Entropy and Spontaneity
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
Tell me about Order and Disorder in the universe
energy and matter disperse
the universe becomes more disordered over time
natural process without the need to add work
can reverse the process at the expense of energy
we can express the degree of disorder of a system as entropy (S)
Tell me about Entropy (S)
the distribution/ dispersal of total available energy/ matter among particles in a system
Ordered states with small energy distribution/localization have low entropy, and disordered states with more energy distribution have higher entropy
What is the 2nd Law of Thermodynamics in layman’s terms?
As time passes, matter and energy become more disordered so entropy of universe increases
increasing # of particles…
increases opportunity to create disorder
solid → liquid (∆S=?)
∆S=+
solid → gas (∆S=?)
∆S=+
liquid → gas (∆S=?)
∆S=+
liquid → solid (∆S=?)
∆S=-
gas → liquid (∆S=?)
∆S=-
gas → solid (∆S=?)
∆S=-
What is Absolute Entropy?
entropy values tabulated under standard conditions (in data booklet)
(S⁰)
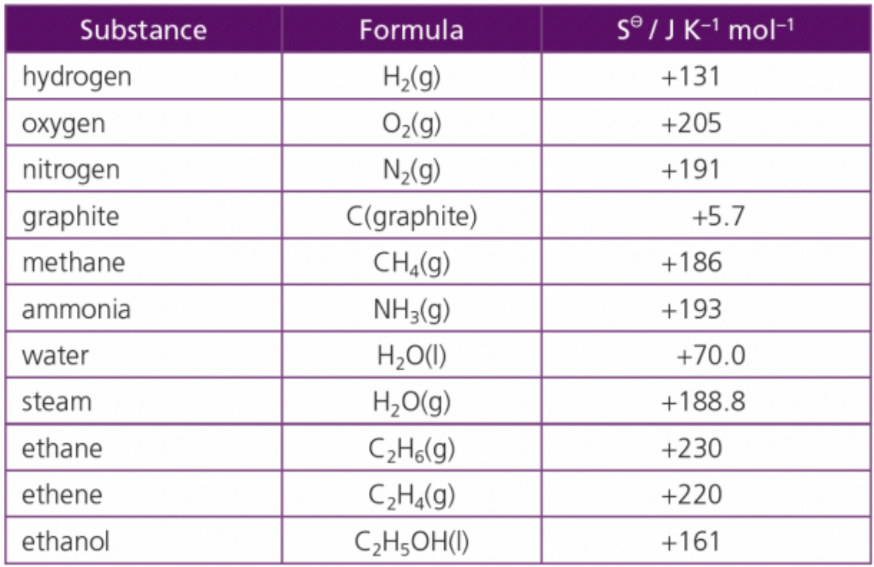
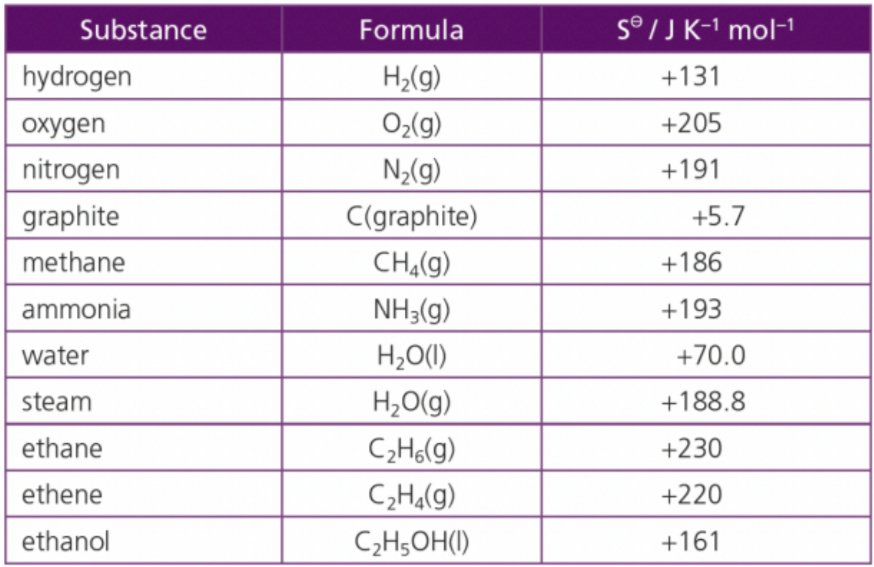
What do you notice about the table?
Entropy increased in gas compared to solids
All states/commands have positive values
perfectly ordered system would have a value of zero
What is the Equation for the Entropy of a Reaction?
∆S°rxn = ∑S(products)-∑S(reactants)
What is the Equation for Total Entropy?
