Molec & Cell Exam 3
1/111
Earn XP
Description and Tags
Notes from lecture
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
112 Terms
Why do we care about cell signaling? (4 examples)
Type I diabetes, pancreatic cells that produce insulin are lost → without insulin signal sugar accumulates leading to dangerous effects
Multiple sclerosis - degeneration of the myelin sheath, affected nerve cells cant transmit signals from one area of the brain to another
If cell communication breaks down in the processes of cell growth and division, uncontrolled cell growth can lead to cancer
Stroke is when blockage causes surrounding tissue to die - dying cells release a lot of glutamate (excitotoxicity) and spreads through the brain killing parts that were not affected leading to widespread brain damage
Cell signaling can occur by (2)
Direct interaction with neighboring cells
Secreting signaling molecules
Direct cell-cell interactions
Critical for regulating the behavior of cells in animal tissues.
Integrins and cadherins (both adhesion and signaling molecules)
Cells express cell surface receptors that interact with neighboring cells that have signaling molecules on their surface.
Important for embryonic development, maintenance of adult tissues
Categories of signaling by secreted molecules (3)
Endocrine
Paracrine
Autocrine
Endocrine signaling
Hormones (signaling molecules) are secreted by endocrine cells and travel through the circulatory system, acting on target cells at distant sites in the body
ex. Estrogen is produced by the ovaries and stimulates development and maintenance of female reproductive system and sex characteristics
In animals, 50 different hormones are produced by the endocrine glands (pituitary, thyroid, pancreas, adrenal, gonads)
Paracrine signaling
Signaling molecules act locally to effect behavior of neighboring cells. Molecule released by one cell acts on neighboring target cells
ex. neurotransmitters
Autocrine signaling
Cells respond to signaling molecules that they themselves produce.
ex. immune response: T-lymphocytes respond to antigenic stimulation by synthesizing growth factor. Drives its own proliferation. Increases the number of responsive T-lymphocytes, amplifies immune response.
Abnormal autocrine signaling leads to cancer
Steroid hormones
Hormones secreted by the adrenal cortex. Some receptors are intracellular proteins located in the cytosol or the nucleus. Signaling molecules are small hydrophobic and able to diffuse across the plasma membrane to directly reach these receptors (rather than a secondary messenger)
i. Sex steroids: Testosterone, progesterone, estrogen
ii. Glucocorticoids: stimulate production of glucose (adrenal gland)
Nuclear Receptor Family
Transcription factors that contain relation domains for ligand binding, DNA binding and transcriptional activation.
Ligand binding regulates their function as activators or repressors of their target genes → signaling molecules directly regulate gene expression
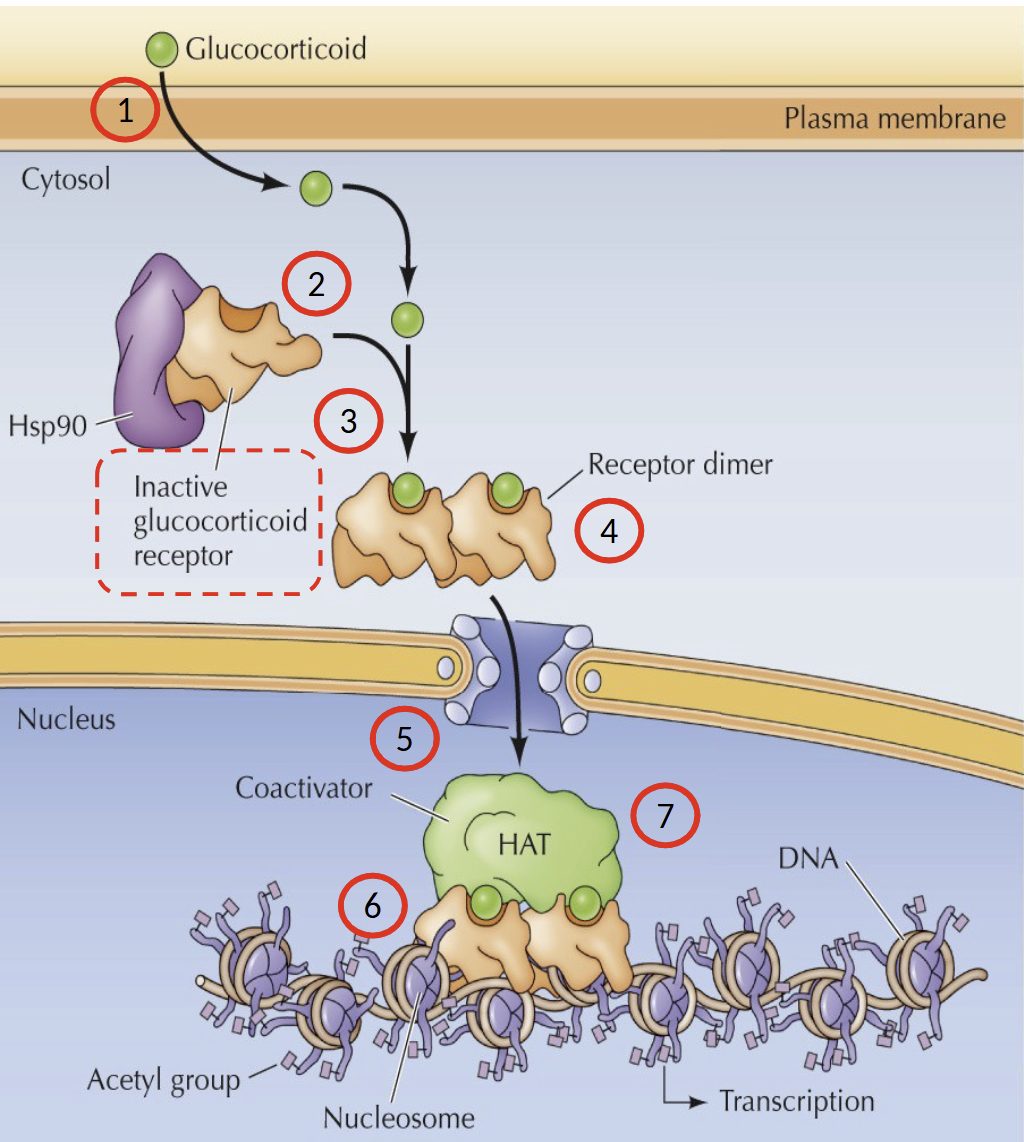
Describe the process of glucocorticoid signaling pathway
