water of crystallisaton Calculations
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
The Molar Mass of Hydrated Salts:
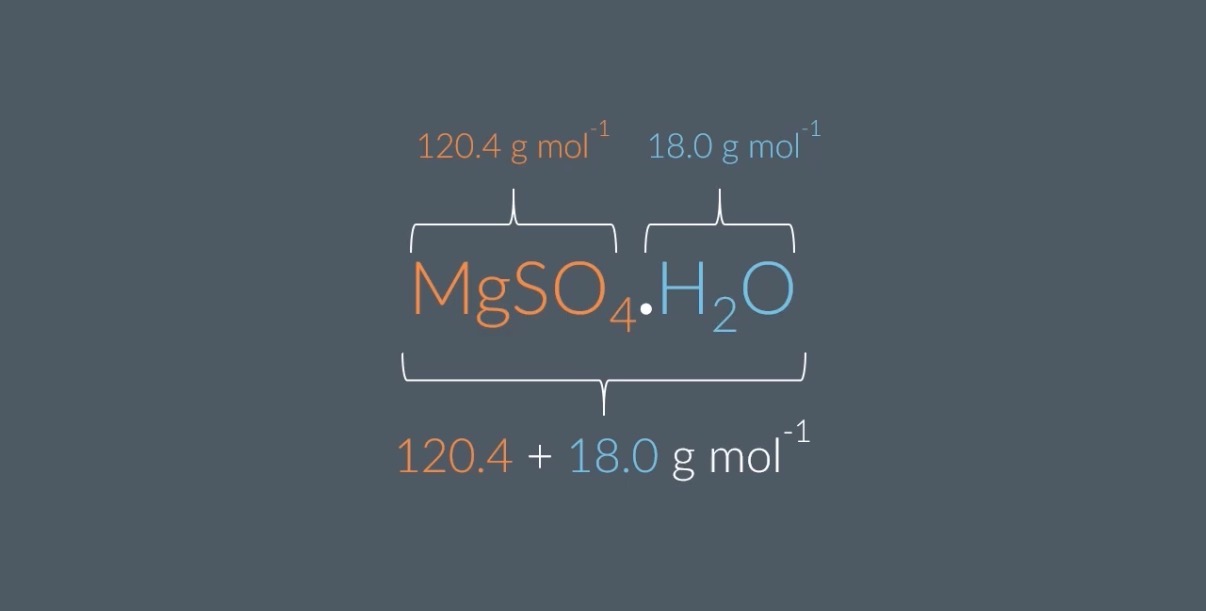
What is the molar mass of MgSO4?
Give your answer to 1 decimal place.
=g mol−1
note:Magnesium (Mg): 24.3
• Sulfur (S): 32.1
• Oxygen (O): 16.0
What is the molar mass of H2O?
Give your answer to 1 decimal placeWhat is the molar mass of MgSO4.7H2O?
The molar mass of MgSO4=120.4g mol−1 and the molar mass of H2O=18.0g mol−1
A: 120.4+4(18.0)g mol−1
B: 7(120.4)+18.0g mol−1
C: 120.4+18.0g mol−1
D: 120.4+7(18.0)g mol−1
What is the molar mass of CuCl2.2H2O?
Give your answer as a number to one decimal place.
note:• Copper (Cu): 63.5
• Chlorine (Cl): 35.5
• Hydrogen (H): 1.0
• Oxygen (O): 16.0
In terms of x, what is the molar mass of MgSO4.xH2O?
The molar mass of MgSO4=120.4g mol−1 and the molar mass of H2O=18.0g mol−1
Correct answersYour answers
A: 120.4+x(18.0)g mol−1
B: x(120.4)+18.0g mol−1
C: 120.4+18.0+x g mol−1
D: 120.4+18.0g mol−1
The molar mass of CaCl2.xH2O is equal to...
A: molar mass of CaCl2+x(18.0)
B: molar mass of CaCl2+18.0
C: molar mass of CaCl2+x
D: molar mass of CaCl2+18.0+x
120.4 g mol-1. 2. 18.0 g mol-1
3.D
170.5 g mol-1 (Explanation
Molar mass of CuCl2=134.5g mol−1
Molar mass of H2O=18.0g mol−1
Molar mass of CuCl2.2H2O=134.5+2(18.0)= 170.5g mol−15.A. 6.A
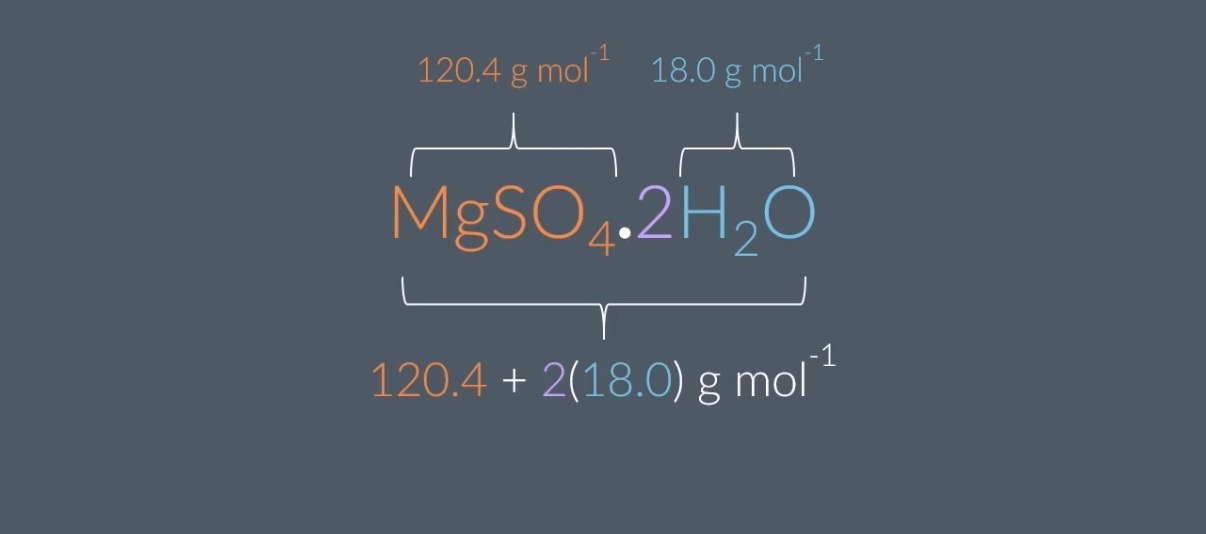
Equations Involving Hydrated Salts
We can turn a hydrated salt into an anhydrous salt by…. (This is decomposition)
Correct answersYour answers
A: heating it gently
B: heating it vigorously
C: dissolving it in water
D: adding a drying agent
Complete the equation for the decomposition of CuCl2.2H2O (s).
