Module 2 Bio
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
4 main macromolecules
carbohydrates, lipids, proteins, nucleic acids
Monosaccharides
monomers that make of carbohydrates
Disaccharide
2 monosaccharides joined together by a covalent bond
carbohydrate function
energy and structure
glucose
useful source of energy for cells, ring-like structure, and has MANY isomers
Monosaccharides are joined by
glycosidic linkages
Glycosidic Linkage (linear)
attached by 1,4 g-bonds
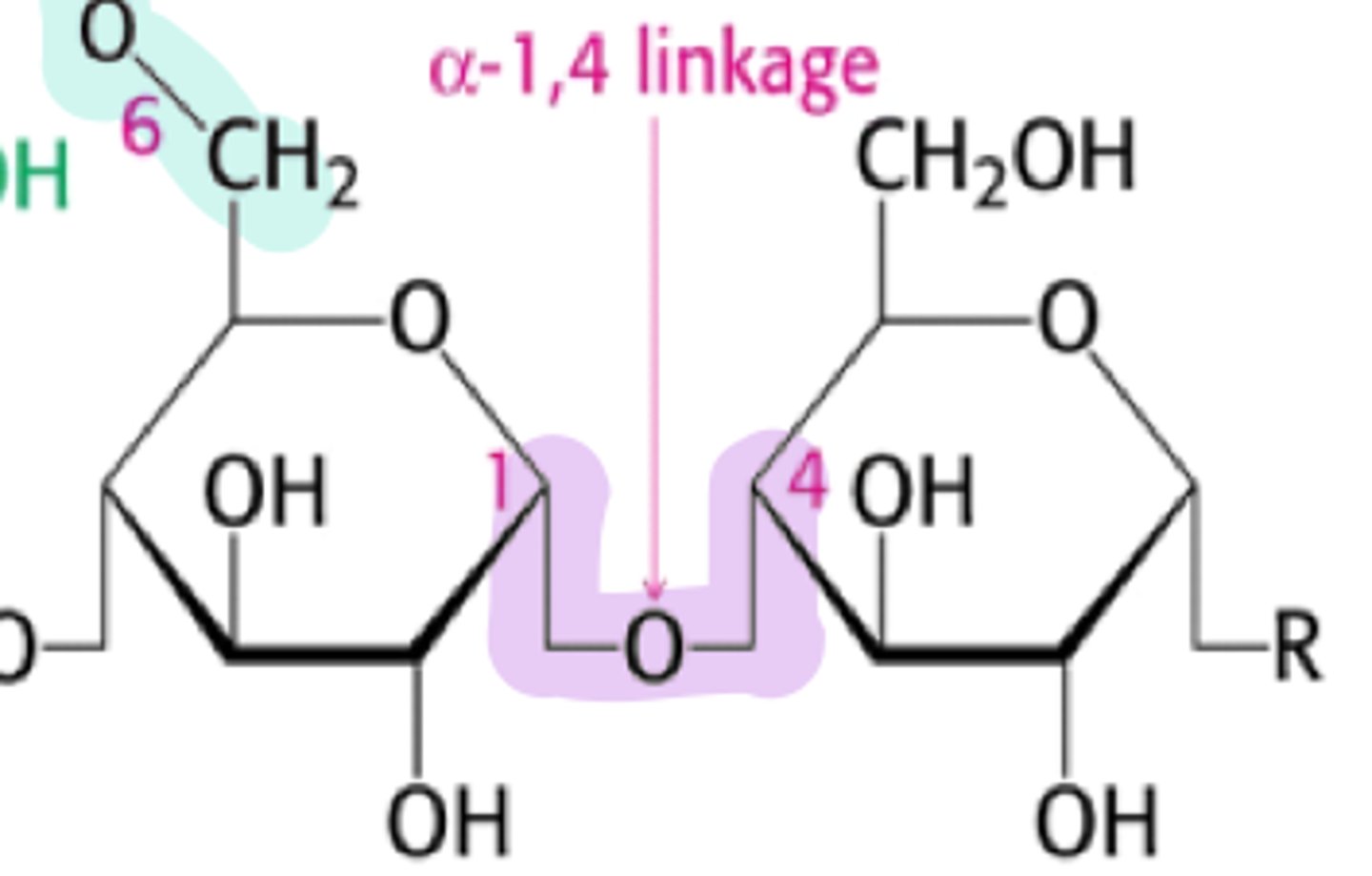
glycosidic linkage (branched)
attached by the 1,6 g-bonds
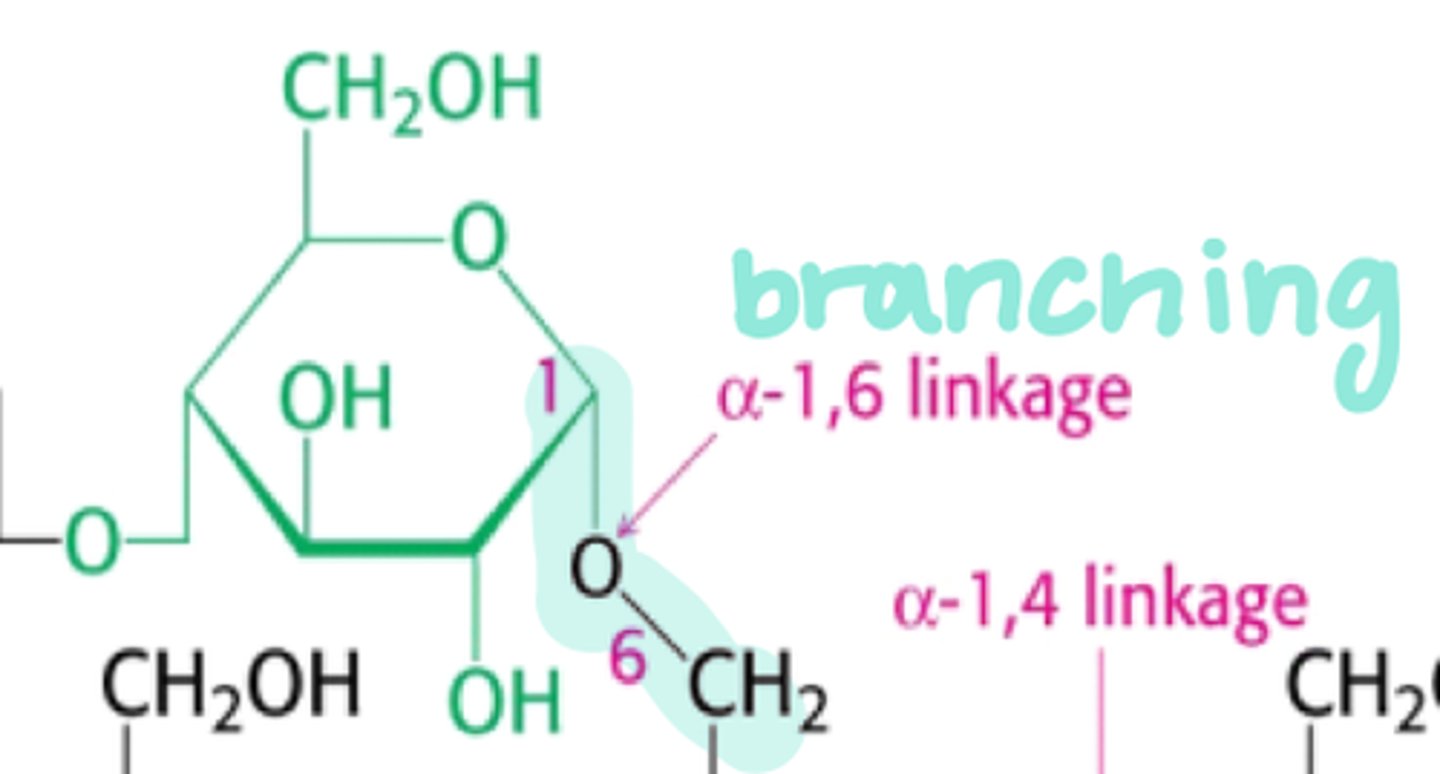
why are glycogen and starch branched?
so it can be broken down to glucose
chitin
VERY strong exoskeleton of animals
are lipida polymers?