Glucocorticoid diffuses across plasma membrane into cytosol and binds to inactive glucocorticoid receptor/Hsp90 (chaperone) complex. Receptor is then dimerized and the nuclear localization signal is exposed → dimer transported across nuclear envelope. Receptor dimer binds to recognition sites in DNA.
Association with co-activators with Histone Acetyltransferase (HAT) activity to stimulate transcription
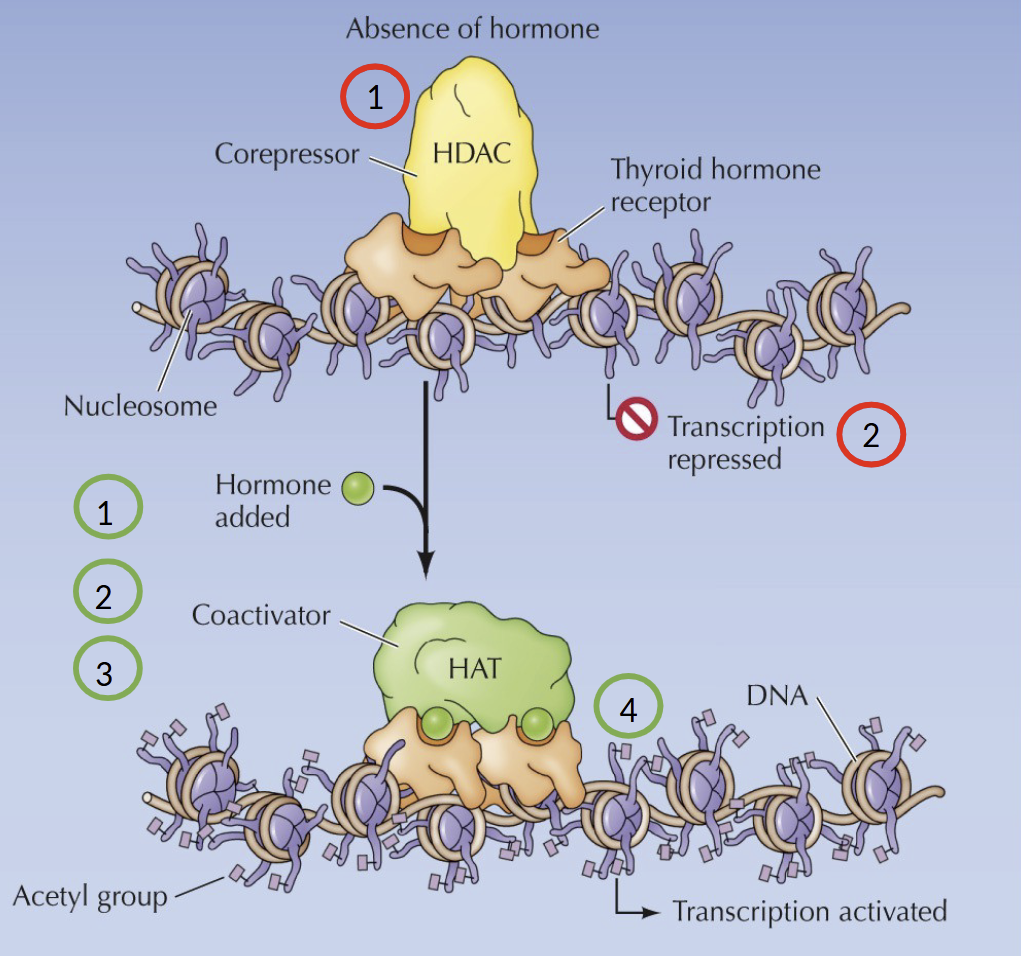
What happens in the absence of a hormone in the Nuclear Receptor Family
The receptor associates with co-repressors with histone deacetylase (HDAC) activity → repression of target gene expression
What happens in the presence of a hormone in the Nuclear Receptor Family
Hormone binds to the receptor, inducing a conformational change. The co-repressor is displaced and association with co-factors with histone acetyltransferase (HAT) activity
What is weird about thyroid hormone receptor
Binds to its own recognition sites in DNA in the presence or absence of the hormone
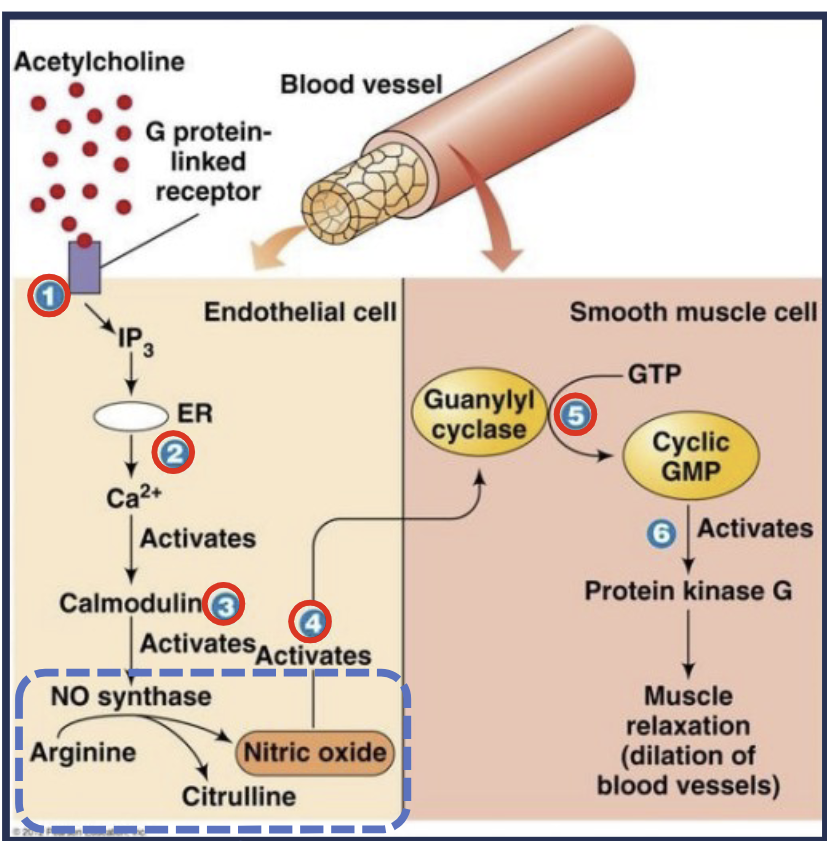
Describe small molecule signaling
Neurotransmitters are released (acetylcholine), and act on endothelial cells to stimulate nitric oxide (NO) synthesis. NO diffuses to neighboring smooth muscle cells and activates guanyl cyclase, synthesizing cyclic GMP leading to muscle cell relation and blood vessel dilation
Application: Nitroglycerin dilates blood vessels
What does an epidermal growth factor (EGF) do
Proliferates many types of cells. These molecules are not able to cross the plasma membrane.
3 internal disulfide bonds
How do cells send signals across cell membranes?
Ligands bind to a receptor on the cell surface initiating signal transduction (intracellular reactions)
What are G-proteins
The largest family of cell surface receptors for many hormones and neurotransmitters.
Responsible for smell, sight and taste
GPCR
7 transmembrane alpha helices. Intracellular ligand binding domain has guanine nucleotide exchange factor (GEF) activity. Carbohydrates are required for proper folding of receptor. Transmit via the G-proteins
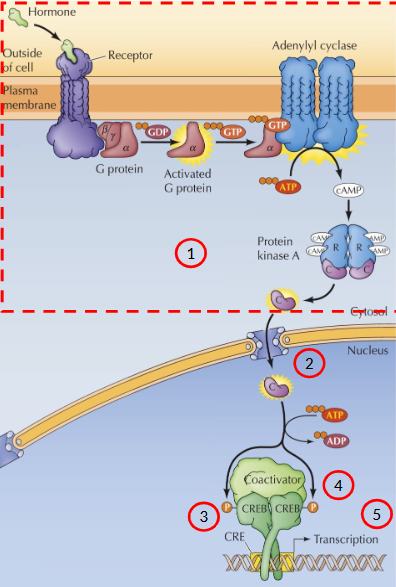
Activation of adenylyl cyclase by epinephrine
Epinephrine binds to the extracellular domain of the receptor → conformational change in the intracellular domain → activates the G protein associated with it.
G protein dissociates from the receptor and carries the signal to the intracellular target (enzymes, ion-channels)
ex. Epi regulates the synthesis of cAMP
What is cAMP
An important sencondary messenger that mediates a number of ceullular responses.
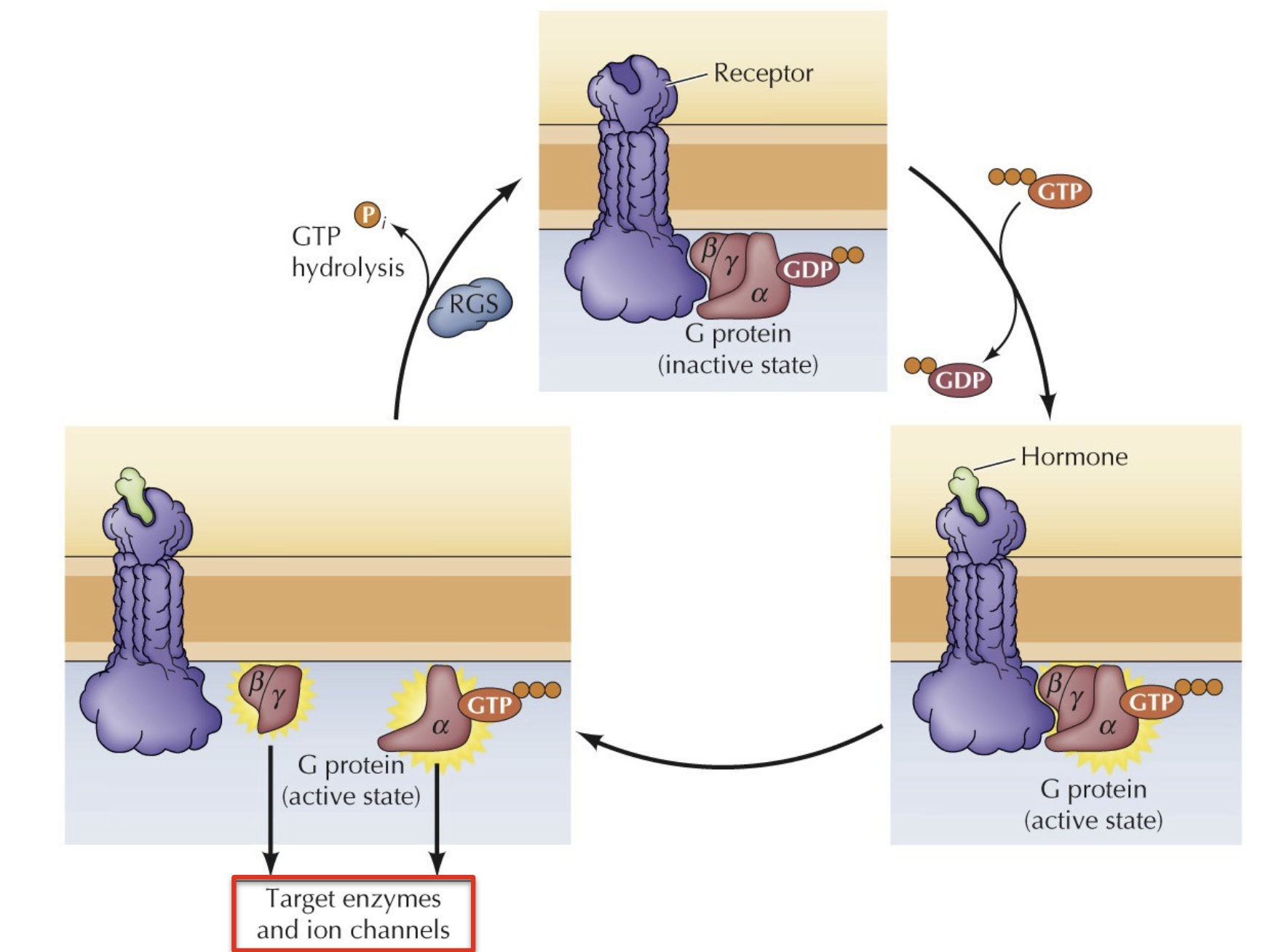
In the absence of a hormone, what does the GPCR look like
In the absence of hormone, GPCR inactive with alpha subunit bound to GDP in a complex with beta and gamma
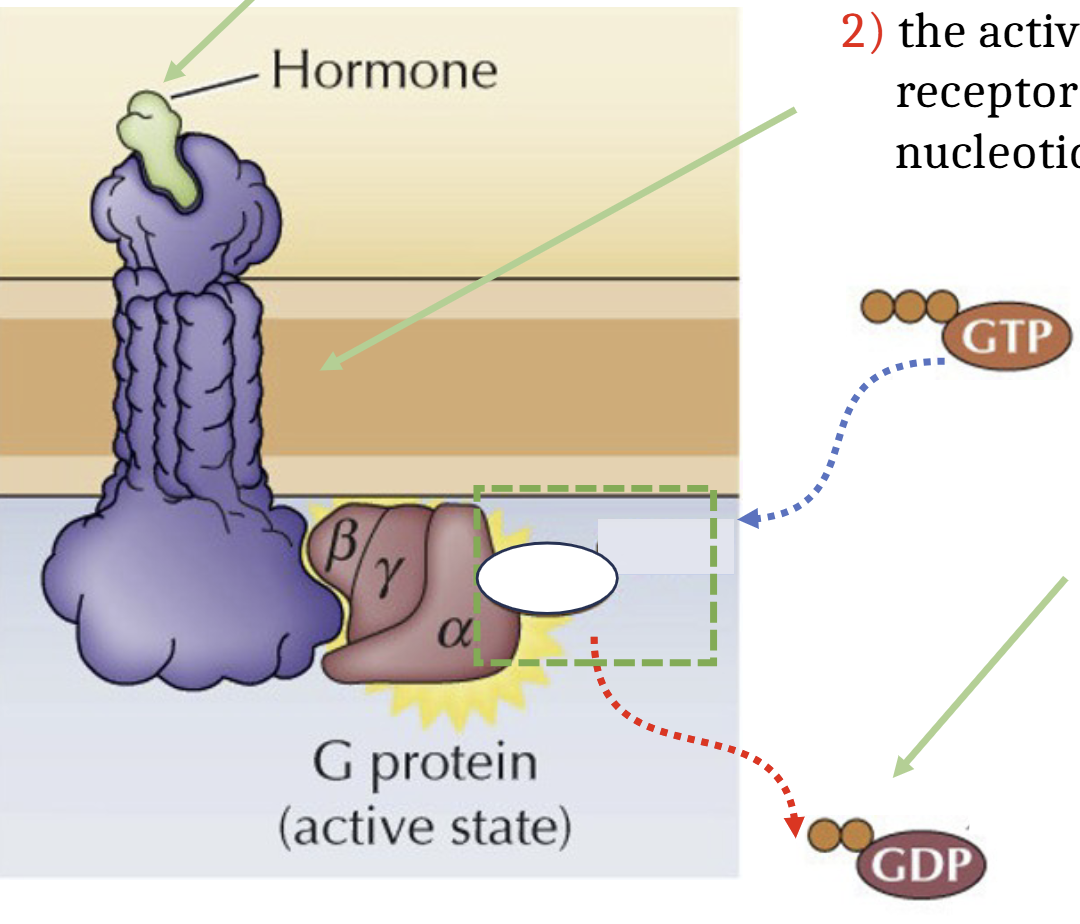
With hormone what does the GCPR process look like
Hormone binds to extracellular domain of GCPR, activating the GPCR causing it to act as a guanine nucleotide exchange factor, stimulating the release of GDP. GDP is exchanged for GTP. G protein in active state.