Only include the products and state symbols in your answer
Dissociation
Write the complete equation for the dissociation(dissolving in water to seperate the water ions and CuCl2 ions) of CuCl2.2H2O (s).
Include state symbols in your answer
Formation
4.Which of the following reactions will form a salt…
A: Acid + metal
B: acid + metal oxide
C: acid + metal carbonate
We represent forming CuCl2.2H2O (s) from CuCO3(s)and HCl (aq) like…
Correct answersYour answers
A: CuCO3(s)+2HCl (aq)→CuCl2.2H2O (s)+…
B: CuCO3(s)+HCl (aq)→CuCl2.2H2O (s)+…
C: CuCl2.2H2O (s)→CuCO3(s)+2HCl (aq)+…
D: CuCl2.2H2O (s)→CuCO3(s)+HCl (aq)+…
note: We just make sure the ions which form the salt are balanced, rather than focusing on balancing the whole thing.
B (Gentle heating is not usually enough to remove the water of crystallisation)
CuCl2(s)+2H2O(g)
CuCl2∙2H2O(s)+aq→ CuCl2(aq) + 2H2O(l)
(note- The aq represents the dissociating in water)
A,B,C (A-salt+hydrogen. B-Salt+water. C-Salt+water+CO2)
A(The Chlorines and coppers are balanced on each side)


Answer image q?
•At a level, there are 3 common types of reaction that contain hydrated salts which are…2?
Decomposition, dissociation, formation

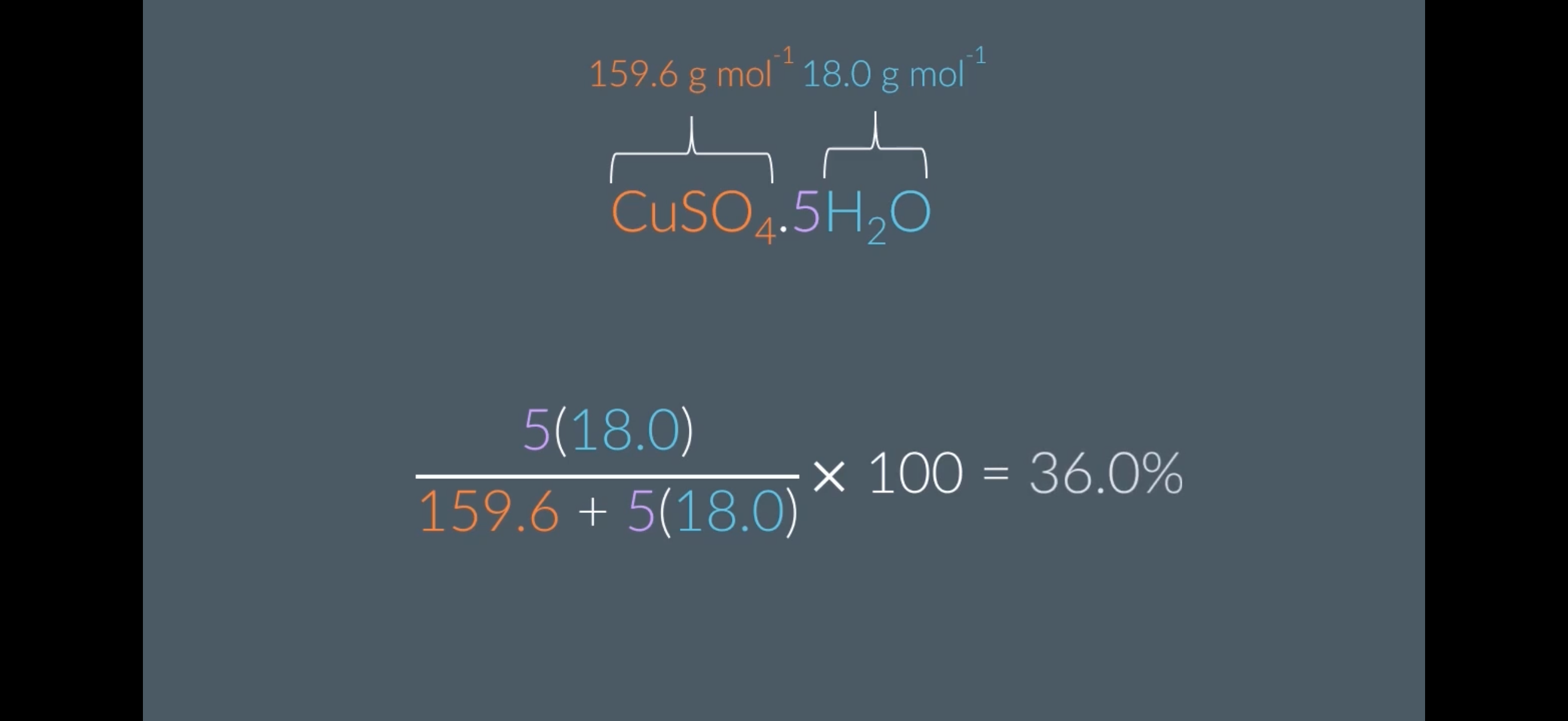
Calculating % Water Content
The molar mass of CuSO4 is…
Give your answer to 1 decimal place
The molar mass of H2O is…
Give your answer to 1 decimal place
Note:Copper (Cu): 63.5
• Sulfur (S): 32.1
• Oxygen (O): 16.0
•Hydrogen (H): 1.0
(Image shows example of the % water content)
2.The percentage water content (by mass) of Na2CO3.10H2O is…
Note: • Sodium (\text{Na}): 23.0 \text{g mol}^{-1}
• Carbon: 12.0 {g mol}^{-1}
• Oxygen : 16.0 {g mol}^{-1}
• Hydrogen ): 1.0{g mol}^{-1}
Give your answer to 1 decimal place
Answer the image question that’s in the other side?