NOOOOOOO
function of lipids
long term energy storage, pigments in plants, structural component of steroids, insulation, waxy coating, cell membrane
4 major lipids
fatty acids, triglycerides, phospholipids, steroids
fatty acid
long hydrocarbon
are lipids polar or nonpolar
both!
amphipathic
both polar and nonpolar
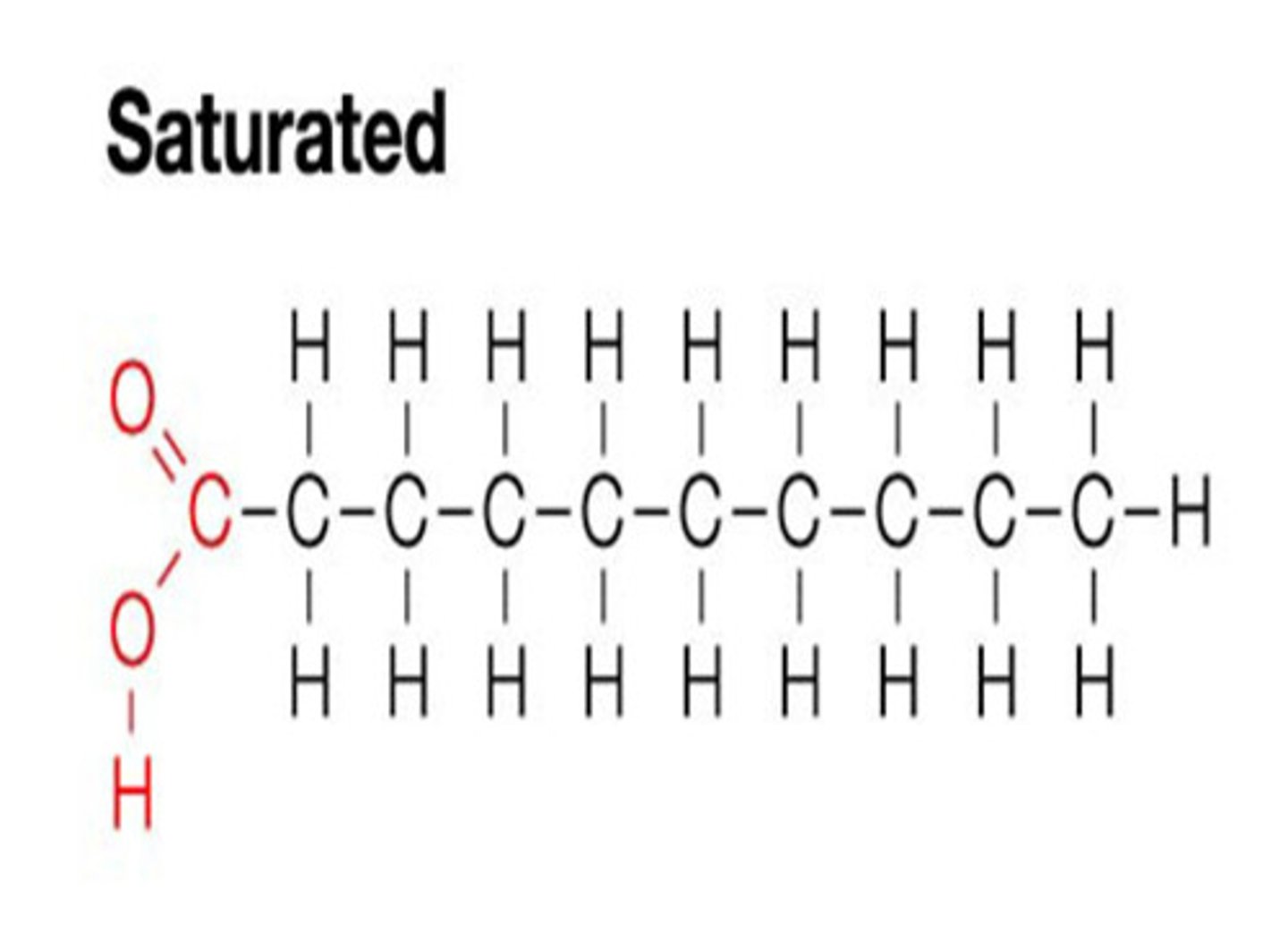
saturated fat