∆S°total = ∑S(system)-∑S(surroundings)
What is important to remember in terms of Entropy of 0? How do you find the entropy of the surroundings? What are the units of entropy?
you need to still consider the entropy of surroundings
J K⁻¹ mol⁻¹
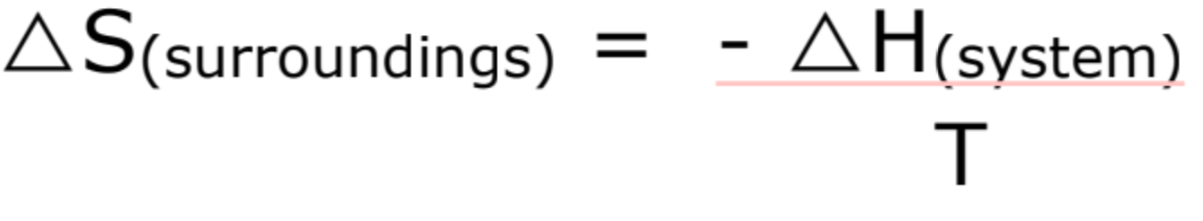
What are the conditions for a spontaneous reaction?
∆Stotal = ∆Ssystem - ∆Ssurroundings > 0
∆Stotal = ∆Ssystem - (∆H ÷ T) > 0
*important to consider system and surroundings for both enthalpy and entropy when determining feasibility of a reaction
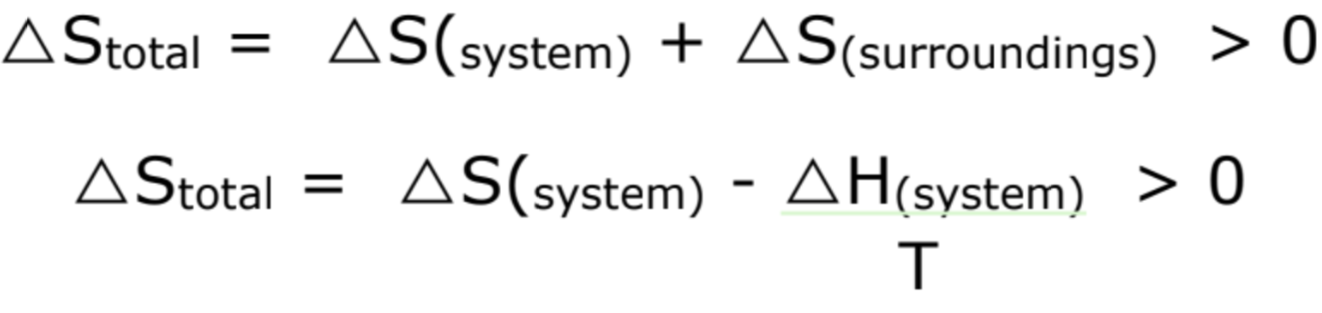
Define Gibb’s Free Energy. What should we know about it? What is its equation
gives a measure of the quality of energy available to do useful work
factors in entropy and enthalpy of a system to help predict spontaneity
denoted by ∆G
must be negative to be spontaneous
△G(system) = △H(system) - T△S(system)
For Gibb’s Free Energy, if the temp. is low then… and what does it mean?
△G(system) = △H(system)
exothermic reactions can occur at low temperatures
For Gibb’s Free Energy, if the temp. is high then… and what does it mean?
△G(system) = -T△S(system)
at high temps., even if the reaction is endothermic, it can still be spontaneous
If a ∆H is (-) and ∆S is (+), what does that mean for ∆G and Spontaneity?
reaction is spontaneous at all temps
If a ∆H is (+) and ∆S is (-), what does that mean for ∆G and Spontaneity?
reaction is never spontaneous
If a ∆H is (-) and ∆S is (-), what does that mean for ∆G and Spontaneity?
at low temps, reaction is spontaneous
at high temps, reaction is not spontaneous
If a ∆H is (+) and ∆S is (+), what does that mean for ∆G and Spontaneity?
at low temps, reaction is not spontaneous
at high temps, reaction is spontaneous
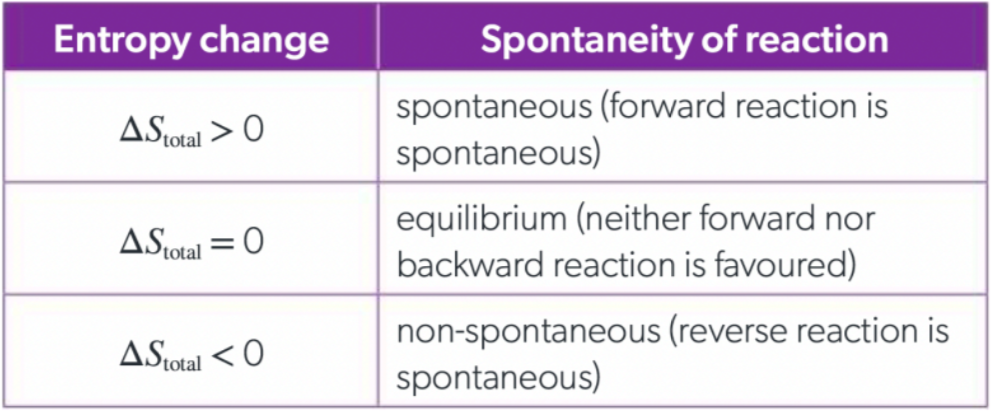
What is the relationship between total entropy change and spontaneity?