The molar mass of CUSO4 is 159.6 g mol-1
molar mass of H2O is 18.0 g mol-1
62.9%
A

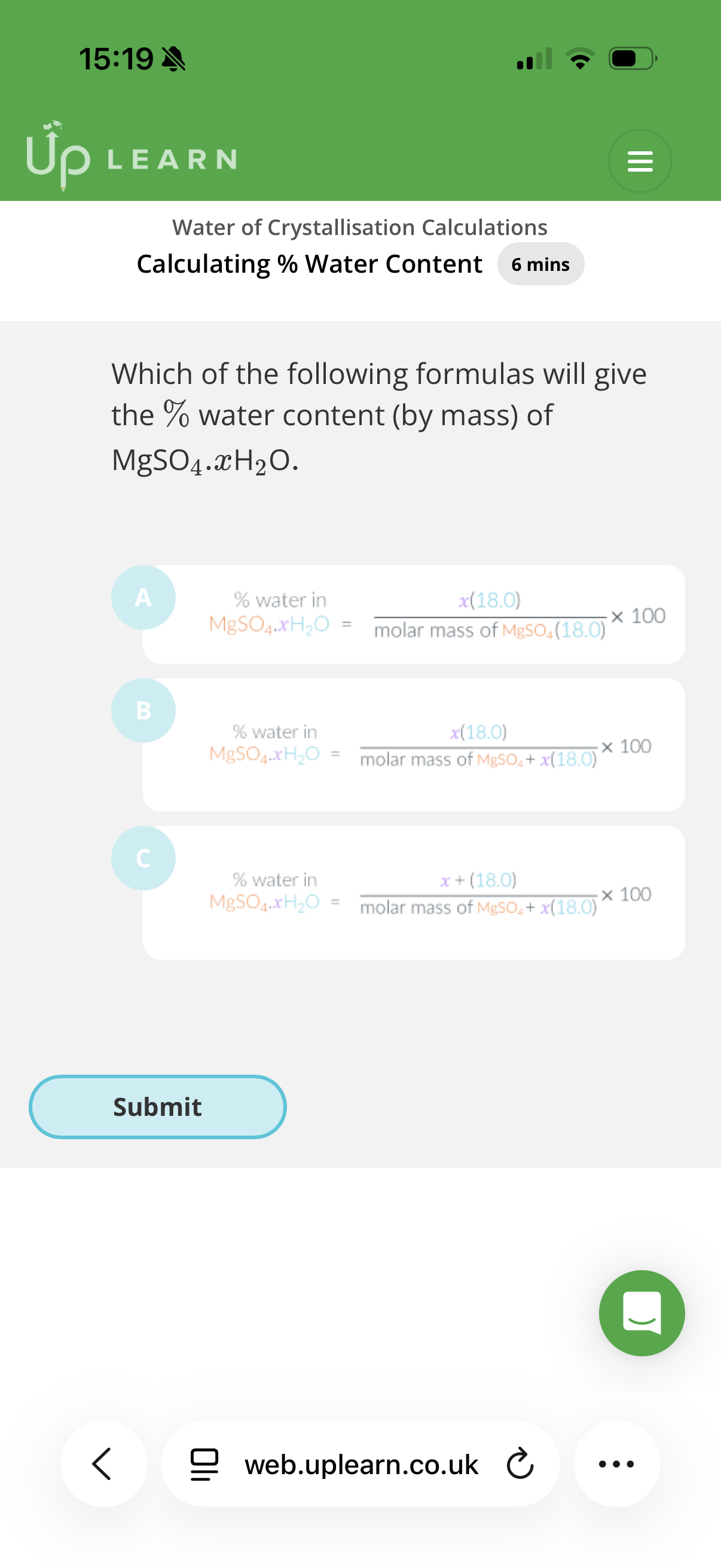
Answer image q?
B
Solving For x Using Molar Mass
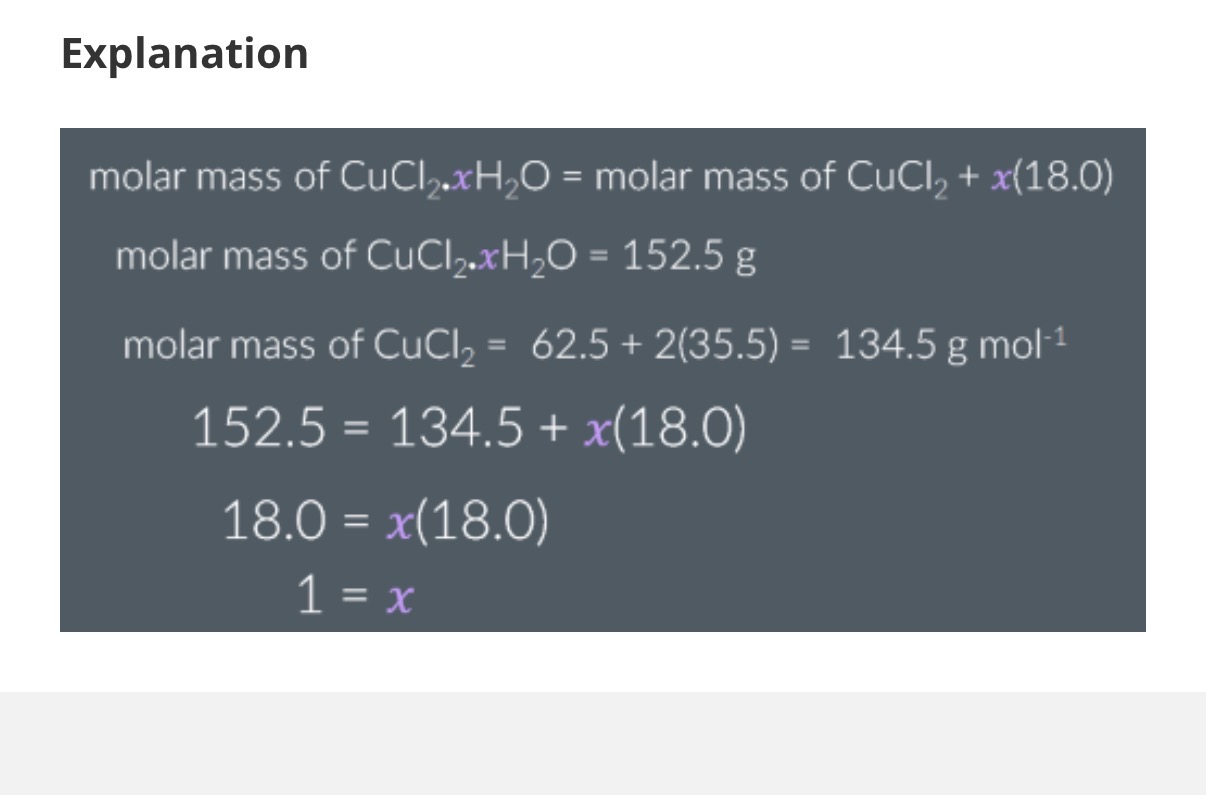
1.The molar mass of CuCl2.xH2O is 152.5g mol−1. What is the value of x?
Note: First, we use standard atomic masses:
• Copper (Cu): 63.5
• Chlorine (Cl): 35.5
• Hydrogen (H): 1.0
• Oxygen (O): 16.0
Give your answer to the nearest whole number. (Answer is in other side image)
2.The molar mass of hydrated sodium phosphate(PO4*3-) is 380.0g mol−1. Calculate the number of moles of water of crystallisation per mole of salt.