Activate GT-bound alpha subunit and beta-gamma complex dissociate from the receptor and interact with their targets (enzymes and ion channels)
GAP protein hydrolyzes GTP → GDP, sets G protein into inactive state.
What happens without GAP protein
G proteins are constitutively on because of their slow hydrolytic activity and GEFs constantly replacing GDP with GTP. This results in unregulated cell division and the formation of tumors
What do Gs alpha subunits do
Stimulate target enzymes (adenylyl cyclase)
What do Gi alpha subunits do
Inhibit target enzymes (Adenylyl cyclase)
Describe the process of signal amplification through epinephrine
x1 signal molecule → x100 signal amplification
Epi stimulates adenylyl cyclase to synthesize cAMP
Most effects of cAMP are mediated by the action of cAMP-dependent kinases (Protein Kinase A = PKA)
PKA is a tetramer consisting of 2 regulatory subunits and 2 catalytic subunits (inactive state)
cAMP binds to regulatory subunits → induces a conformation changes that causes dissociation of the catalytic subunits
Catalytic subunits are now enzymatically active - phosphorylate serine residues in target proteins
Catalytic subunit phosphorylates phosphorylase kinase, which in turn phosphorylates glycogen phosphorylates
Glycogen phosphorylase then catalyzes the breakdown of glycogen to glucose-1-phosphate
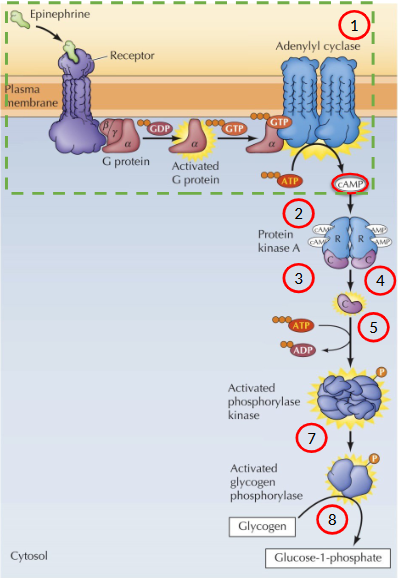
Describe the process of signal amplification through a generic hormone
Signal leads to the activation of the catalytic domain of PKA
Signal translocates from the cytoplasm to the nucleus
PKA catalytic subunit the phosphorylates a transcription factor called CRE-binding protein (CREB)
Co-factor binds CREB
Transcription of cAMP-inducible genes
cAMP activates specific transcription factors that contain a sequence called the cAMP response element
Kinases vs phosphatase
Kinase phosphorylate
Phosphatases dephosphorylate
Actions done by PKA is reversed by action of protein phosphatase 1
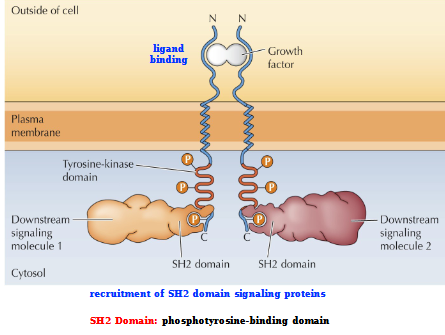
Signaling by Receptor tyrosine kinases
Ligand binding (Growth factor)
Recruitment of SH2 (phosphotyrosine-binding domain) domain signaling proteins
= Downstream signaling
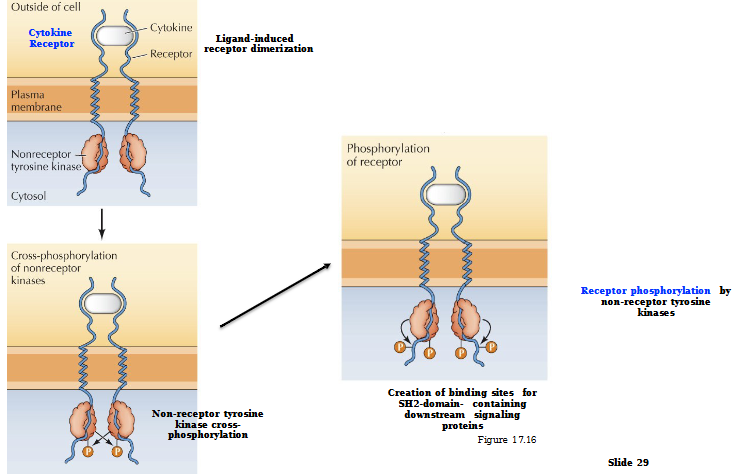
Non-receptor Tyrosine Kinase Activation by Non-Enzymatic Receptors
Ligand-induced receptor dimerization
Non-receptor tyrosine kinase cross-phosphorylates
Creation of binding sites for SH2 domain - containing downstream signaling proteins
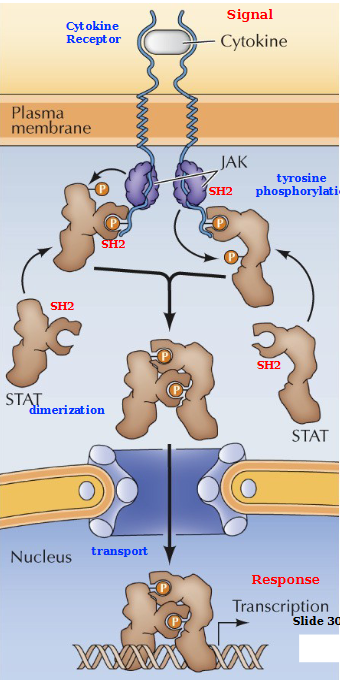
What is Janus Kinase Family (JAK)
non-receptor tyrosine kinase
STAT Proteins
Signal transducer and activator of transcription
Describe the JAK/STAT Pathway
Cytokine (signal) binds to the cytokine receptor. JAK (non-receptor tyrosine kinase) Cross phosphorylates and directly activates STAT proteins. These STAT proteins dimerize and are then transported across the membrane into the nucleus where they transcribe the gene to produce a response.