bonded by ONLY single bonds and creating a tight bond
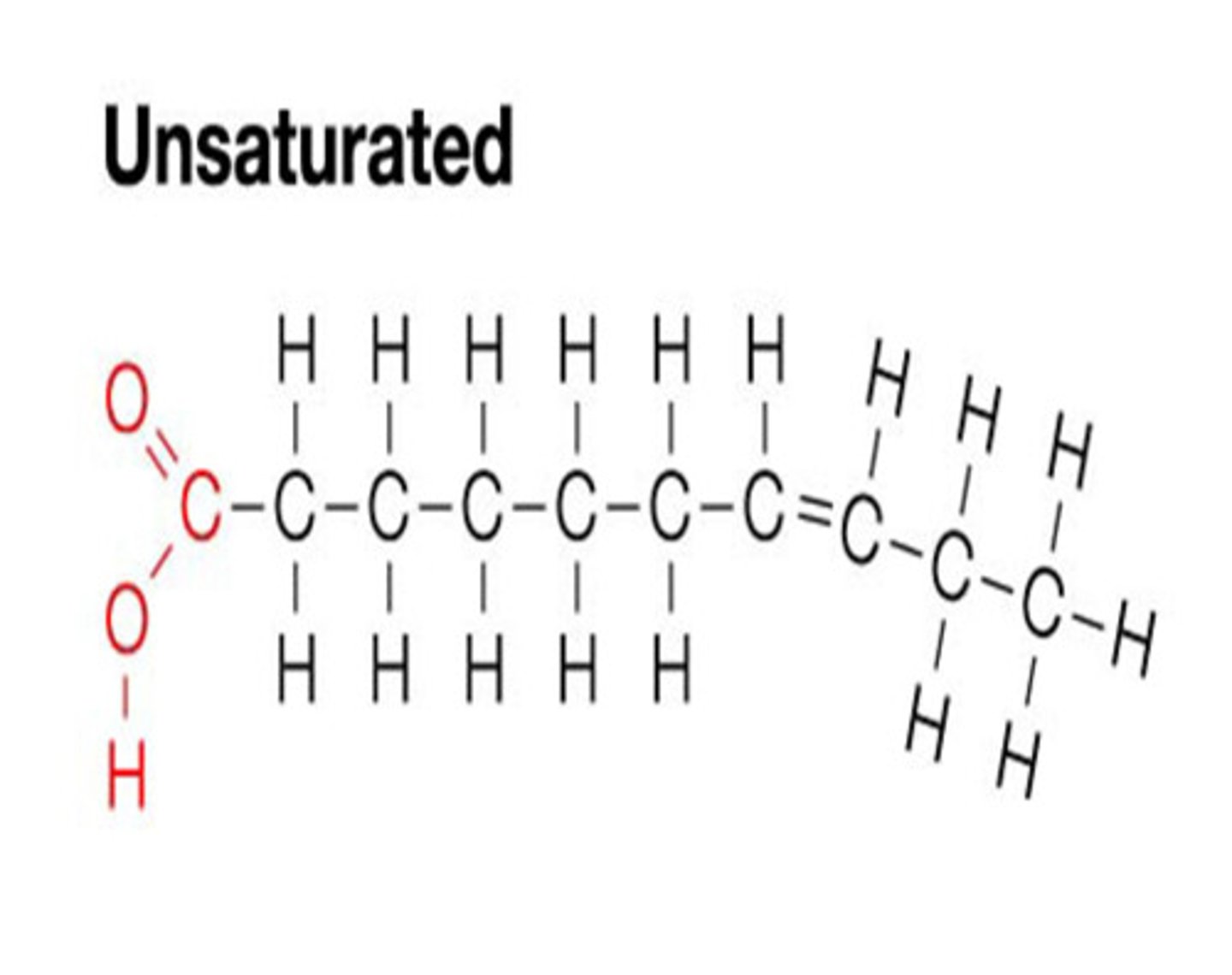
unsaturated fat
bonded by single AND double bonds which causes KINKS AND BENDS
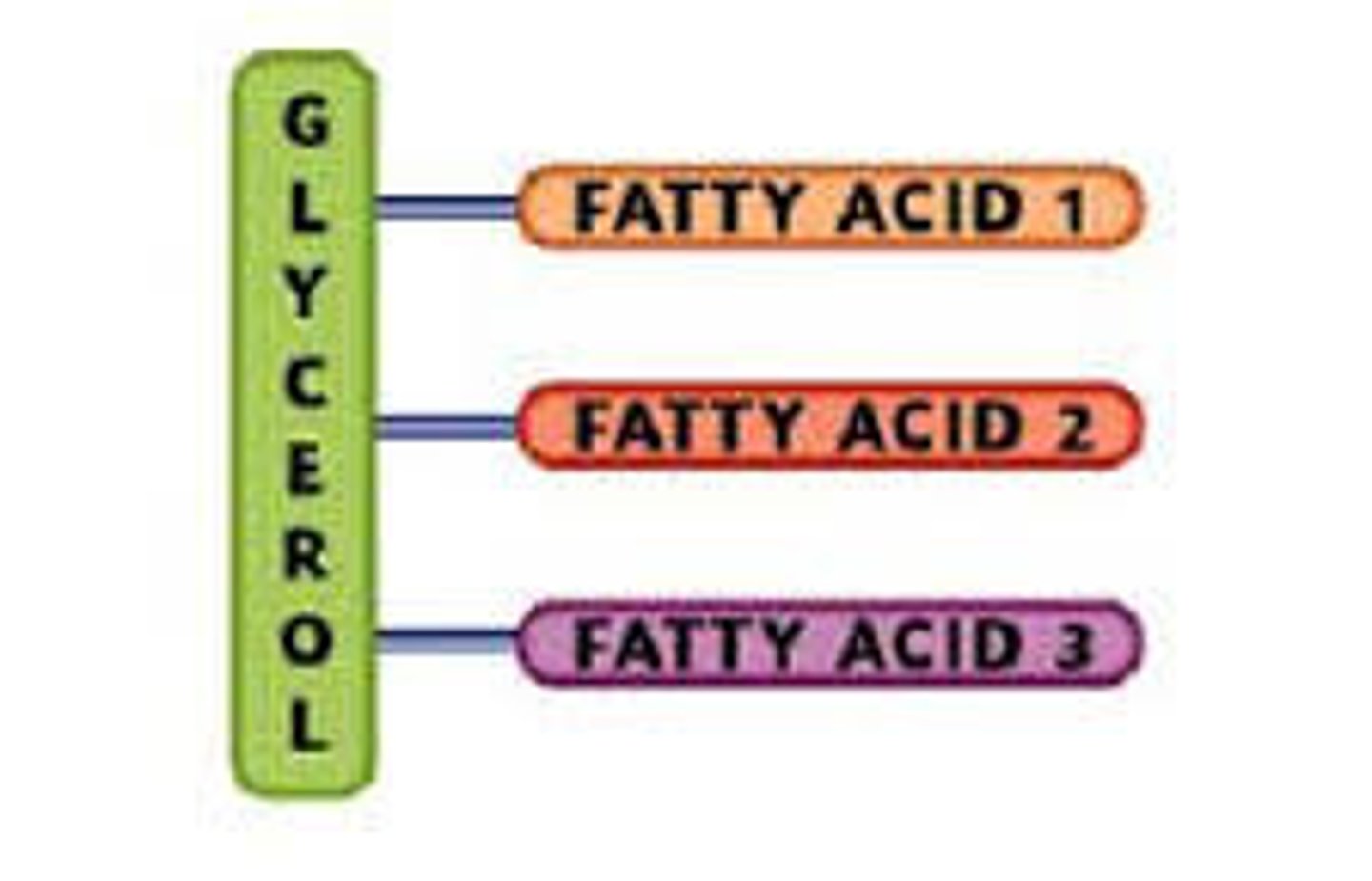
Triglycerides
3 fatty acids that are attached to a molecule called glycerol
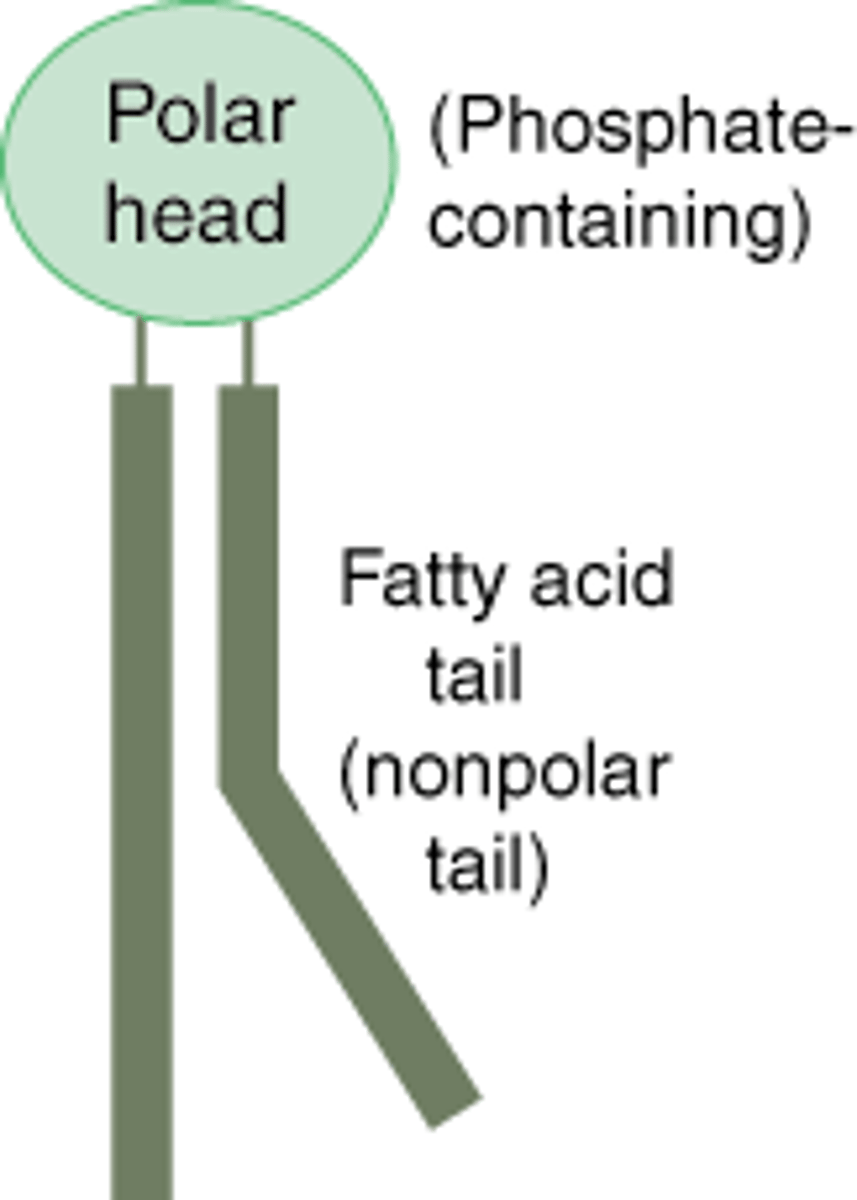
phosolipid
