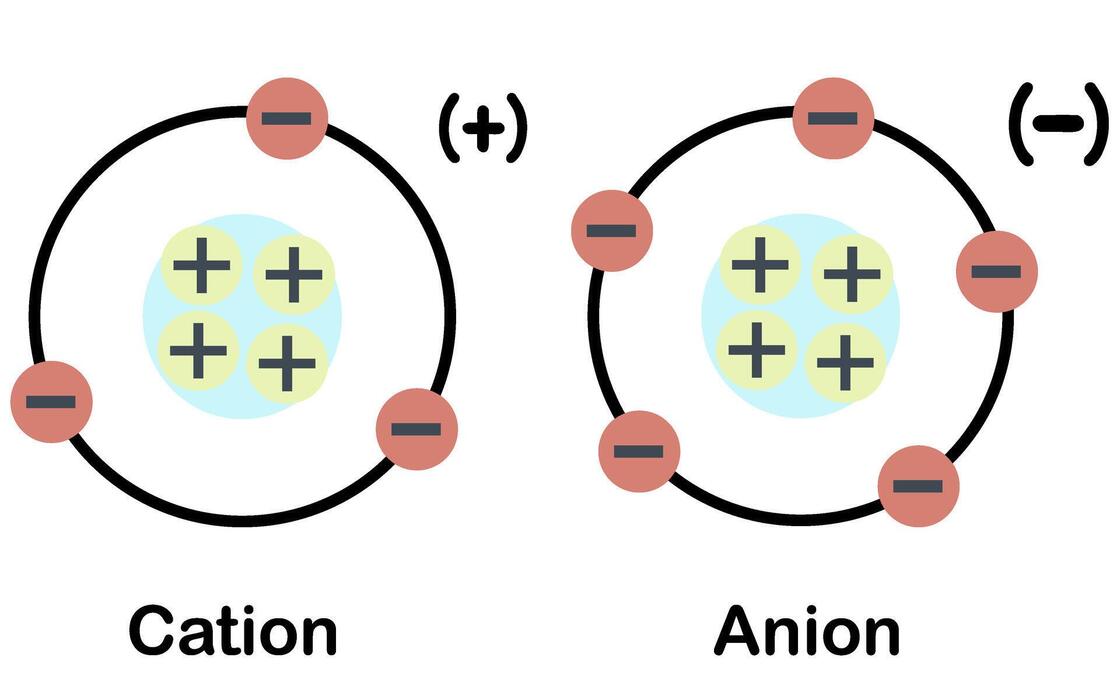

What is a cation?
Positively charged ion
Paw-sitive

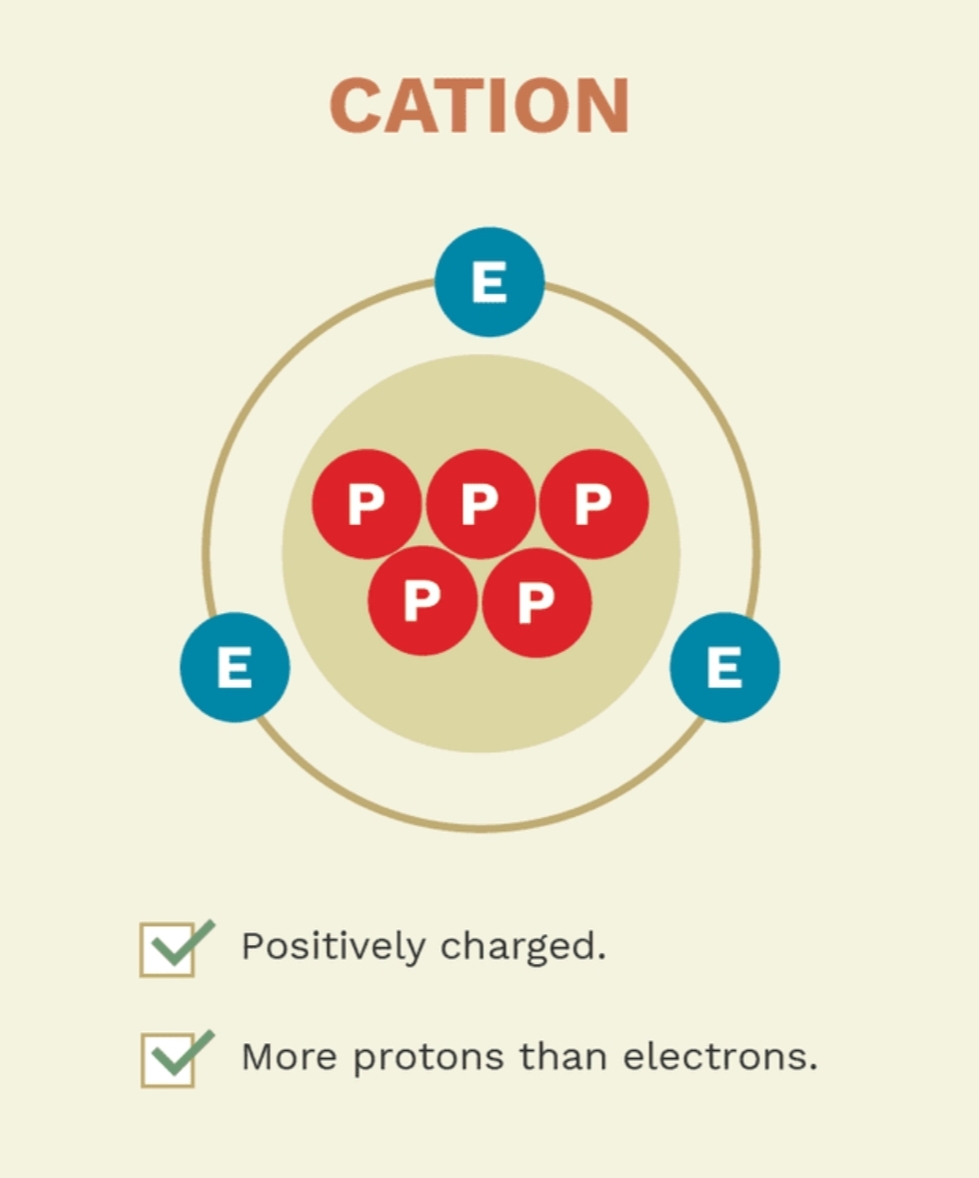
How is a cation formed?
Formed when an atom loses electrons
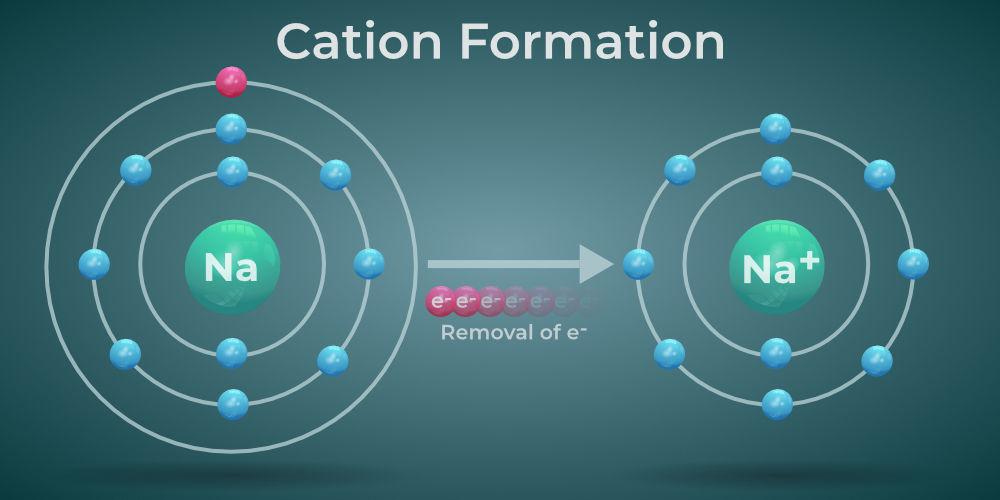
What are examples of cations?
Sodium ion (Na+)
Calcium ion (Ca²+)
Potassium ion (K+)
Losing negative electrons makes the atom:
Postive
What is an anion?
Negatively charged ion
How is an anion formed?
Formed when an atom gains electrons
What is an example of an anion
Chloride ion (Cl–)
Fluoride ion (F⁻)
Gaining negative electrons makes the atom:
Negative
Why do ionic bonds form?
Because opposite charges attract.
Postive cations are attracted to negative anions
What happens when opposite charges bond?
Charges balance out so the compound has no net charge
What is an ionic compounds?
When cations and anions bond
What are examples of Ionic Compounds? Name 2.
Sodium Chloride [NaCl]
Potassium Chloride [KCl]
Calcium Phosphate [Ca3(PO4)2]
![<p>Sodium Chloride [NaCl]</p><p class="has-focus">Potassium Chloride [KCl]</p><p class="has-focus">Calcium Phosphate [Ca3(PO4)2]</p><p class="has-focus"></p>](https://knowt-user-attachments.s3.amazonaws.com/210164b7-6dbe-42ff-8ef1-563cf3f837cf.jpg)
Ionic Bond Review:
Involves electron transfer
Produces ions
Bonds are based on charge attraction
What are Covalent Bonds?