What are integrins
Major receptors responsible for the attachment of cells to the ECM
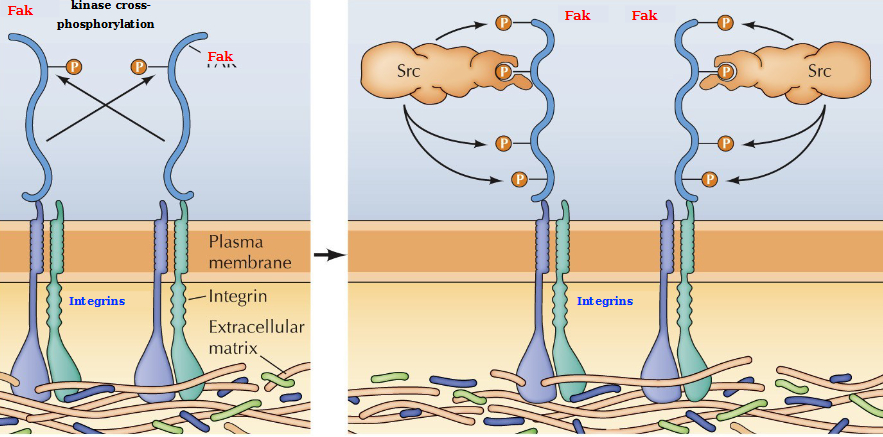
Non-receptor tyrosine kinases Fak and Src
Binding of integrins to the ECM leads to integrin clustering and activation of FAK by autophosphorylation. SRC then binds to the FAK autophosphorylation site and phosphorylates FAK on additional tyrosine residues, which serve as binding sites for downstream signaling molecules
What is a MAP kinase
Mitogen-activated protein kinase
Activated in response to a variety of growth factors and Inflammatory cytokines or cell stress
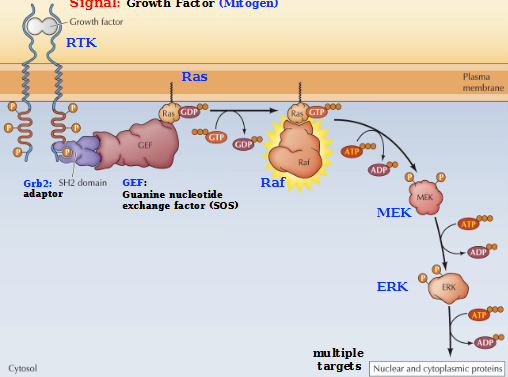
Describe the MAP kinase cascade with a growth factor signal
Activation of RAS (small GTP-binding proteins)
RAS converts to the active state (GTP-bound) via guanine nucleotide exchange factors (GEFs)
Active RAS complex activates RAF protein kinase, which phosphorylates and activates MAK that activates ERK, a MAP kinase
ERK phosphorylates a variety of nuclear and cytoplasmic protein targets. (Leads to a response: proliferation, differentiation, cell survival)
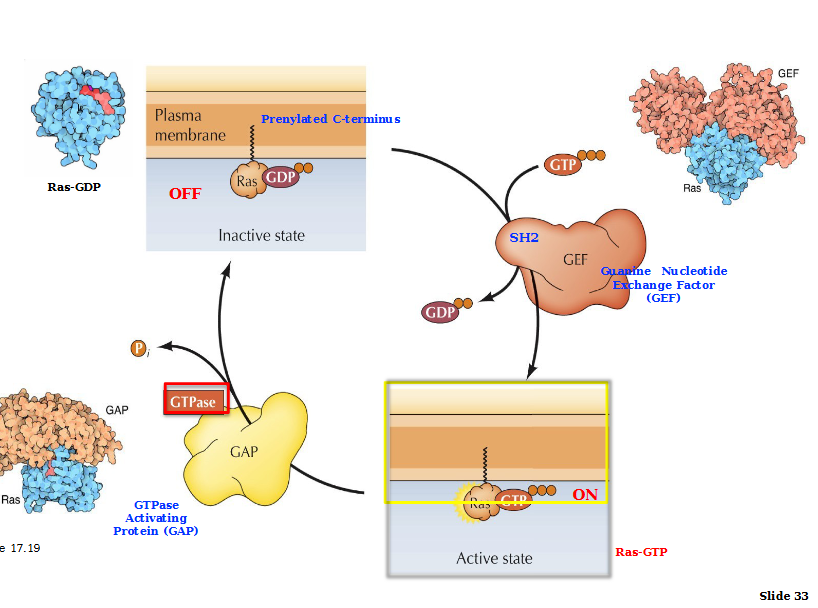
Regulation of RAS proteins
Initiation: Ras is inactive when GDP bound. SH2 domain guanine nucleotide exchange factor (GEF) activates Ras via GDP/GTP exchange.
Termination: Stimulated by GTPase- activating proteins (GAPs) interactions with Ras-GTP. These GAPs hydrolyze GTP into GDP, returning Ras into inactive state.
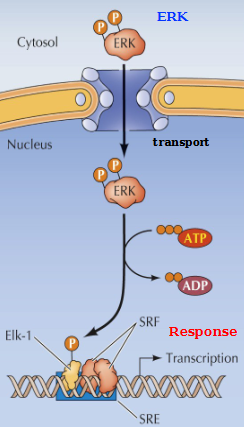
Induction of Gene expression by ERK
Activated ERK (in the cytosol) translocates to the nucleus, where it phosphorylates the TF Elk-1
Elk-1 in turn binds to the serum response element (SRE) in a complex with serum response factor (SRF). This activation leads to immediate-early gene expression
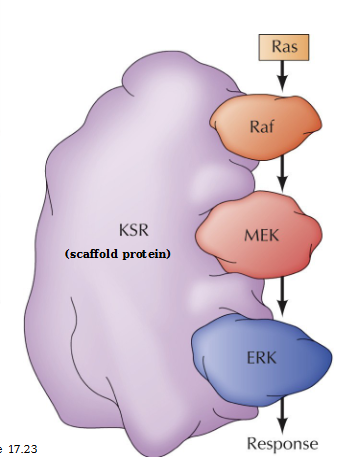
What do scaffold proteins do
Organize complexes of signaling molecules and maintain the specificity of MAP kinase signaling
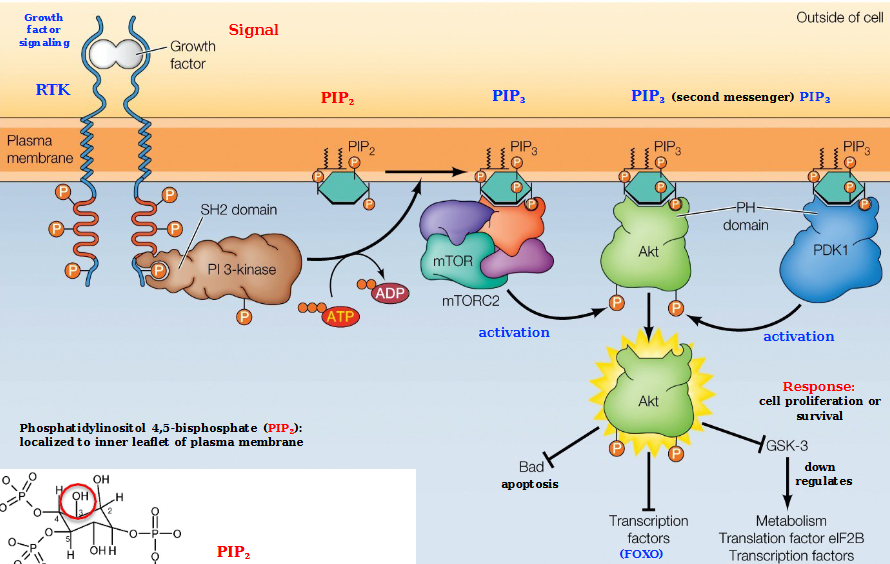
PI 3-Kinase/Akt Pathway
Growth factor signaling triggers the formation of an SH2-domain which recruits PI 3-Kinase.
PI 3-kinase phosphorylates PIP2 into PIP3
PIP3 recruits ATK binding (must have 3 total PIP3 for ATK to activate)
ATK is activated via PDK1 and mTORC2 phosphorylation by protein kinases
ATK phosphorylates a number of proteins that regulate cell proliferation and survival
Absence of growth factor with FOXO
No growth factor → no activation of
Transcription of FOXO-induced genes expresses genes that inhibit proliferation or induces apoptosis
No FOXO transcription leads to…
Cell proliferation or survival
What is autophagy in the mTOR pathway
Turnover and recycling of cell componenets
Describe the mTOR pathway with high cellular energy
Initiated with a growth factor signal binding to the N-terminus of the transmembrane receptor
This causes phosphorylation, allowing the SH2 domain to bind PI 3- kinase
mTORC2 and PDK1 (all with PIP3) bind to the phosphates on Akt, releasing an AKT subunit that goes and binds to TSC (a GTPase-activating protein)
The causes Rheb - a small STP-binding protein - to attach to mTORC1.
mTORC1 stimulates translation by phosphorylating eIF4E-BP1, relieving inhibition of an essential translation initiation factor eIF4E
In addition, mTORC1 inhibits autophagy by phosphorylating 2 of the Atg proteins required for autophagosome formation (S6 kinase and ribosomal protein S6)
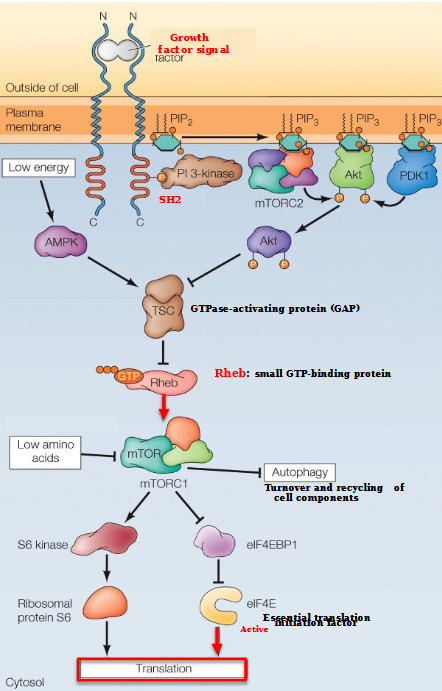
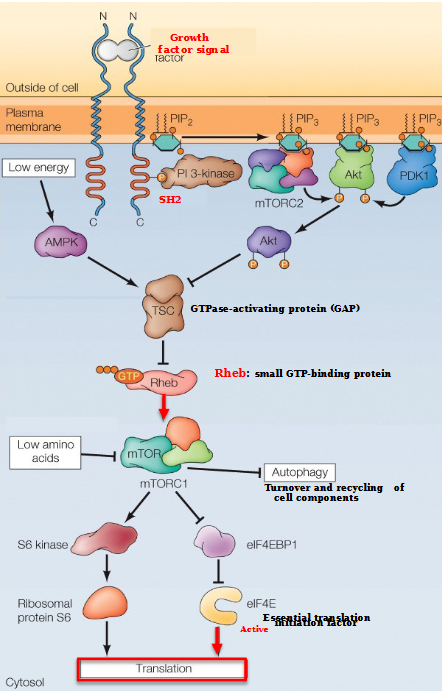
When is the mTOR pathway inhibited?