Atomic Masses
• Sodium (Na): 23.0
• Phosphorus (P): 31.0
• Oxygen (O): 16.0
• Hydrogen (H): 1.0
Give your answer to the nearest whole number
X=12

Solving For x Using % Water Content
Note: the image is an example of the calculation.
Sodium carbonate can occur as a hydrated compound, Na2CO3.xH2O.
Analysis of this hydrated compound showed that it contained 62.9% by mass of water.
Determine the value of x.Individual Atomic Masses (A_r)
• Sodium (Na): 23.0
• Carbon (C): 12.0
• Oxygen (O): 16.0
Give your answer to the nearest whole number
10 (calculation in image)
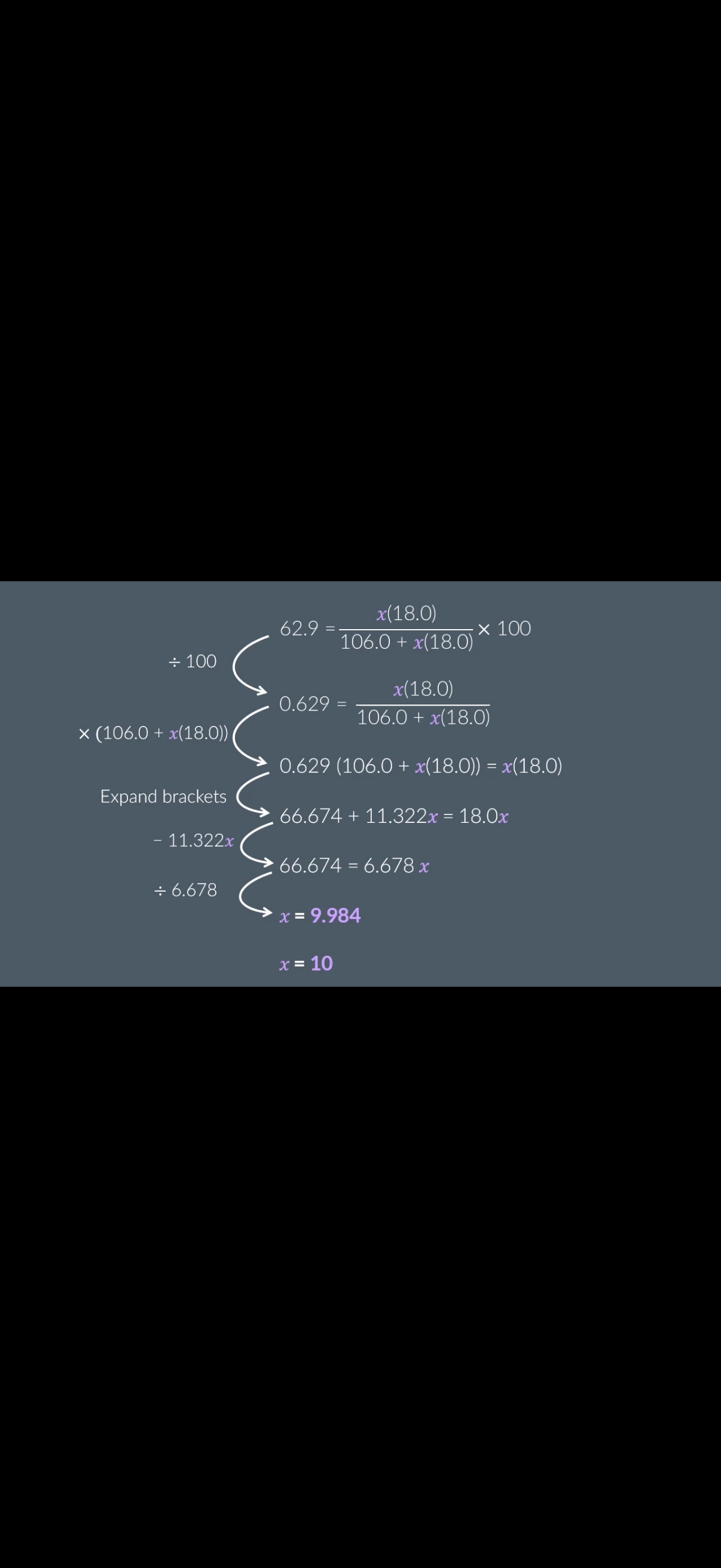

Finding x Using Experiments
Answer image q?
If we have a sample of CuCl2.xH2O with a known mass, we can find x by...
A: heating the sample vigorously and measuring its mass afterwards
B: dissolving the sample in water and measuring its mass afterwards
C: adding a small volume of water to the sample and measuring its mass after
D: reacting the sample with an acid and measuring the volume of gas produced.
3.If we have a sample of CuCl2.xH2O with a known mass, we can find x by heating the sample vigorously (to remove the water of crystallisation) and measuring its mass afterwards.
During this experiment, we make sure all the water of crystallisation has escaped by...
A: heating the sample at 1000°C
B: adding a drying agent to the crucible
C: heating the sample until its mass stops changing
D: performing the experiment in a arid climate
A. 3.C


Answer image q?

