Biochem Exam 2 - Protein function
1/49
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
50 Terms
what are all the functions of proteins?
transport, structural, motor function, catalysis, immunity, regulation of gene expression
function of myoglobin
transport o2 throughout muscles
function of hemoglobin
transport o2 through blood
function of actin
forms myofilaments
function of tubulin
forms microtubules
function of keratin
forms intermediate filaments found in hair, nails, and skin
function of collagen
major protein in connective tissue
function of myosin
interacts with actin to facilitate muscle movement
function of kinesin
moves along microtubules to help transport cellular cargo
explain catalysis
enzymes accelerate chemical reactions
what is a heme
o2 binding component of myoglobin and hemoglobin
prosthetic group - specifically porphyrin
what is a prosthetic group
organic molecules bound to a protein that aids in protein function
what is the structure of heme
a porphyrin ring with iron in the center
iron is chelated by four nitrogen atoms
how many coordinate bonds does iron form?
6 total
4 nitrogen
1 o2
1 histidine residue
what is the function of His residues on a heme
stabilize the heme-o2 interaction
F8 his = coordinates directly with iron
E7 his = helps stabilize the bound o2 molecule
what is anemia and how is it treated?
iron deficiency
treated with iron supplements or iron rich diet
how can binding of o2 to myoglobin be described?
Mb + o2 = Mbo2
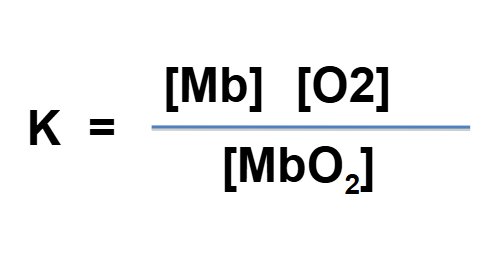
what does K stand for in this equation
dissociation constant = measure of how easily mbo2 falls apart and the po2 at which half the myoglobin is saturated
low k = strong bonding
high k = weak bonding
what does the myoglobin and o2 graph represent
hyperbolic trend
what happens when the oxygen conc and dissociation constant are equal?
binding is half maximal
what percentage are Mb and Hb identical in their primary sequence?
18%
which structure level are Mb and Hb similar?
secondary and tertiary
what does the similarity in structure and sequence between Mb and Hb indicate?
common evolutionary origin
how many heme groups can Hb and Mb bind to?
Hb = 4
Mb = 1
what is cooperativity?
when one subunit binds to o2 it triggers a conformational change that inc the affinity for the next
what type of graph data indicates cooperativity
sigmoidal
how is the deoxy (T) state represented
HbH+ + o2
how is the oxy (R) state represented
Hbo2 + H+
what is the relationship between ph and o2 affinity
as ph inc, o2 affinity inc
what is BPG
modulates Hb ability to bind to o2
negatively charged
only binds to Hb when in t/deoxy state
dec Hb’s o2 affinity which releases more o2 to tissues
at high altitudes does BPG inc or dec in people
inc to prevent hypoxia
What would be the expected result of a Lys residue being replaced by a Ser residue in the BPG binding site of hemoglobin?
BPG would bind less tightly because of the loss of a positive charge
In the ____ form of Hb, the iron atom is out of the plane of the porphyrin ring.
tense state
Deoxyhemoglobin is unfavorable to oxygen binding because it is in the _____ conformation
T
What statement regarding the fractional saturation of myoglobin is FALSE?
It increases linearly at all oxygen concentrations.
A plot of the binding of oxygen to myoglobin as a function of pO2 gives a _____ shape; a similar plot for hemoglobin gives a _____ shape.
hyperbolic; sigmoidal
How does CO2 affect hemoglobin-oxygen binding?
CO2 is converted to bicarbonate and H+ which promotes the deoxy state
explain the residue change in sickle cells
glutamate is replaced with valine
glutamate is neg charged while valine is nonpolar
explain sickle cell rbcs
less flexible so more likely to rupture
harder to pass through small capillaries can cause blockages
deoxygenated Hb becomes insoluble and forms polymers that aggregate
describe the structure of actin
globular actin subunits associate in a double chain to form microfilaments
microfilaments have a a pos and neg end
pos end has faster addition
what is treadmilling
when the rate of addition to one end matches the rate of removal at other end
describe the alpha tubulin
guanine nucleotide is buried and inaccessible
describe beta-tubulin
nucleotide is more exposed and can be hydrolyzed
describe the microtubule structure
alpha and beta tubulin join to create a heterodimer which is the fundamental unit of the microtubule which is hollow
how is the tubular structure able to be seen
cryoelectron microscopy
what drugs affect microtubules
colchicine = causes microtubules to depolarize
paclitaxel = prevents microtubule depolymerization
what is the structure of keratin
forms a coiled-coil structure made from alpha helices
what is the structure of collagen
made from 3 left handed gly-rich helical polypeptides that form a triple helix
covalently cross linked which stabilizes the structure
every 3rd aa is gly
30% of remaining aa are pro or hydroxyproline
what is the structure of myosin
two heads and a long tail
describe the sequence of events in the myosin-actin cycle
ATP binds to myosin, myosin release from actin, myosin binds to another actin subunit, release of Pi and ADP, stretched myosin returns to original conformation