AMPK activates TSC, leading to the inhibition of Rheb and mTORC1.
mTORC1 is also inhibited by amino acid starvation
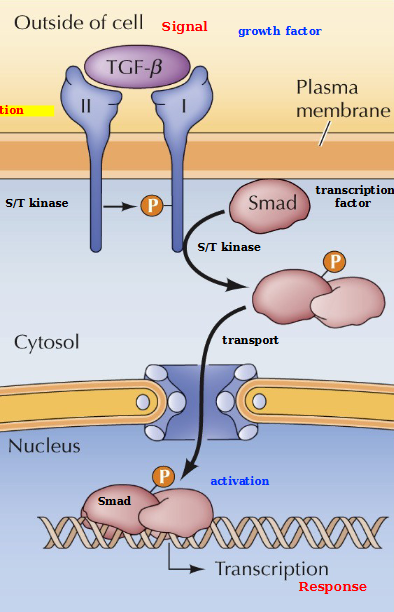
What are TGF-β receptors
Serine/Threonine (S/T) kinases that phosphorylate Smad transcription factors. They are dimers of type I and II polypeptides
Describe NF-xβ signaling
TGF-β type II receptor phosphorylates and activates type I (through heterodimerization), which phosphorylates a SMAD protein. Phosphorylated SMADs form complexes and translocate to the nucleus to activate transcription of target genes. This leads to Proliferation, differentiation, and survival?
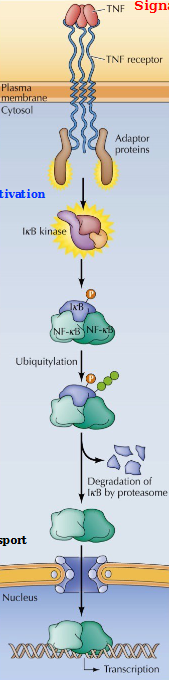
What is a TNF
Tumor necrosis factor - a cytokine that induces inflammation and cell death
What is IkB
An inhibitor of NF-kB: sequesters NF-kB in the cytoplasm
Describe NF-kB signaling from the TNF receptor
A TNF binds to a TNF receptor, recruiting the adaptor proteins in the cytosol and activating them.
The adaptor proteins activate IkB kinase, which ubiquitizes IxB, causing its degradation by the proteosome
This allows the NF-kB to translocate to the nucleus and activate transcription of its target genes
This mechanism diplays feeback inhibition as well: the transcription of IkB mRNA by NF-kB forms an IkB complex that then plops on the NF-kB dimer and inhibits translocation to the nucleus and therefore transcription.
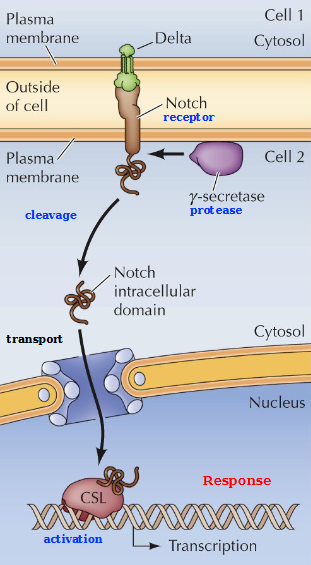
Describe Notch signaling
The binding of Delta leads to proteolytic cleavage of Notch by γ-secretase complex
This releases Notch intracellular domain which translocates to the nucleus and interacts with CSL - a transcription factor to induce gene expression. This controls cell fate during development.
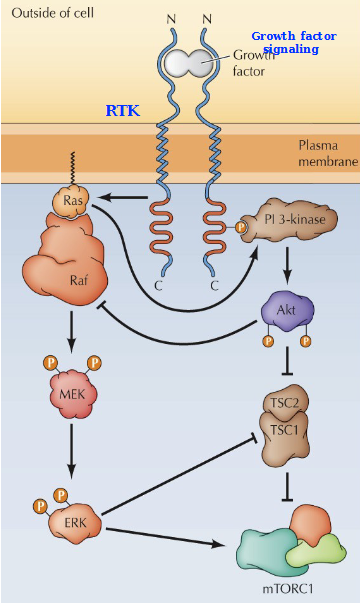
Crosstalk between the ERK and PI 3-Kinase signaling pathways
The Ras/Raf/MEK/ERK and PI 3-Kindase/AKT/mTORC1 pathways are connected by both positive and negative crosstalk, including activation of PI-3 Kinase by Ras, inhibition of Raf by Akt, Inhibition of TSC by ERK, and activation of mTORC1 by ERK
Phases of the cell cycle
Interphase
G1 - Cell grows and copies organelles
S - DNA synthesis, duplicates centrosome
G2 - Cell grows more, makes proteins and organelles, begins to reorganize contents
Mitosis - Cell divides (cytokinesis)
Embryonic cell cycle
Rapid, synchronous divisions tha oscillate between S and M phases (DNA synth. and cytokinesis), skipping G1 and G2.
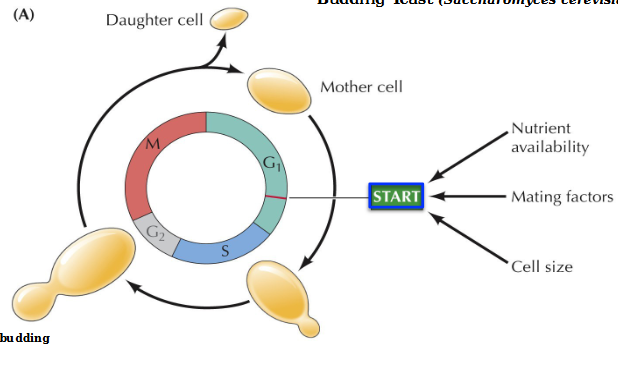
What is the start point in the cell cycle of budding yeast
Start is a major regulatory point in late G1 controlling progression from G1 to S phase. Requires adequate nutrient availability, mating factors, and cell size
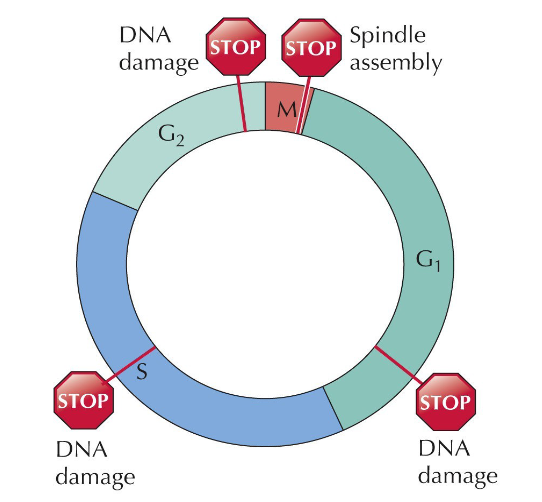
Regulation of the Cell Cycle - checkpoints
Late in G1 is a restriction point - if not enough growth factors are present, the cell will restart at G0.
