drug design and toxicity?
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
What are the roles and contributions to binding energy
Charge interactions (salt bridges)
H-bond donors and acceptors (HBD/HBA)
Charge interactions (salt bridges): Strong electrostatic attraction; high binding energy.
H-bond donors/acceptors (HBD/HBA): Directional bonds; moderate binding energy; increase specificity.
What are the roles and contributions to binding energy of:
Aromatic stacking
Hydrophobic interactions
Aromatic stacking: π–π interactions between rings; moderate contribution; stabilizes positioning.
Hydrophobic interactions: Nonpolar groups cluster to avoid water; major contribution; entropy-driven.
What is the pharmacophore concept and its significance in drug design?
Defines the relative positions of key groups involved in binding.
Derived from an active conformation of the molecule.
Enables lead hopping: moving from one chemical structure to a different one while retaining activity.
what are the different concepts for modifying molecules ?
-simplification concept
-rigidification
What is the Simplification Concept in drug design and why is it useful?
Natural lead compounds are often complex and hard to synthesize.
Simplifying makes analogues easier, quicker, and cheaper to make.
Simpler structures may fit binding sites better and increase activity.
Removing excess functional groups can improve selectivity and reduce toxicity.
oversimplification may result in
decreased activity + selectivity
simpler molecules have more ... and are more likely to .....
conformation
more likely to interact with more than 1 target binding site
What is rigidification in drug design and its effects?
Flexible endogenous leads (e.g., adrenaline) can bind multiple targets.
Rigidify to limit conformations (conformational restraint).
Increases activity (more chance of desired conformation).
Increases selectivity (less chance of undesired conformation).
May make molecule more complex and harder to synthesize.
pharmacokinetic drug design looks at approaches to affecting the ... and ...
-absorption of a drug
-coping with metabolism/ isosteres/bioisosteres
What are key solubility and polarity requirements for drugs?
Polar: soluble in water, interact with targets.
Lipophilic (“fatty”): cross membranes, avoid rapid excretion.
Balanced: drugs need both hydrophilic and lipophilic properties.
How can drug permeability be modified, what are the disadvantages, and what methods are used?
Modification: Vary alkyl group size → larger groups increase hydrophobicity.
Disadvantage: May interfere with target binding (steric hindrance).
Methods:
Change alkyls on heteroatoms → feasible.
Change alkyls on carbon skeleton → difficult, often requires full synthesis.
Why is increased hydrophobicity beneficial for drugs?
Improves membrane permeability=which are largely lipid (fatty) in nature.
Reduces rapid excretion=increasing bioavibilty
Must be balanced with aqueous solubility=so can still dissolve in aqeous envrioenemnt
What is the rationale and disadvantage of masking or removing polar groups in drug design?
Rationale: decreases polarity → increases hydrophobicity
Disadvantages:
Polar group may be needed for target binding
Unnecessary polar groups often already removed during simplification
What is the effect and rationale of adding polar groups to a drug?
Effect: increases polarity, decreases hydrophobicity
Rationale / Uses:
Reduce gut absorption (useful for gut-targeted drugs)
Limit CNS side effects
Limited by gut transport time
How does modifying the pKa of a drug affect its absorption?
Varying pKa changes the percentage of ionised drug.
Ionised drugs are generally poorly absorbed and too water-soluble.
How can pKa be modified in amines to influence drug properties?
Modify alkyl substituents on amine nitrogen (electron-withdrawing groups decrease basicity).
Modify aryl substituents on aromatic amines (electron-withdrawing groups make the amine less basic).
How can metabolic stability of a drug be improved?
Introduce bulky groups as a steric shield.
Protect susceptible functional groups (e.g., esters) from hydrolysis.
Hinders attack by nucleophiles or metabolizing enzymes.
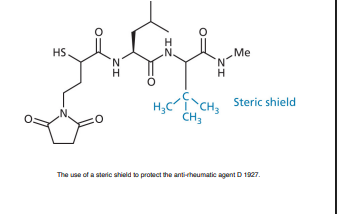
How are steric and electronic effects combined to improve drug stability?
Steric shielding + electronic stabilization used together.
Increases both chemical and metabolic stability of the molecule.
What are isosteres in drug design?
Rational variation of a functional group.
Maintain similar size, shape, and electronic properties.
What are bioisosteres and why are they used in drug design?
Replace a susceptible group with a different group without affecting activity.
Can improve pharmacokinetic properties.
Not necessarily structurally identical to the original group; may look quite different.
How can metabolism of drugs be blocked to increase stability?
Drugs are metabolized at specific susceptible sites.
Introduce blocking groups at these sites.
Increases metabolic stability and drug lifetime.
How can labile groups in drugs be replaced to improve metabolic stability?
Metabolism often occurs at specific susceptible groups.
Remove or replace these groups with metabolically stable groups
Alternatively, move the metabolic target to a less critical position.
What is a pro-drug and how is it activated?
Inactive compound converted to an active drug in the body.
Activation often occurs via metabolic or mechanistic events.
What are the main uses of pro-drugs in drug design?
Improve membrane permeability
Prolong activity
Mask toxicity and side effects
Adjust water solubility
Enable drug targeting
Improve chemical stability
How are carboxylic acids masked in pro-drugs, and what are key considerations?
Mask polar/ionisable carboxylic acids.
Hydrolysed in blood by esterases to release active drug.
Carboxylic acid required for target binding.
Leaving group (alcohol) should be non-toxic.
Example: Enalapril → Enalaprilate (antihypertensive).
What is the rationale behind anti-drugs?
Designed to decrease metabolic stability and shorten drug lifetime.
Useful for drugs that linger too long and cause side effects.
How are anti-drugs designed and what is an example?
Add groups susceptible to Phase I or Phase II metabolism.
Example: Anti-arthritic agents with shortened in vivo lifetime.
How does the diazepam prodrug LDZ reduce side effects?
LDZ is a prodrug of diazepam.
Avoids high initial plasma levels.
Reduces drowsiness side effects associated with regular diazepam.