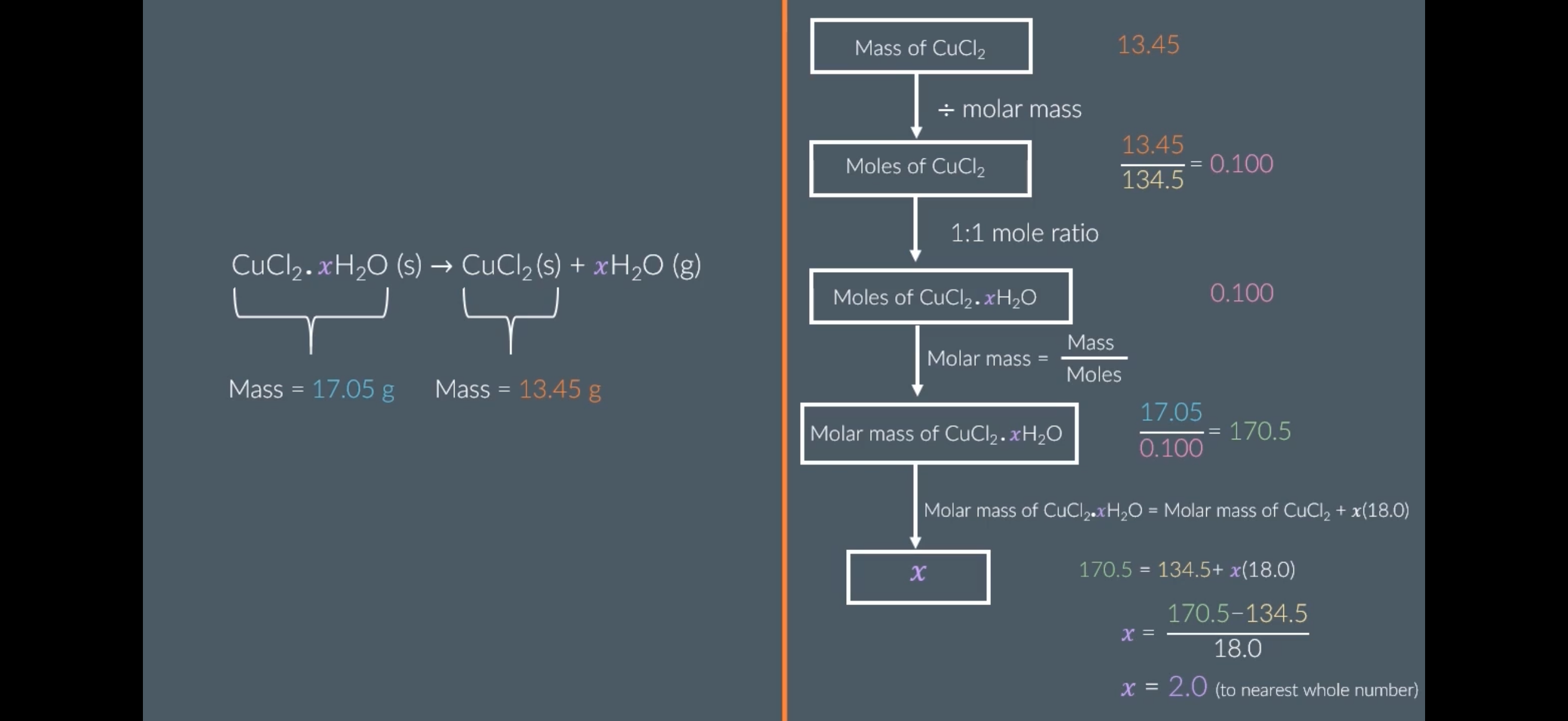
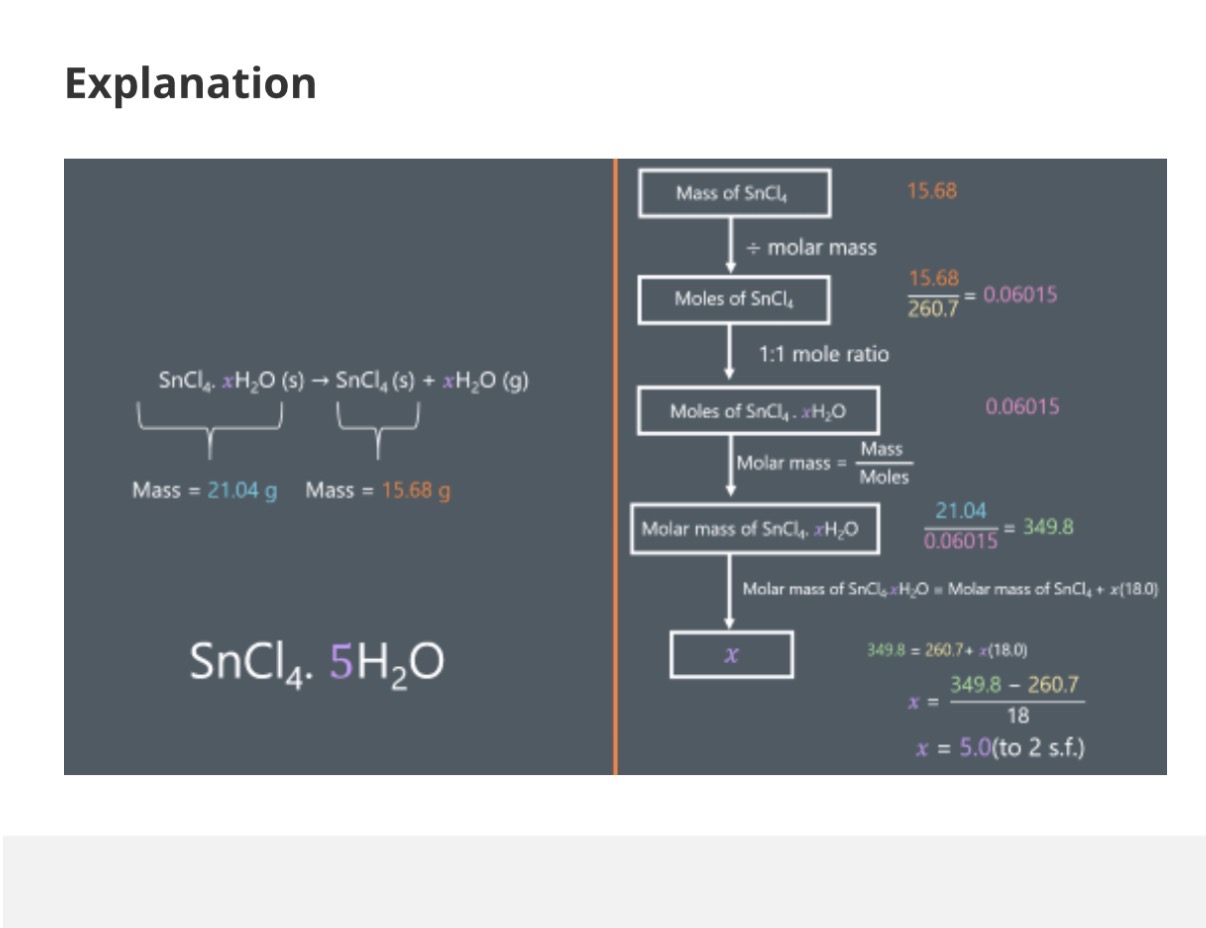
Solving For x Using Experiments:
Image: The image shows an example of calculating the moles by using experimental values.