Checkpoints in late G1, mid S, and late G2 phases all indicate DNA damage and immediately stop the process of cell division. In Mitosis, division is halted it an error in spindle assembly occurs
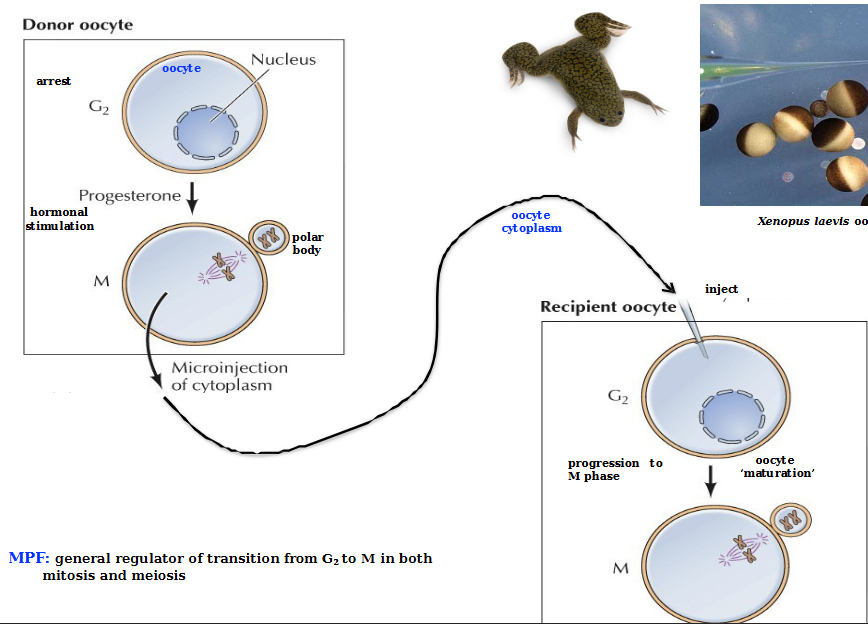
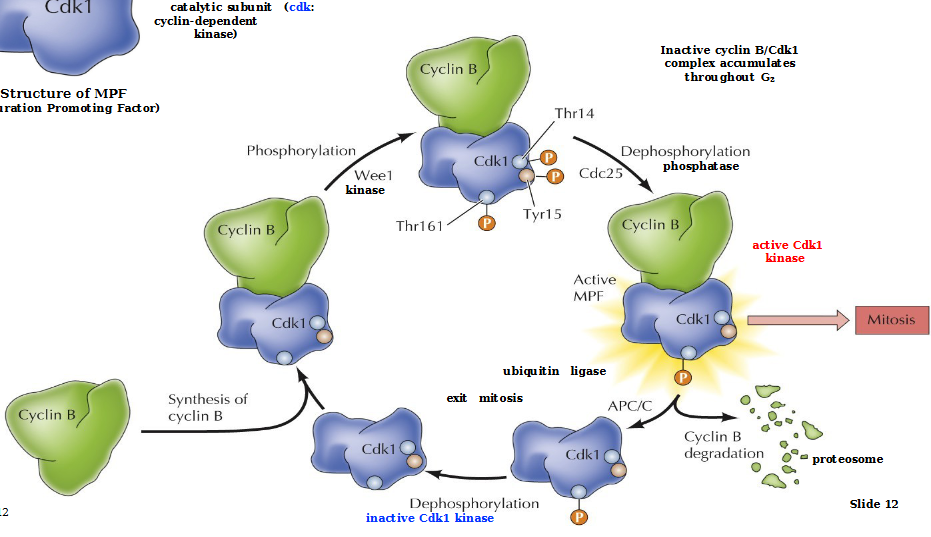
MPF
Maturation Promoting Factor - general regulator of transition from G2 to M in both mitosis and meiosis
Consists of a regulatory subunit (Cyclin B) and a catalytic subunit Cdk1 (cyclin-dependent kinase)
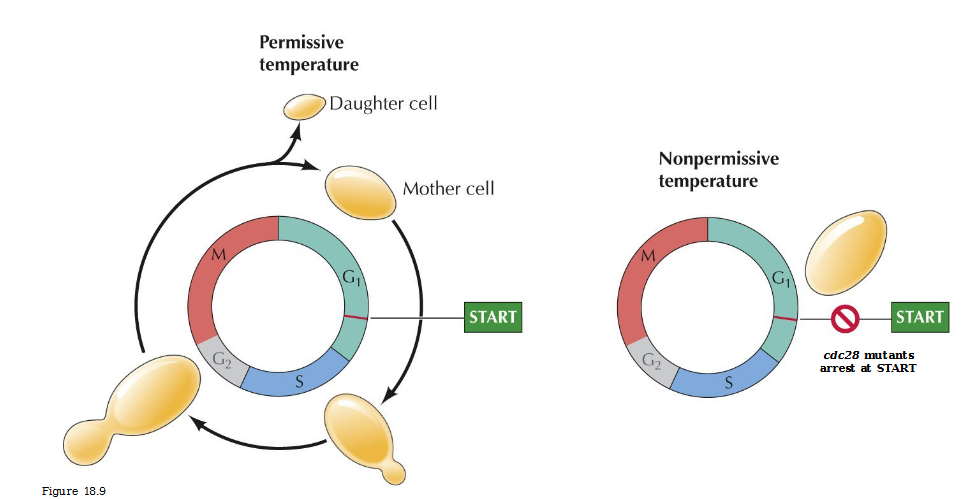
Cell division cycle (csc) mutants
Temperature sensitive mutats defective in cell cycle progression
Cdc28 mutatns arrest at START
Cyclin levels in sea urchin embryos
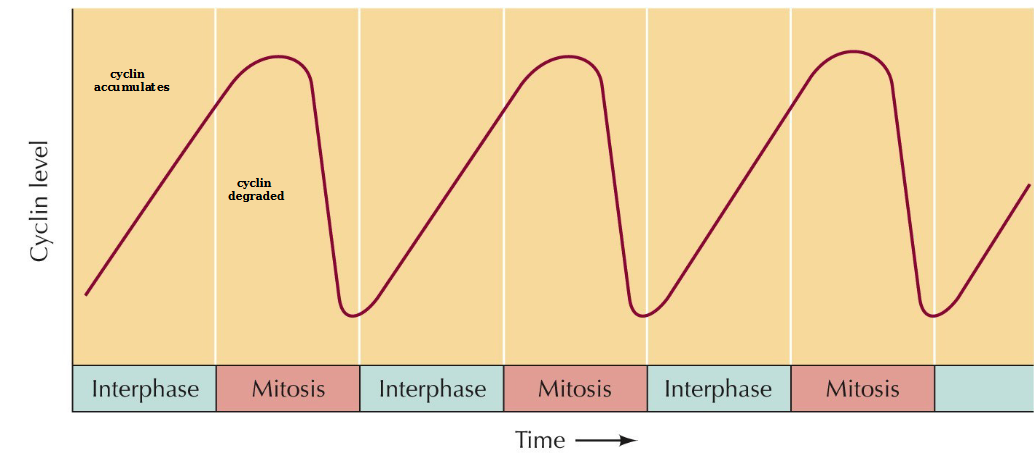
Describe MPF regulation
Cyclin B binds to inactive Cdk1
Wee1 kinase phosphorylates all the blobs on Cdk 1:Thr161 (required for kinase activity), Thr14/Tyr15 (inhibitory)
Cdc25 phosphatase dephosphorylates Thr14/Tyr15 to produce an active Cdk1 kinase MPF which allows for mitosis to occur.
Cyclin B is degraded by proteosomes and Cdk1 is dephosphorylated to start all over again
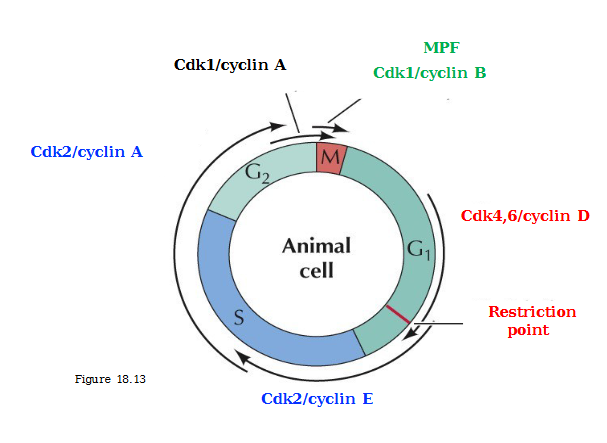
Regulation of Cdk activity
Association with a specific cyclin (controlled by synthesis and degradation
Activation requires phosphorylation (CAK: Cdk-activating kinase)
Inhibitory phosphorylation (Wee1 protein kinase)
Activation requires removal of inhibitory phosphate (Cdc25 protein phosphatase)
CKIs - Cdk inhibitory proteins (Ink4 family proteins, Cip/Kip family proteins)
Induction of D-type cyclin synthesis in response to growth factor stimulation
Growth factors → Ras/Raf/MEK/ERK pathway → Synthesis of D-type cyclins Cdk4, 6/CycD
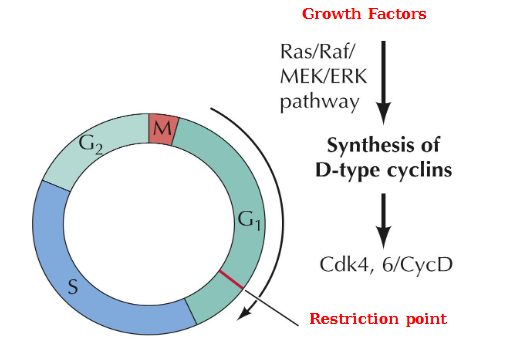
Key target of active Cdk4, 6/CycD?
Rb/E2F complex. Inhibition of this complex by Cdk4, 6CycD allows transcription to be activated and cell cycle progression
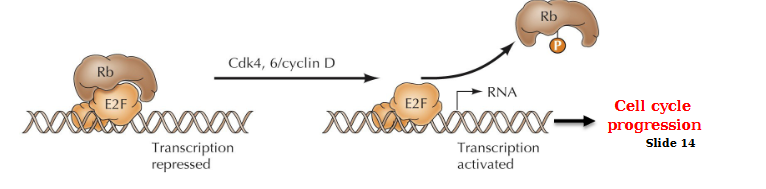
E2F activation transcription
Cyclin E
DNA synthesis proteins
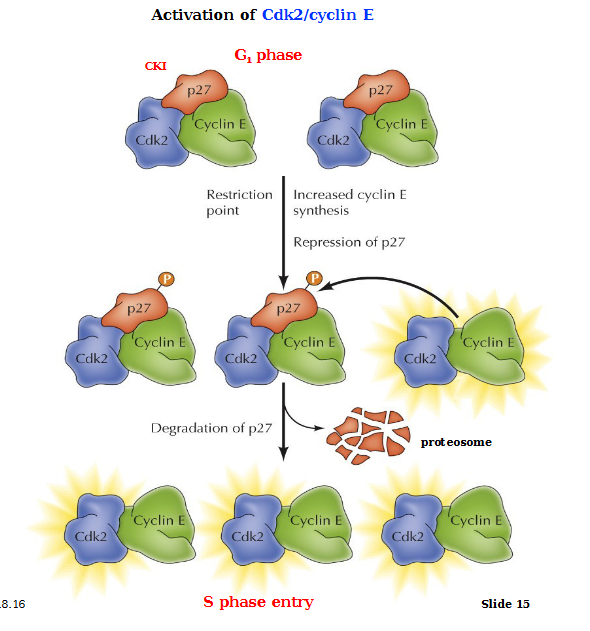
p27: Cdk inhibitor (CKI)
Activation of Cdk2/CycE
In the G1 phase, CKI p27 inhibits Cyclin E and Cadk2 complexes. Passage through the restriction point induces the synthesis of Cyclin E via activation of E2F. Growth factor signaling inhibits synthesis of p27. Cdk2 becomes activated, it phosphorylates and targets p27 for degradation, resulting in further activation of Cdk2/CycE complexes. This allows for progression into the S phase
Cdk2/CycE also inhibits ubiquitin ligase, preventing cyclin E degradation
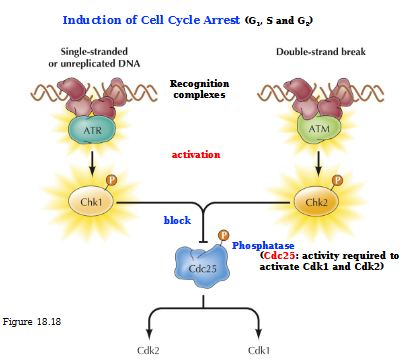
ATR and ATM
Protein kinases activated by DNA damage
Describe the induction of cell cycle arrest (G1, S, and G2)
Single-stranded or unreplicated DNA: ATR is the recognition complex that activates Chk1. Activated Chk1 blocks Cdc25 so no Cdk2 or Cdk 1 is produced
In a double-stranded DNA, ATM is the recognition complex that activates Chk2. Activated Chk2 blocks Cdc25 no no Cdk2 or Cdk1 can be produced
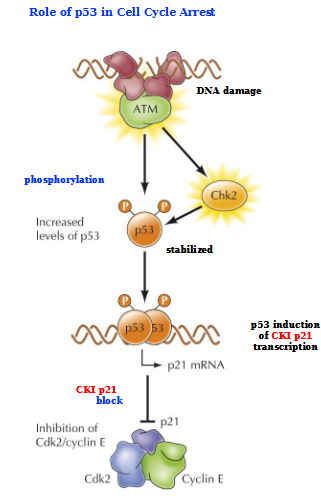
Role of p53 in Cell Cycle Arrest
Activated ATM (indicating DNA damaged) activates Chk2 and phosphorylates p53 (stabilizes it)
p53 induces CKI p21 transcription
p21 sits on top of Cdk2/Cyclin E complex
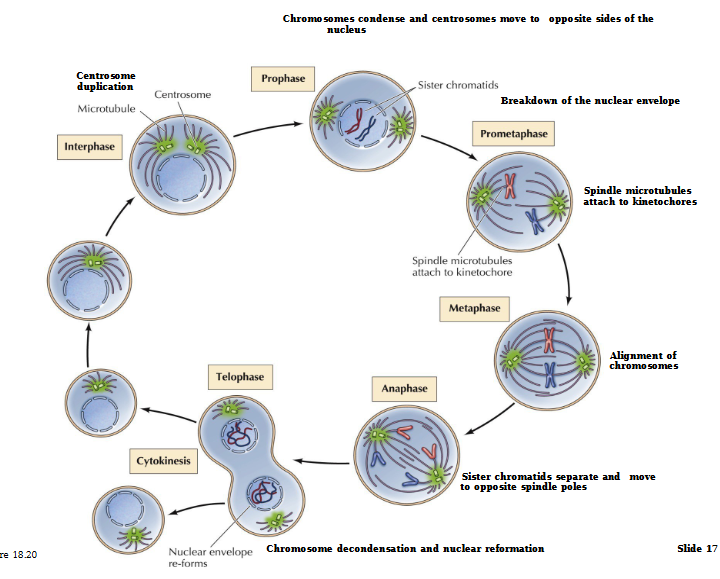