Forms when two atoms share electrons
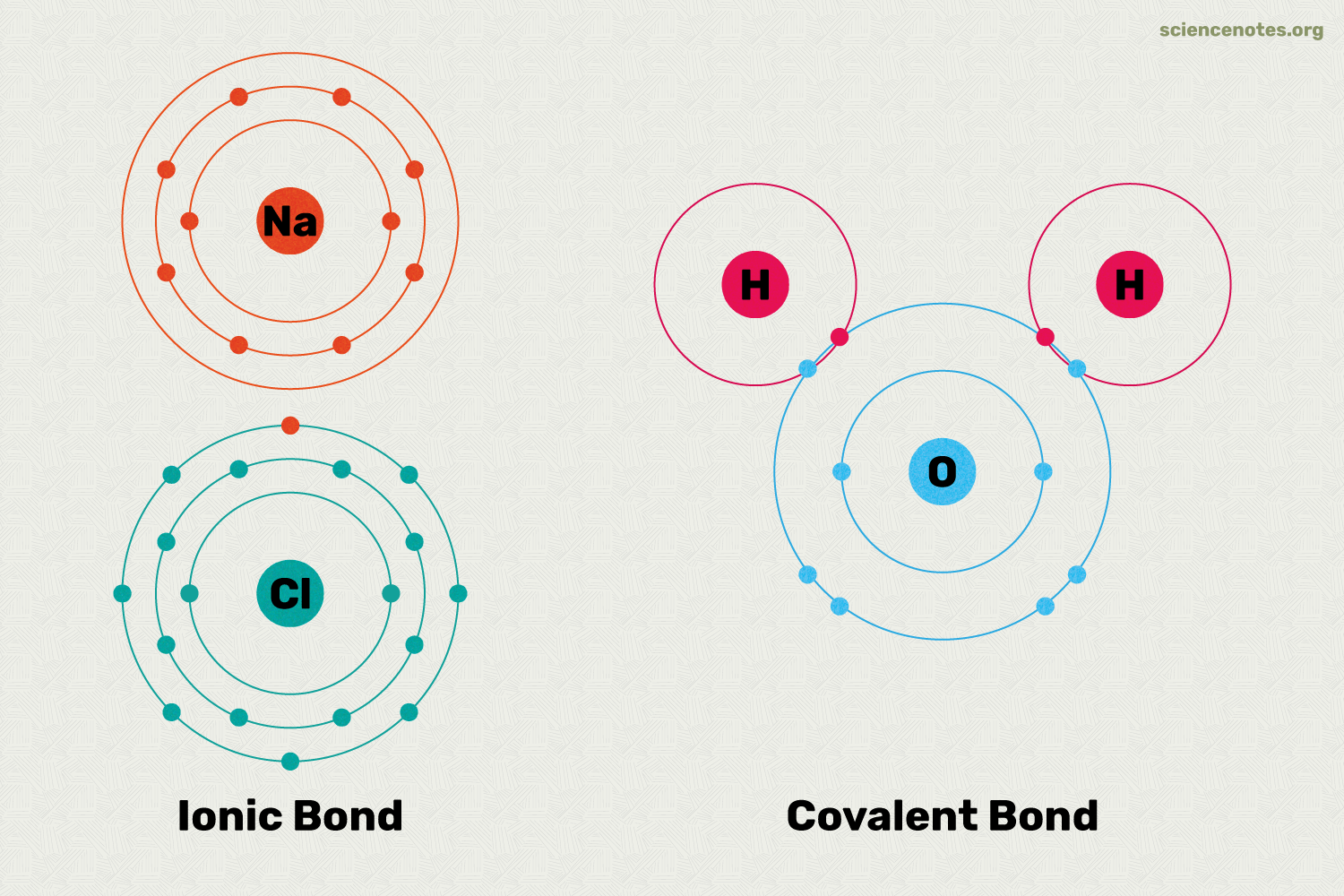
T/F: Covalent bonds form molecules, not salts.
True
Ionic bonds form salts.
What are the two types of Covalent bonds?
Nonpolar
Polar
What are Nonpolar Covalent Bonds?