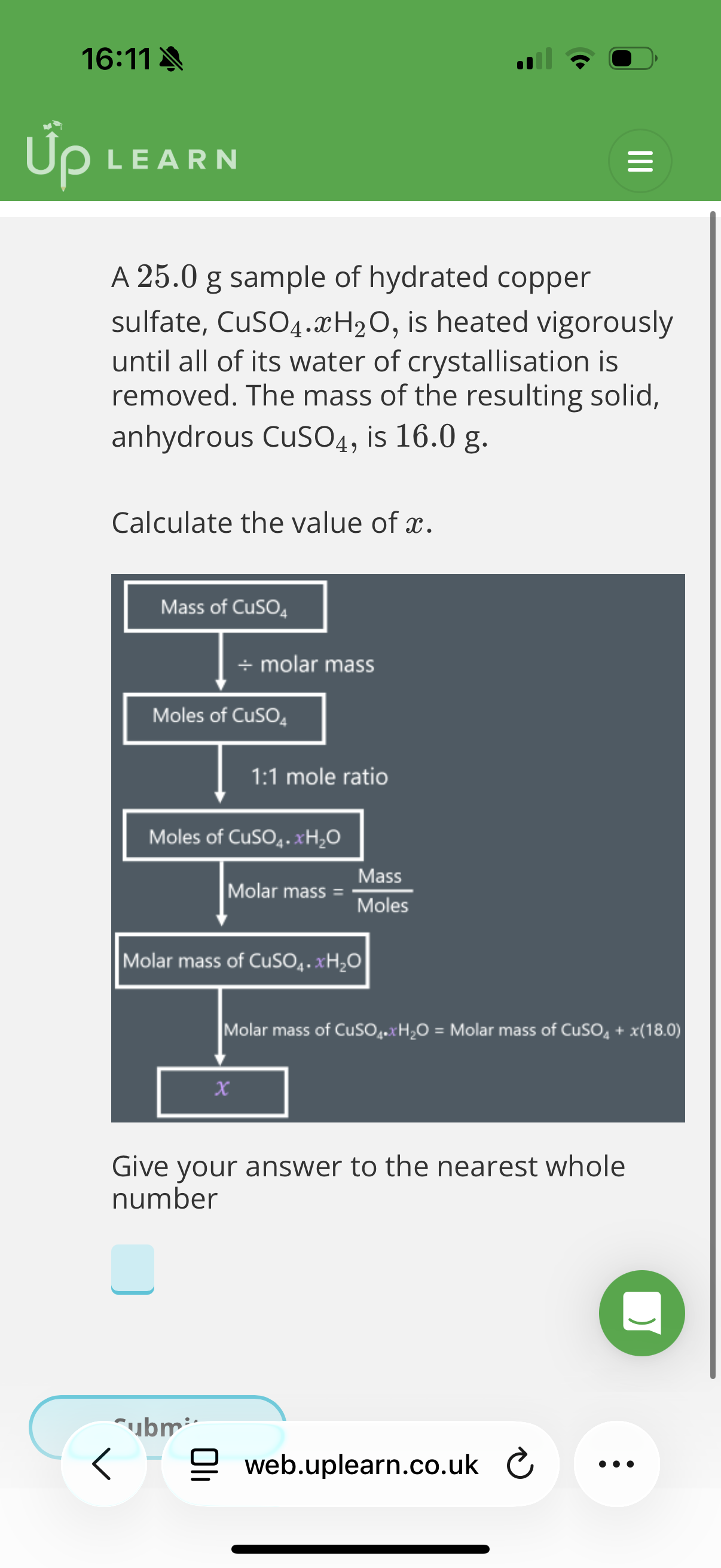
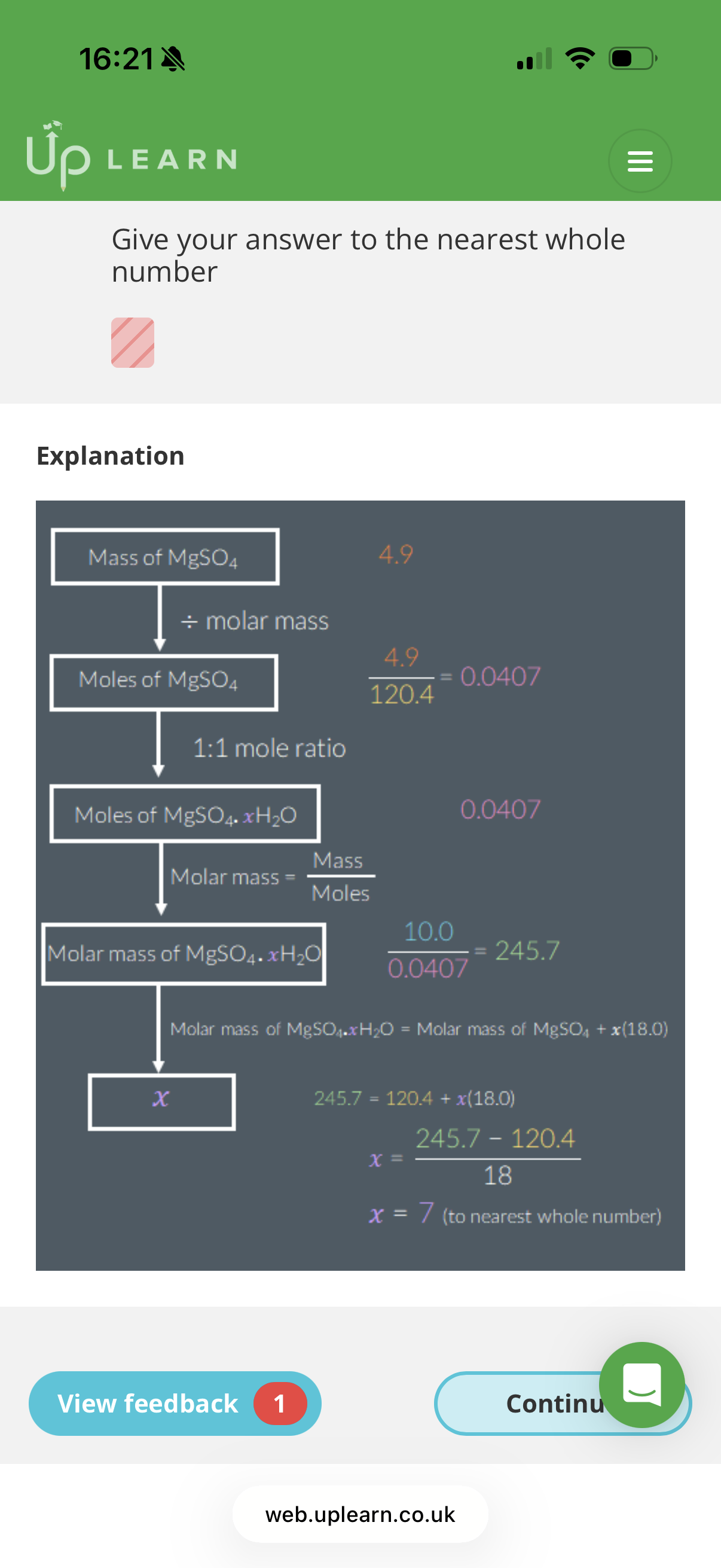
Answer image question?