Stages of Mitosis
Interphase (G1, S, G2) - Duplication of DNA, growth of cell and proteins, duplication of organelle
Prophase - Chromosomes become visible. Centrioles separate and move to poles
Prometaphase - Nuclear envelope disintegrates. Chromosomes move toward metaphase plate
Metaphase - Chromosomes line up and attach to spindle fiber at the centromere
Anaphase - Spindle pulls apart sister chromatids into separate chromosomes
Telophase - Chromosomes decondense. Nuclear envelope reforms
Cytokinesis - Cytoplasm separates and 2 identical daughter cells form
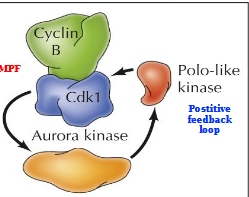
Mitotic Protein Kinases
Aurora kinase and Polo-like kinase act in a positive feedback loop with MPF (Cyclin B/Cdk1). Leads to:
Chromatin condensation - phosphorylation of condensins and cohensins
Nuclear envelope breakdown - Phosphorylation of lamins, nuclear pore complexes, and inner nuclear membrane proteins
Fragmentation of Golgi apparatus - Phosphorylation of Golgi matrix proteins
Spindle formation - Phosphorylation of centrosome, kinetochore, and microtubule-associated proteins
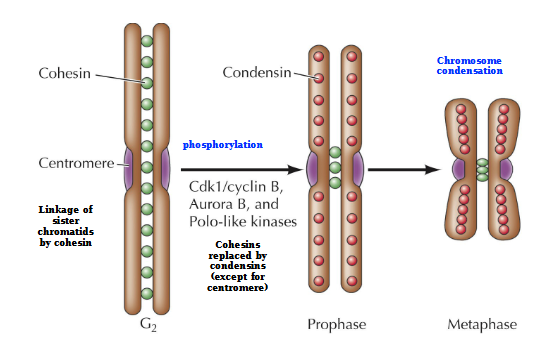
Action of Cohesins and Condensins
G2 - Cohesins links the chromosomes
Phosphorylation of Cdk1/CycB, Aurora B, and Polo-like kinases
Prophase - Cohesins replaced by condensins (except for centromere)
Metaphase - Chromosome condensation
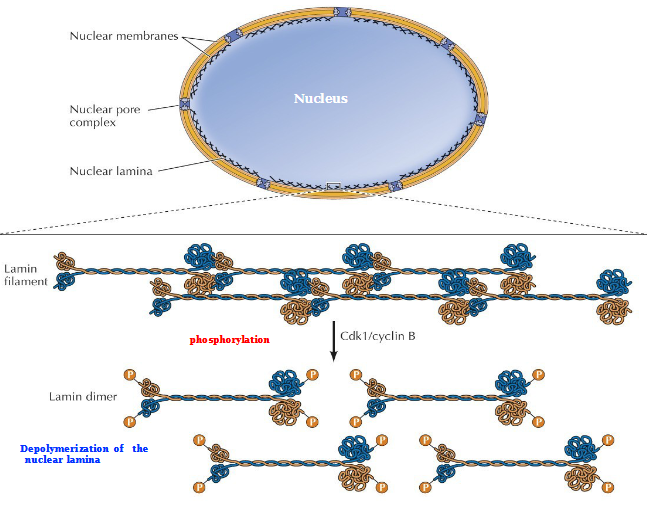
Breakdown of the nuclear envelope during mitosis in animal cells
Cdk1/Cyclin B phosphorylates the lamin filament → separation of lamina into lamin dimers
Depolymerization of the nuclear lamina
Centromere
Specialized chromosomal region that connects sister chromatids and attaches them to the mitotic spindle
Kinetochore
Protein structure attached to the centromere that mediates attachment and movement of chromosomes along the mitotic spindle
Organization of types of microtubules in metaphase
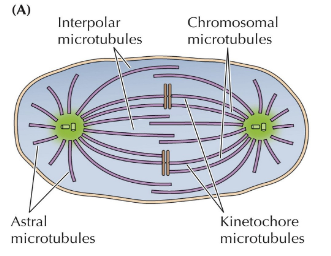
Spindle Assembly Checkpoint
Monitors chromosome alignment on the metaphase spindle
MCC: mitotic checkpoint complex, blocks APC/C from binding meaning an inactive ubiquitin ligase
Progression to anaphase mediation
Activated APC/C ubiquitin ligase (activated with Cdc20) will:
Tags MPF with Ubiquitin causing the degradation of Cyclin B and an inactive Cdk1 complex
Tags Securin - the inhibitory subunit of seperase - causing the degradation of securing and an active separase. The active separase degrades cohesin and allows for the separation of chromosomes
Contractile ring
Actin an myosin II filaments
Properties of fibroblasts
Dispersed through connective tissue, secrete collagen
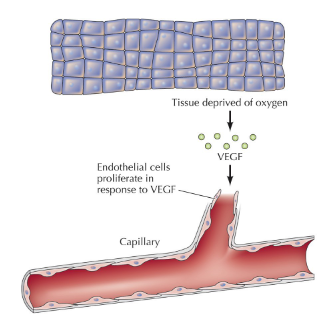
Endothelial cells
Line the lumen of blood vessels.
Endothelial cell proliferate in response to VEGF
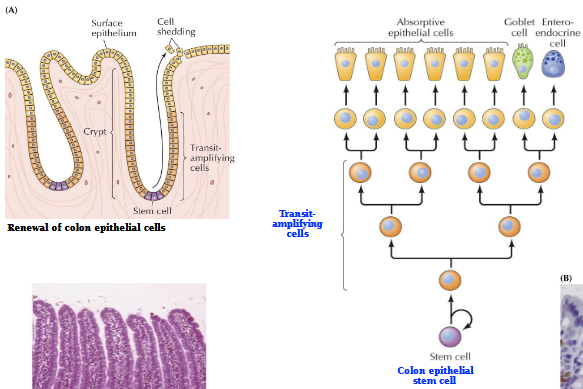
Process of Stem cell proliferation
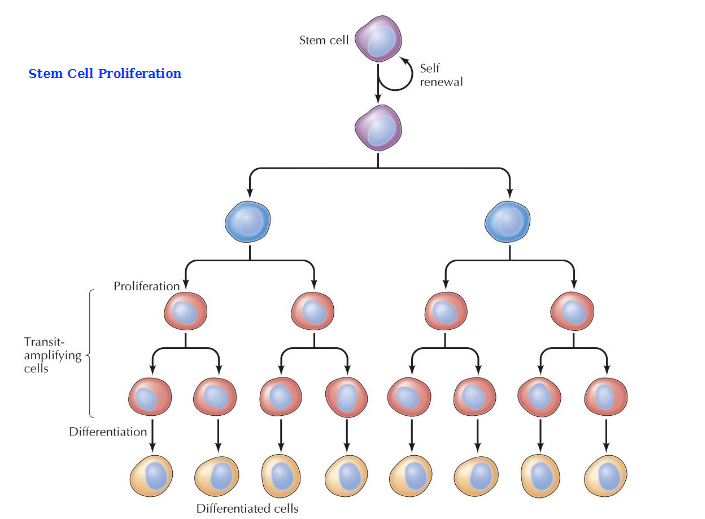
What are transit-amplifying cells (TACs)
Non-differentiated, proliferating cell population, an early intermediate in tissue regeneration. Bridge the gap between stem cells and fully differentiated progeny
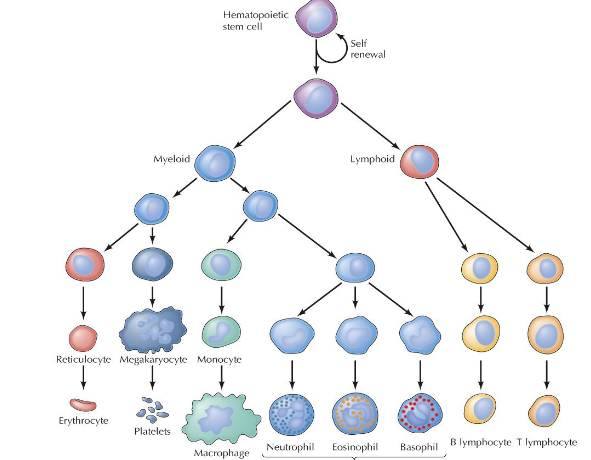
What are hematopoietic stem cells (HSCs) and how is their differentiation process different from other stem cells?