very similar structure to a triglyceride, but a charged phosphate molecule replaces one fatty acid
steroid
hormones
proteins are made up of
amino acids
what makes up an amino acid
amino group, carboxyl group, carbon central atom, and a R group
peptides
short polymers of amino acids
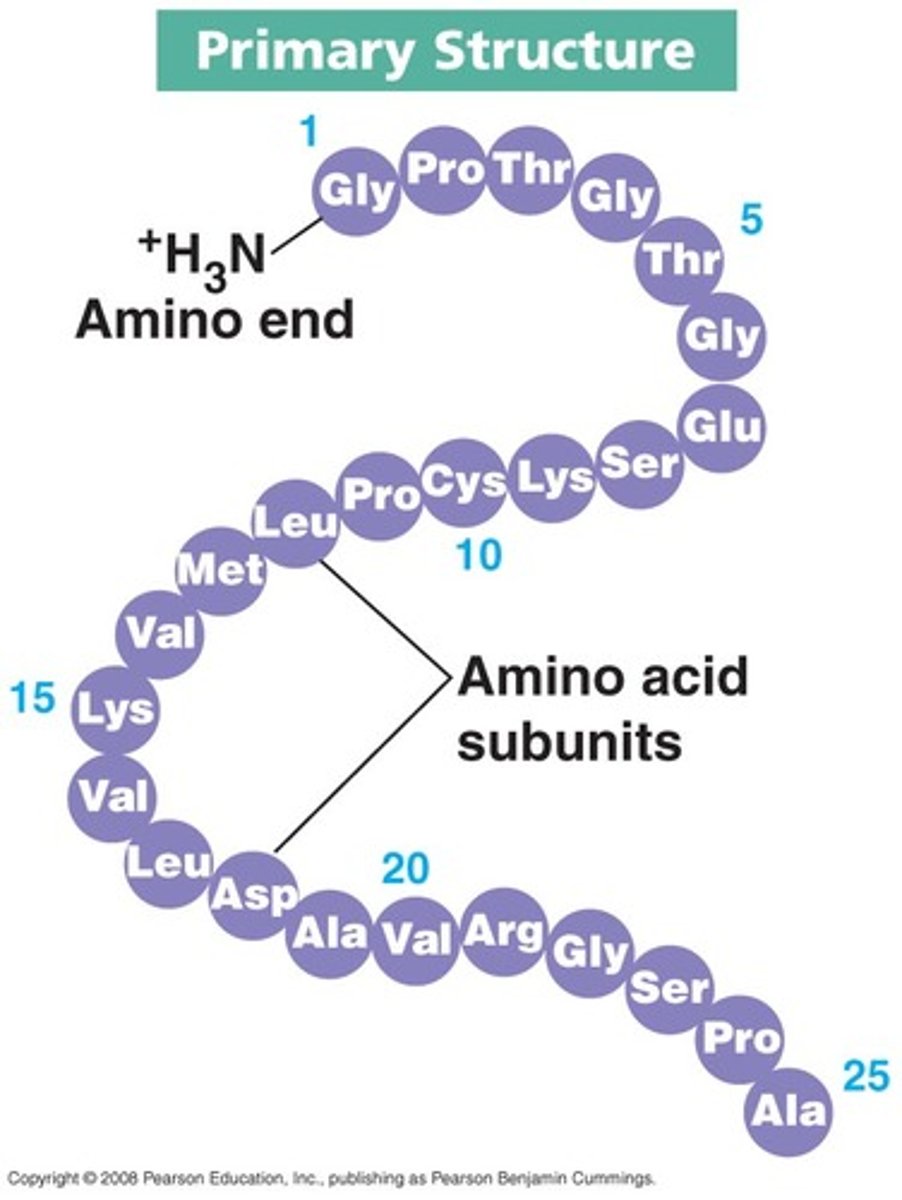
primary structure
sequence of amino acids formed by peptide bonds (polar)