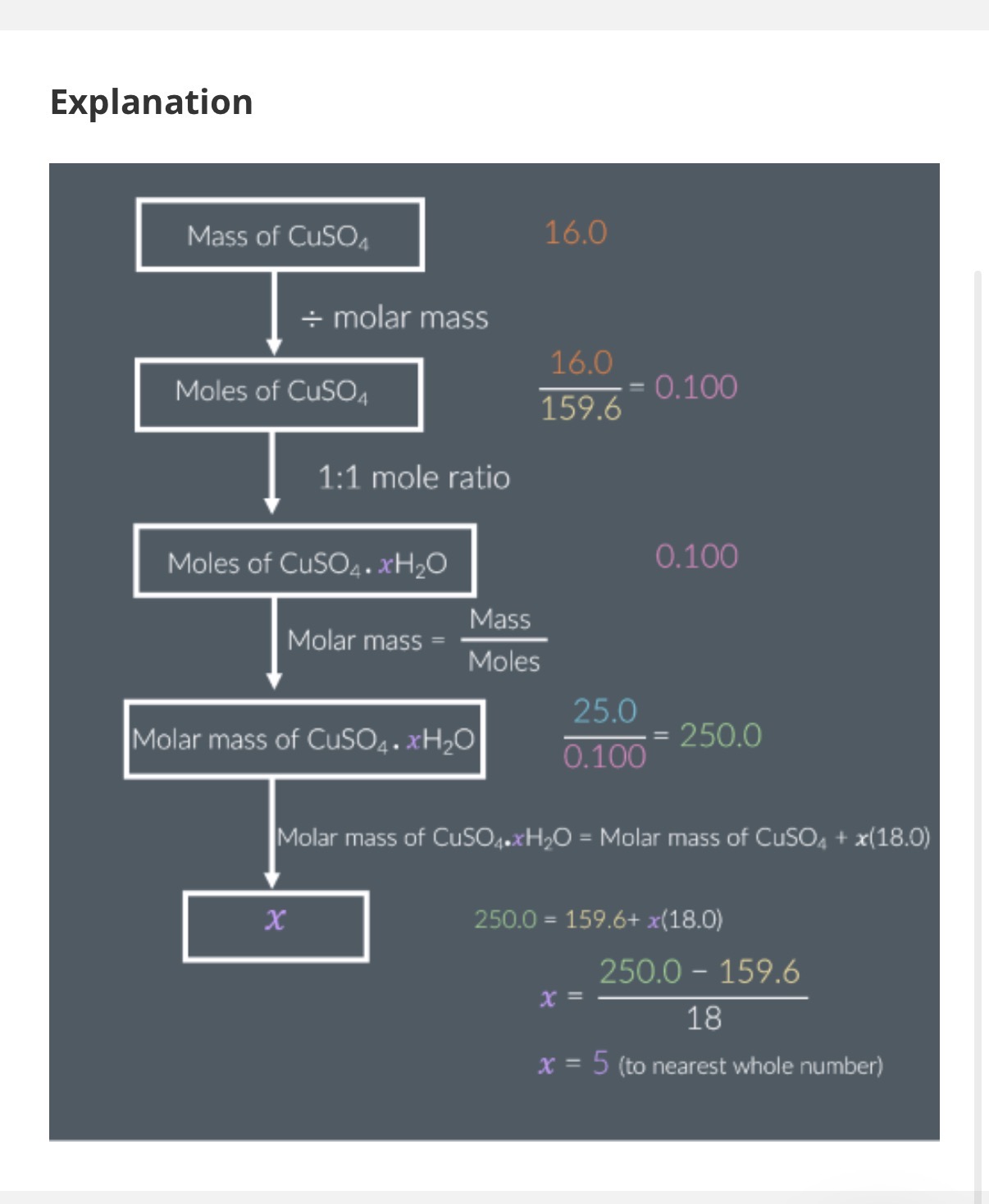
A 10.0g sample of hydrated magnesium sulfate, MgSO4.xH2O, is heated vigorously until all of its mass stops changing. The mass of the resulting solid is 4.9g.
Calculate the value of x.
Ar: Mg-24.31 S:32.06 O: 16.00
Give your answer to the nearest whole number
Does x Have to be a Whole Number?
Jas performs an experiment heating hydrated copper sulfate, but fails to heat the crystals enough to remove all the water of crystallisation. This would mean the measurement he uses for...
Correct answersYour answers
A: the mass of anhydrous copper sulfate is too high
B: the mass of hydrated copper sulfate is too high
C: the mass of anhydrous copper sulfate is too low
D: the mass of hydrated copper sulfate is too low
What experimental errors could lead to an incorrect value for x.
Select all that apply
A: The hydrated crystals weren’t heated to remove all the water of crystallisation
B: The hydrated crystals lost some water to the surroundings before they were weighed
C: The anhydrous crystals formed after heating absorbed some water from the surroundings before they were weighed
D: The resultant anhydrous crystals lost some water to the surroundings before they were weighed