Self-renewing multipotent (can only become blood cells) cells found mainly in the bone marrow or umbilical cord blood. They function as the source for all blood cells, differentiating into RBCs, WBCs, and platelets. Crucial for lifelong blood formation, immune responses, and regeneration.
Very organized branching pathway, differentiate into myeloid or lymphoid and proliferate through the structure from there.
Intestinal epithelium self-renewal process
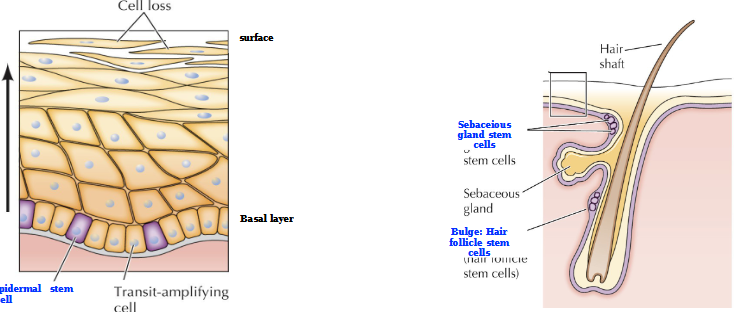
Self renewal of the skin and hair
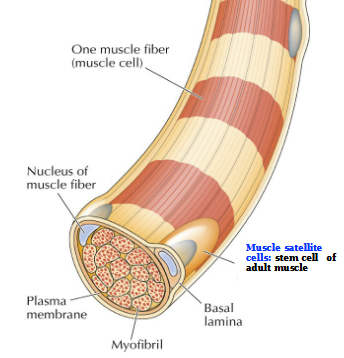
Muscle fibers
large multinucleated cells formed by cell fusion
Muscle satellite cells
stem cell of adult muscle
Self-renewal of skeletal muscle cells in response to tissue damage
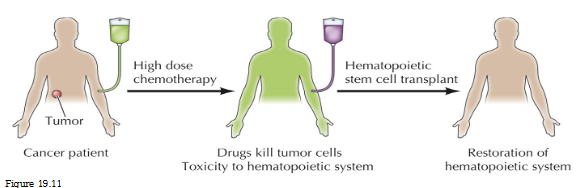
Hematopoietic stem cell transplatation
Cancer patient receives high dose chemotherapy
Drugs kill tumor cells
Toxicity to hematopoietic system
Hematopoietic SC transplant = restoration of hematopoietic system
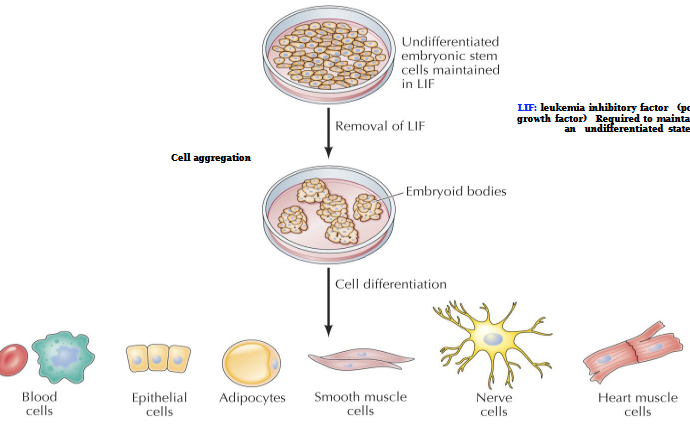
Leukemia inhibitory factor (LIF)
Polypeptide growth factor. Required to maintain cells in an undifferentiated state
Specific cell differentiation can be directed by the addition of appropriate growth factors
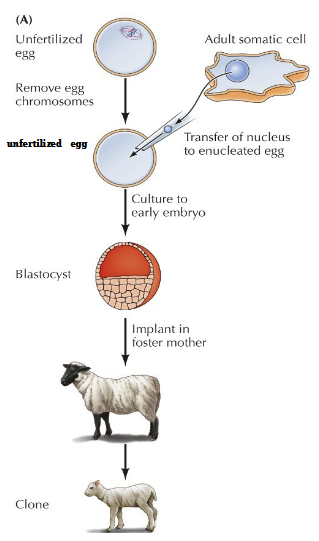
Reproductive cloning by somatic cell nuclear transfer (1997)
Unfertilized egg with chromosomes removed
Transfer of nucleus to enucleated egg
Culture to early embryo = blastocyte
Implant in foster mother = 1st cloned mammal
Blastocyte
Undifferentiated, early embryonic cell found during the blastula stage of development.
Critical to early pregnancy, especially in implantation
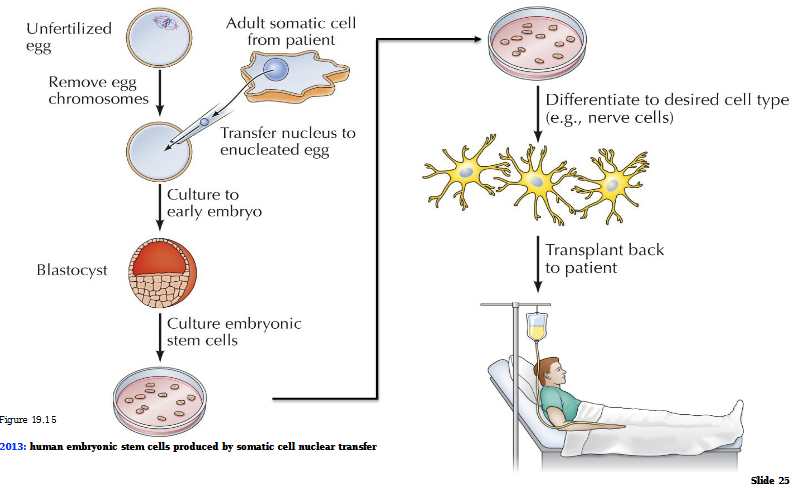
Therapeutic cloning by somatic cell nuclear transfer (2013)
Unfertilized egg with chromosomes removed
Transfer of nucleus to nucleated egg
Culture to early embryo = blastocyte
Culture embryonic stem cells
Differentiate to desired cell type (ex. neurons)
Transplant back to patient
Induced pluripotent stem cells (iPSCs)
Adult somatic cells (skin or blood) reprogrammed to a pluripotent state, allowing them to differentiate into any cell type in the body.
Adult mouse fibroblast + infection with retroviruses carrying genes for Oct4, Sox2, Klf4, and c-Myc transcription factors = induced pluripotent stem cells
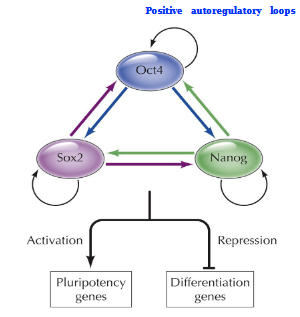
Pluripotency transcriptional program
Positive autoregulatory loop. A self-sustaining network governed by core TFs Oct4, Sox2, and Nanog that maintain ESCs in an undifferentiated state while enabling self-renewal.
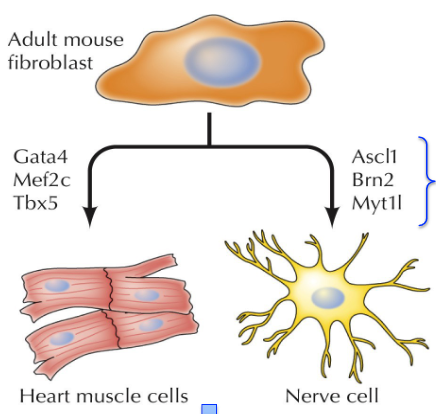
Transdifferentiation of somatic cells
Adult mouse fibroblast + lineage specific transcription factor sets = desired cell type → transplantation therapy
Apoptotic Process
DNA fragmentation, Chromatin condensation
Fragmentation of nucleus
Fragmentation of the cell
Apoptotic bodies
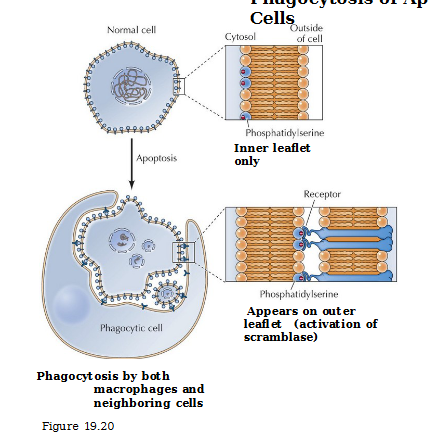
Phosphatidylserine
Presence on exterior of cell recognized by receptors on phagocytic cells (‘eat me’ signal)
Phagocytosis of apoptotic cells
In normal cells, phosphatidylserine on the inner leaflet only. In an apoptotic cell, it appears on the outer leaflet via activation of scramblase and binds to receptor of macrophages or neighboring cells that consumes it.