Chapter 6 - Principles of Stereochemistry
1/18
Earn XP
Description and Tags
Studying stereoisomers, which are compounds that have the same atomic connectivity but a different arrangement of atoms in space.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
Absolute configuration
refers to the spatial arrangement of atoms in a chiral molecule, typically described using the Cahn-Ingold-Prelog priority rules to assign R or S configuration at each asymmetric carbon atom.
Chiral
molecules that have non-superimposable (aka different) mirror images, meaning they exist in two configurations that are mirror images of each other.
Chiral carbon atom
aka asymmetric carbon; a carbon atom that is bonded to four different groups.
Chiral center
the IUPAC term for an atom holding a set of ligands in a spatial arrangement that is not superimposable on its mirror image. asymmetric carbon atoms are the most common chirality centers.
Allene
compounds having two C=C double bonds that meet at a single carbon atom, C=C=C. the two outer carbon atoms are trigonal planar, with their planes perpendicular to each other. many substituted allenes are chiral!
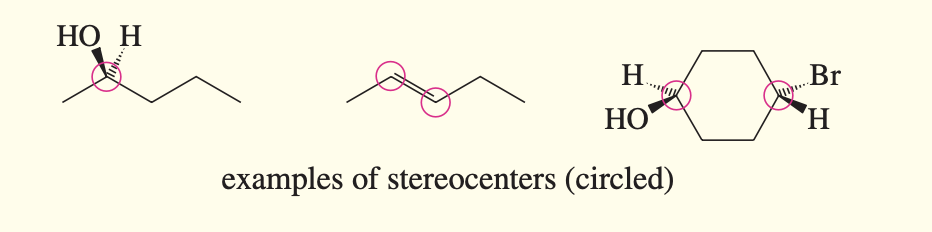
Stereocenter
aka stereogenic atom; an atom that gives rise to stereoisomers when its groups are interchanged. asymmetric carbon atoms and double-bonded carbons in cis-trans alkenes are the most common stereocenters!!
Cis
on the same side of a ring or double bond.
Geometric isomers
aka cis-trans isomers; isomers that differ in their geometric arrangement on a ring or double bond; a subclass of diastereomers.
Stereoisomers
aka configurational isomers; isomers whose atoms are bonded together in the same order but differ in how the atoms are oriented in space.
Structural isomers
aka constitutional isomers; isomers that differ in the order in which their atoms are bonded together.
Conformational isomers
aka conformers; structures that differ only by rotations about single bonds. in most cases, conformers interconvert at room temperature; thus, they are not different compounds and not true isomers.
Diastereomers
stereoisomers that are not mirror images.
Enantiomers
a pair of nonsuperimposable mirror-image molecules: mirror-image isomers.
Isomers
different compounds with the same molecular formula.
Internal mirror plane (σ)
a plane of symmetry through the middle of a molecule, dividing the molecule into two mirror-image halves.
T/F: A molecule with an internal mirror plane of symmetry cannot be chiral.
TRUE
Dextrorotatory (+) or (d)
rotating the plane of polarized light clockwise.
Levorotatory (-) or (l)
rotating the plane of polarized light counterclockwise.
Mesocompound
an achiral (optically compound that contains chirality centers (usually asymmetric carbon atoms).