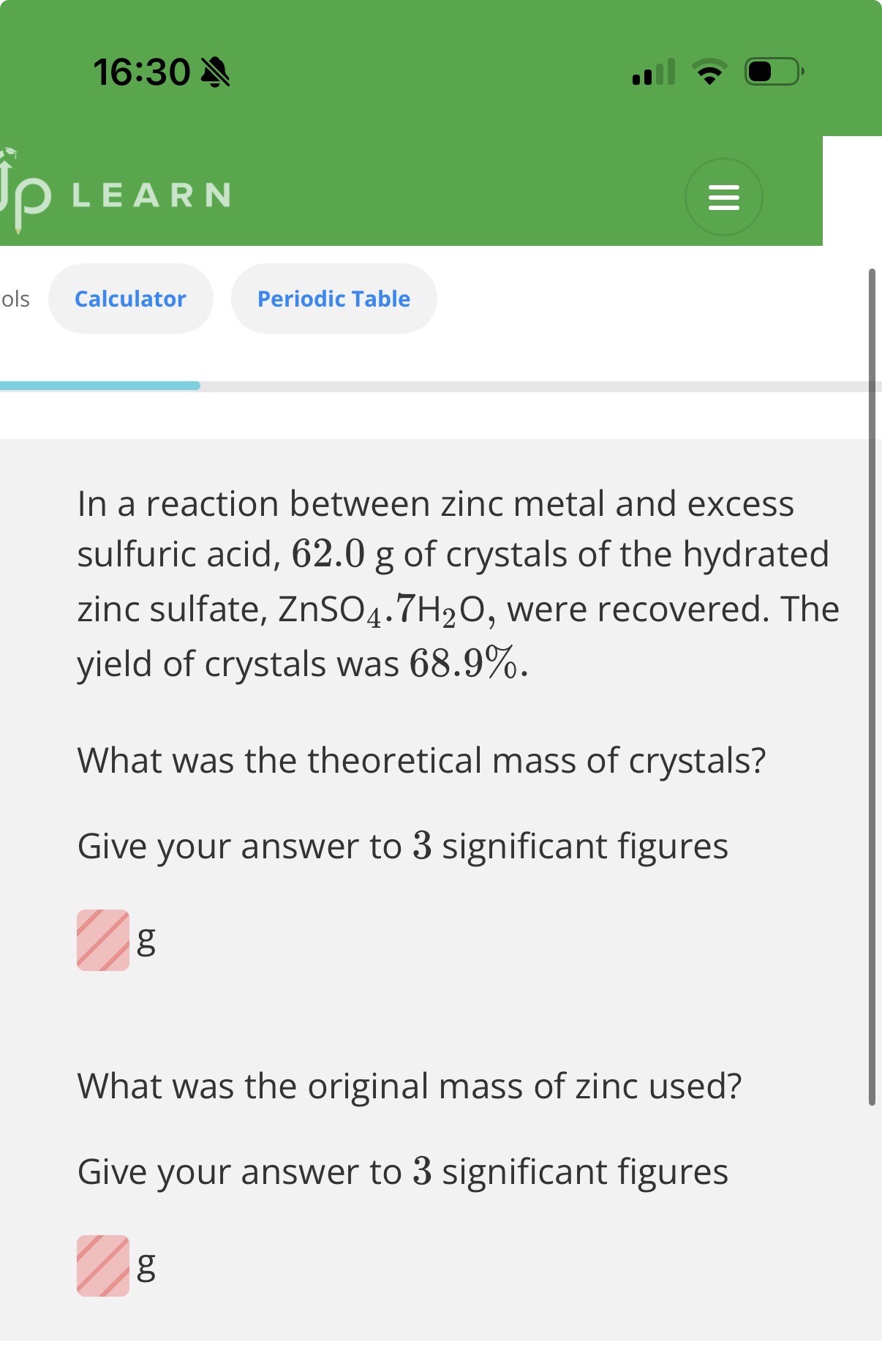
3.A lab technician left a 10.0g sample of hydrated copper sulfate, CuSO4.xH2O in a beaker overnight. The day after she reweighed the sample and found it had a mass of 9.8g.
This change in mass suggests that, over the course of the night, the value of xhas...
Correct answersYour answers
A: decreased, because water of crystallisation is lost to the environment.
B: decreased, because copper sulfate is lost to the environment.
Answer image q?
A. 2.A,B,C. 3.A(Explanation
The mass of hydrated copper sulfate has decreased overnight. This suggests that it has lost some water of crystallisation to the surroundings. Since there are now fewer moles of water of crystallisation in the hydrated salt, the value of x will decrease
90g then 20.5g

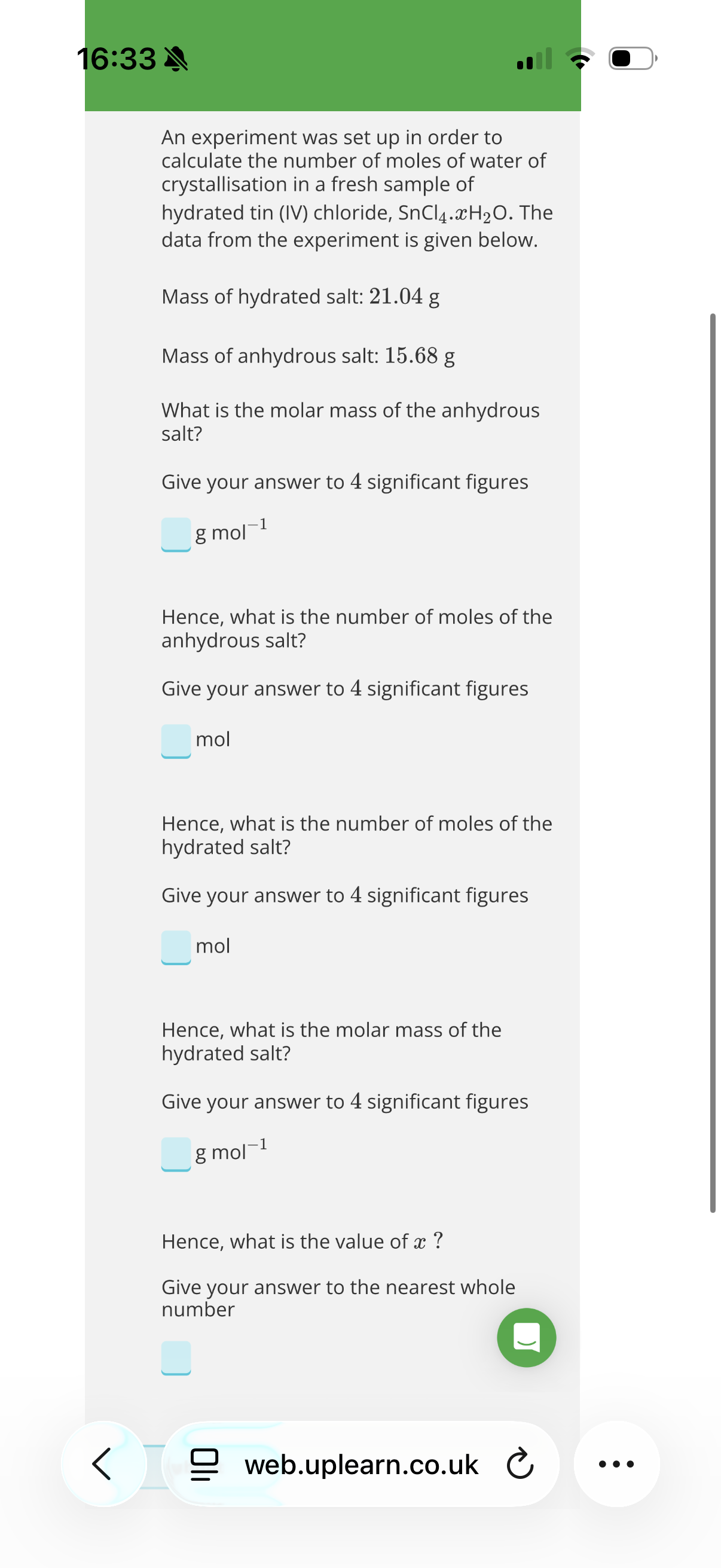
Answer image w?
What would have happened to x if the sample of hydrated salt, SnCl4.xH2O, had been left out overnight before doing the analysis to determine the amount of water of crystallisation in the sample?
Correct answersYour answers
A: x would have increased because the hydrated salt would gain water from the environment.
B: x would have decreased because the hydrated salt would lose water to the environment.
2.B