secondary structure
in a primary structure there is hydrogen. hydrogen create dipoles that can react to other primary structures and form 2ndary structures
2 types of secondary structure
alpha helix and beta pleated sheet
tertiary structure
a bunch of 2ndary structures connected together
formed interactions between R-groups
disulfide bonds, hydrogen bonds, ionic bonds, van der waals interactions
quarternary structure
formed between many tertiary bonds
Denaturing
breaking down quart, and tertiary w/ out breaking peptide
Importance of Denaturation
important in changing pH, making things poplar/nonpolar, temp changes
nucleic acids are made up of
nucleotides
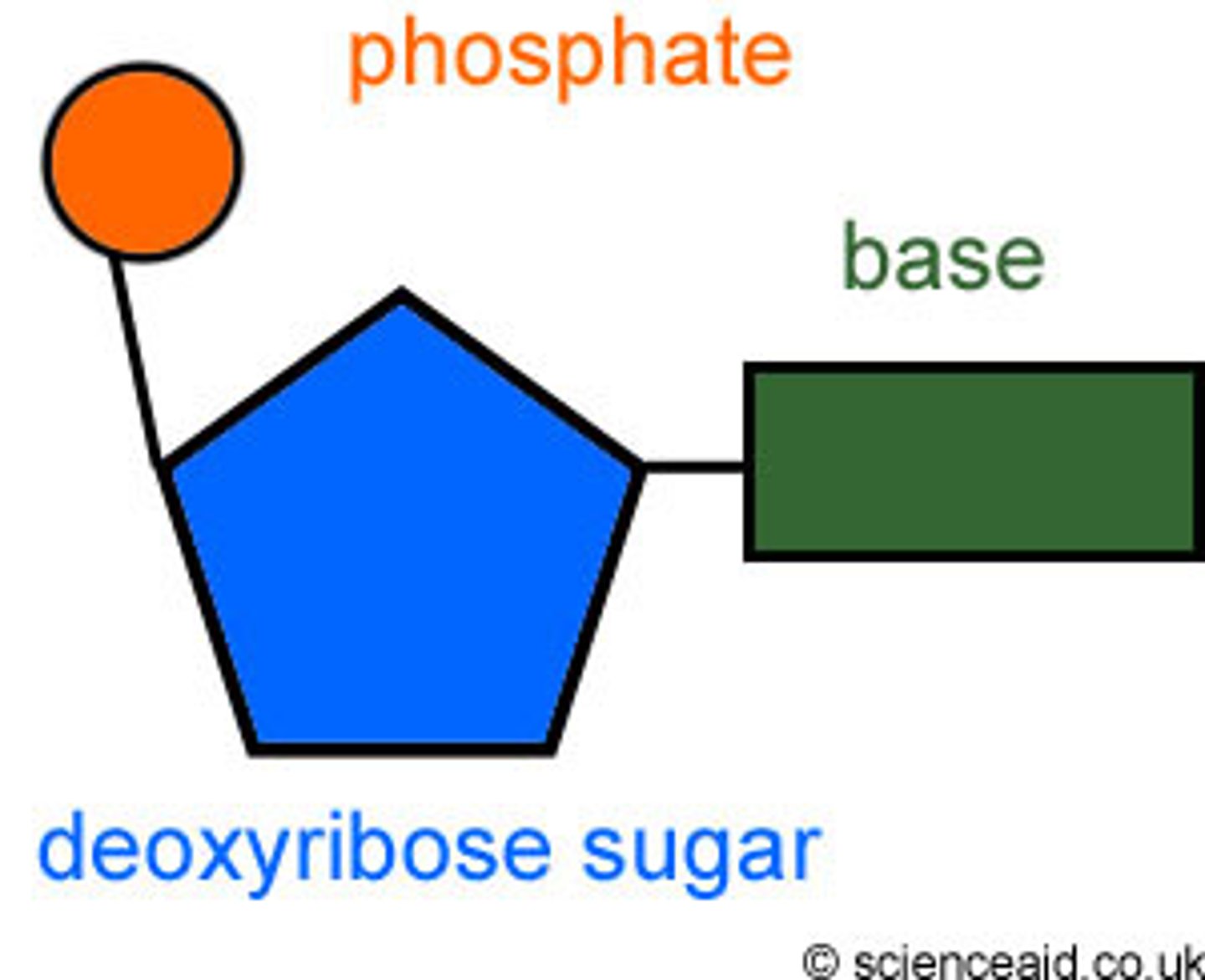
structure of a nucleotide
phosphate group, sugar, nitrogenous base
Pryimidine Bases
single rings, cytosine, thymine, and uracil
purines
double rings, adenine, and guanine
phosphate backbone
link between 5 carbons and 3 carbon and deoxyribose
Antiparrell
two sides run in opposite directions
- atoms arranged in opposite structure
Adenine
Thymine(held by 2 H-bonds)
Cytosine
Guanine(held by 3 H-bonds)