Atoms share electrons equally. Neither atom has a stronger pull on electrons
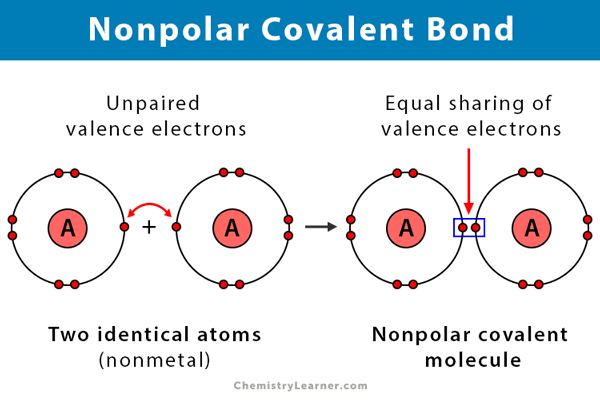
When do you know Nonpolar bonds occur?
When two atoms are:
The same element
Have very similar electronegativity
What are examples of Nonpolar Bonds? Name three.
Hydrogen Gas (H2)
Nitrogen Gas (N2)
Oxygen Gas (O2)
Chlorine Gas (Cl2)

Why is Oxygen Gas (O2) Nonpolar?
Oxygen bonded to Oxygen (O2) means both atoms pull equally.
Notice the symmetry.
What are C—H Bonds?
C-H bonds (carbon-hydrogen bonds) are covalent chemical bonds that connect a carbon (C) atom to a hydrogen (H) atom.
Why is it important to remember C—H Bonds?
The Carbon—Hydrogen (C—H) bond is fairly Nonpolar
Molecules with many C—H bonds are usually nonpolar
What are examples of C–H bonds that are Nonpolar?
Lipids (fats)
Triglycerides
Phospholipids
Fatty Acids
Why are Nonpolar molecules important to remember?
They do not dissolve well in water
They form membranes and insulation (fats)
What are Polar Covalent Bonds?
Atoms share electrons unequally. One atom pulls electrons more strongly.
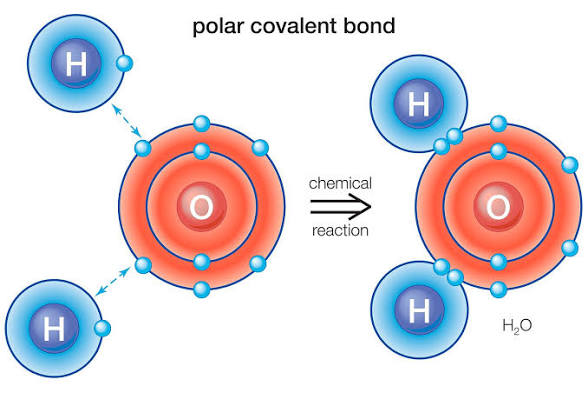
What is Electronegativity?
An atoms ability to attract electrons
T/F: if one atom is more electronegative, the bond is polar.
True, the bond does not have an equal pull, therefore making it a polar bond.
T/F: The electrons spend more time near the more electronegative atom
True
What are Examples of common Highly Electronegative Elements? Name 2.
Oxygen (O)
Nitrogen (N)
Fluorine (F)
Chlorine (Cl)
(O) or (N), 2 common Highly Electronegative Elements, pair with which atoms (name 2), to create polar bonds.
Oxygen or Nitrogen pairs with ____ or ____ to create polar bonds.
Hydrogen (H) or
Carbon (C)
What are examples of Polar Bonds?
O-H (water)
N-H (protein)
C-O
-Slight negative charge near O or N
-Slight positive charge near H
Why do Polar Bonds matter?
It allows Hydrogen binding which enabled
Water’s unique property
Protein folding
Enzyme activity
Transport of substances
Ionic vs Covalent Bonds?:
Electrons transferred
Ionic
Ionic vs Covalent Bonds?:
Forms molecules
Covalent
Ionic vs Covalent Bonds?:
Partial or no charge
Covalent
Ionic vs Covalent Bonds?:
Electrons shared
Covalent
Ionic vs Covalent Bonds?:
Forms ions
Ionic
Ionic vs Covalent Bonds?:
NaCl
Ionic
Ionic vs Covalent Bonds?:
Weak in water
Ionic
Ionic vs Covalent Bonds?:
H2O
Covalent
Quick memory cues:
Polar
Polar
Unequal sharing
O & N involved
Quick memory cues:
Nonpolar
Nonpolar
Equal sharing
Same elements
Quick memory cues:
Covalent
Covalent
Sharing electrons
Molecules
Quick memory cues:
Ionic
Ionic
Ions
Cation/anions
NaCl
T/F: covalent bonds create ions
False. That's ionic bonds
T/F: C—H is mostly nonpolar
True.
What's the difference between polar bonds and ionic bonds?
polar bonds involve unequal sharing of electrons,
while ionic bonds involve a complete transfer of electrons