Chemistry
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
Polyethene uses and properties
Properties:
flexible
cheap
electrically insulating
Uses:
plastic bags
plastic bottles
Polypropene uses and properties
Properties:
flexible
strong
Uses:
food packaging
ropes
carpets
Polychloroethene (PVC) uses and properties
Properties:
long-lasting
tough
cheap
Uses:
plastic sheets
artificial leather
drainpipes and gutters
insulation on wires
PTFE (polytetrafluoroethylene) uses and properties
Properties:
very tough
non stick
resistant to high temperatures
Uses:
cookware
pipes
Are nitrates soluble? Exceptions?
All nitrates are soluble
Which three elements are always soluble in compounds
Sodium, potassium, ammonium
Are chlorides soluble? Exceptions?
Most chlorides are soluble apart from silver and lead (II)
Are sulfates soluble? Exceptions?
Most sulfates are soluble apart from barium, calcium and lead (II)
Are carbonates soluble? Exceptions?
Most carbonates are insoluble apart from SPA
Are hydroxides soluble? Exceptions?
Most hydroxides are insoluble apart from SPA
Sulfate ion
SO₄²⁻
Nitrate ion
NO₃⁻
Carbonate ion
CO₃²⁻
Recycling advantages and disadvantages
Advantages:
reduces need for mining and quarrying which damages habitats
uses less energy than extracting new materials
reduces how quickly non-renewable resources are used up
Disadvantages:
difficult to collect and process materials to be recycled
limits product e.g. can only be certain colours
(expensive)
Describe haber process
Stage 1: H2 and N2 are obtained from natural gas and the air respectively and are pumped into the compressor through pipe
Stage 2: the gases are compressed to about 200 atmospheres inside the compressor
Stage 3: the pressurised gases are pumped into a tank containing layers of catalytic iron beds at a temperature of 450°C. Some of the hydrogen and nitrogen react to form ammonia:
N2 (g) + 3H2 (g) ⇌ 2NH3 (g)
Stage 4: unreacted H2 and N2 and product ammonia pass into a cooling tank. The ammonia is liquefied and removed to pressurised storage vessels
Stage 5: the unreacted H2 and N2 gases are recycled back into the system and start over again
Group 1 physical and chemical properties
Physical:
soft
low density
low melting point
reactive
As you go down the group:
density increases
melting point decreases
Noble gases:
uses of helium, argon and neon
properties
as you go down group
Helium is used for filling balloons and weather balloons as it is less dense than air and does not burn.
Argon is used to provide an inert atmosphere for welding and to fill electric light bulbs
neon, argon, xenon in advertising signs
low melting point
As you go down group:
boiling point increases (due to larger molecules)
density increases
Exothermic
definition
types of reaction
uses
In exothermic reactions energy is given out to the surroundings so the temperature of the surroundings increases
Combustion, oxidation, and neutralisation reactions
Hand warmers and self heating food/drink cans
Endothermic
definition
types of reaction
uses
In endothermic reactions energy is taken in from the surroundings so the temperature of the surroundings decreases
-Electrolysis, thermal decomposition reactions and the first stages of photosynthesisice packs
Is bond breaking exothermic or endothermic and does it use more or less energy
Endothermic -
More energy is required to break the bonds than that gained from making the new bonds
has a positive change in energy as products have more energy than reactants
Is bond making exothermic or endothermic and does it use more or less energy
Exothermic - More energy is released when new bonds are formed than energy required to break the bonds in the reactants
So more energy is released than absorbed
Therefore an exothermic reaction has a negative change in energy as products have less energy than reactants
What is the order of crude oil from the bottom and give uses of each one
bitumen - surfacing roads and rooves
fuel oil - ships and power stations
diesel oil - diesel engines
kerosene - jet fuel
petrol - fuels for cars
gases - domestic heating and cooking
As you go down the crude oil tube fractionating collumn what happens to number of atoms in molecules, boiling point, ease of ignition and viscosity
number of atoms in molecules - decreases
boiling point - decreases
ease of ignition - decreases
viscosity - increases
Describe the history of the atmosphere
volcanic activity
Gases produced by volcanoes formed early atmosphere
There was little or no oxygen
Lots of CO2, water vapour and some other gases
The Earth was incredibly hot and molten until some slowly solidified into volcano land massesWater vapour condenses
Gradually, the Earth cooled enough for the water vapor to condense forming oceans
CO2 is a very soluble gas so it dissolved into the oceans lowering levels in the atmosphere
There was still little or no oxygenCyanobacteria
Primitive plants called cyanobacteria grew
Can photosynthesize converting CO2 and water into glucose and O2
Oxygen in atmosphere increases
CO2 in atmosphere decreases
Test for carbonate ion
Add dilute acid and bubble the gas produced through limewater. If the limewater turns cloudy due to calcium carbonate precipitate, then carbonate ions were present
Test for sulfate ion
Add dilute hydrochloric acid and aqueous barium chloride, if present a white precipitate of barium chloride is formed
Test for halide ion
Add dilute nitric acid and silver nitrate solution forming a precipitate of silver halide.
Silver chloride = white
Silver bromide = cream
Silver iodide = yellow
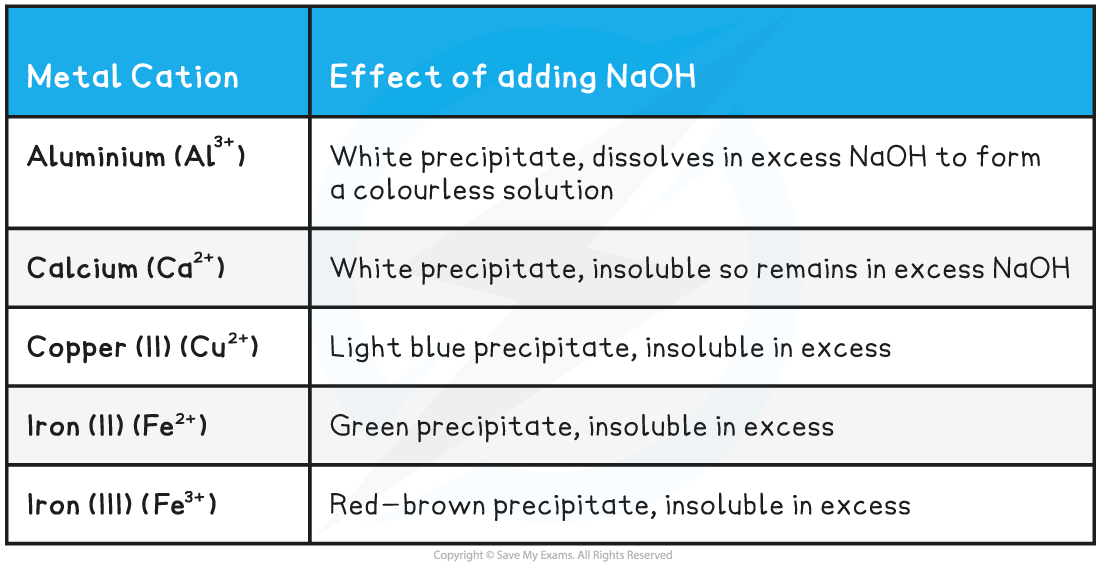
How to test for cations
When in aqueous solution, add sodium hydroxide and the precipitate they form has different colours
What colour precipitate do the ions make