Week 11 Lecture
1/33
Earn XP
Description and Tags
silicates
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
34 Terms
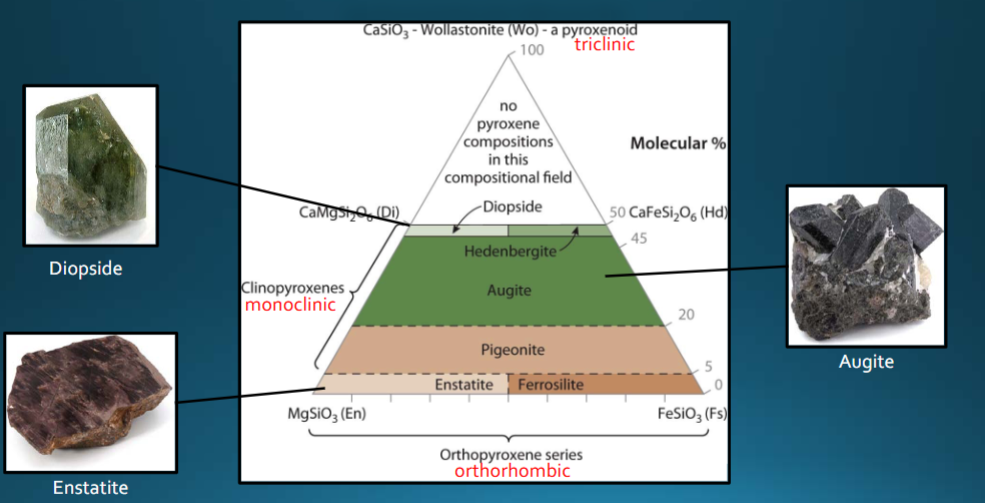
Pyroxenes
single chain silicates. Limited polymerization, include orthorhombic orthopyroxenes (enstatite) and monoclinic clinopyroxenes (augite). Often show exsolution; 1:3 ZO ratio

Orthopyroxene extinction
parallel
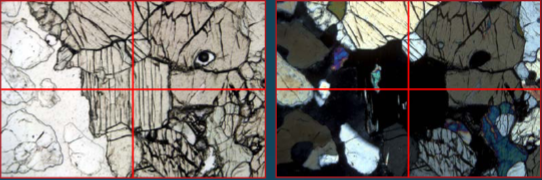
Clinopyroxene extinction
inclined
Pyroxene composition
area above quadrilateral is where exsolution occurs
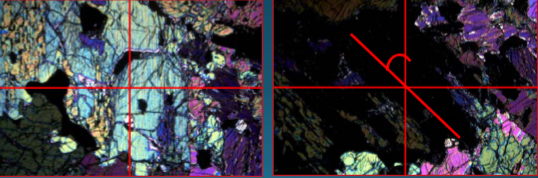
Amphiboles
Double chain silicates; monoclinic clino-amphiboles (hornblende) 4:11 ZO ratio. Common in igneous rocks in subduction zones; have (OH) groups
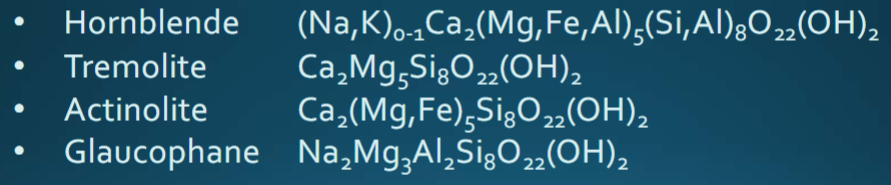
Mica group
Sheet silicates (muscovite)

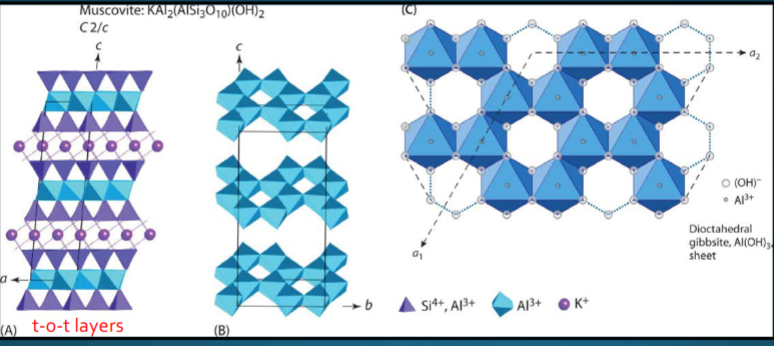
Dioctahedral muscovite (sheet silicates)
one octahedra vacant for every two octahedra that are filled with Al3+. Here, dioctahedral sheet is gibbsite: Al2(OH)6
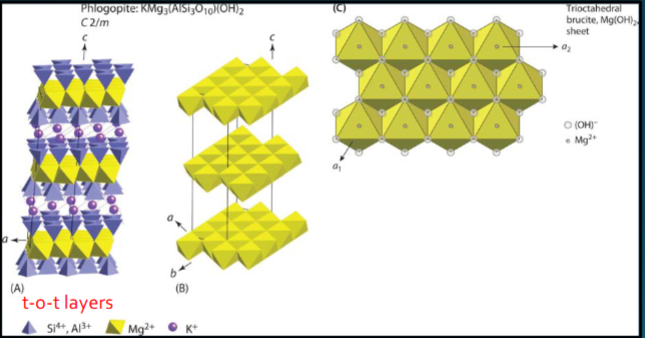
Trioctahedral phlogopite (sheet silicates)
all octahedral occupied and filled with Mg2+. Here, trioctahedral sheet is brucite: Mg(OH)2
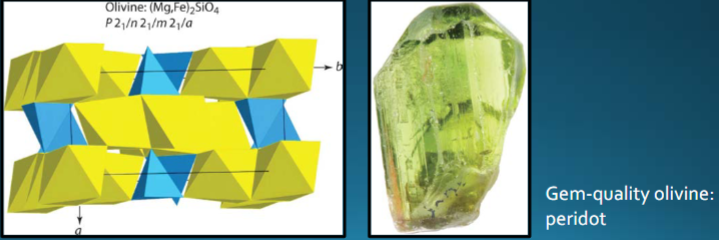
Olivine group
High relief with lack of cleavage. Independent tetrahedra (orthosilicates). Forsterite-fayalite (Mg,Fe)2SiO4. Common in gabbro, basalt, peridotite.
Rock with >90% olivine = dunite
Common accessory minerals in silicates

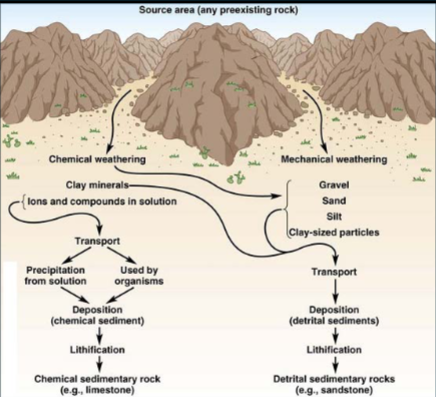
Sediment
Particles derived from the weathering process
Diagenesis
Occurs during and after lithification.
Compaction → cementation → recrystallization (changing of fine-grained rocks into coarser ones)
Dissolution and removal of minerals (leaching) and the formation of clay or other minerals are both common
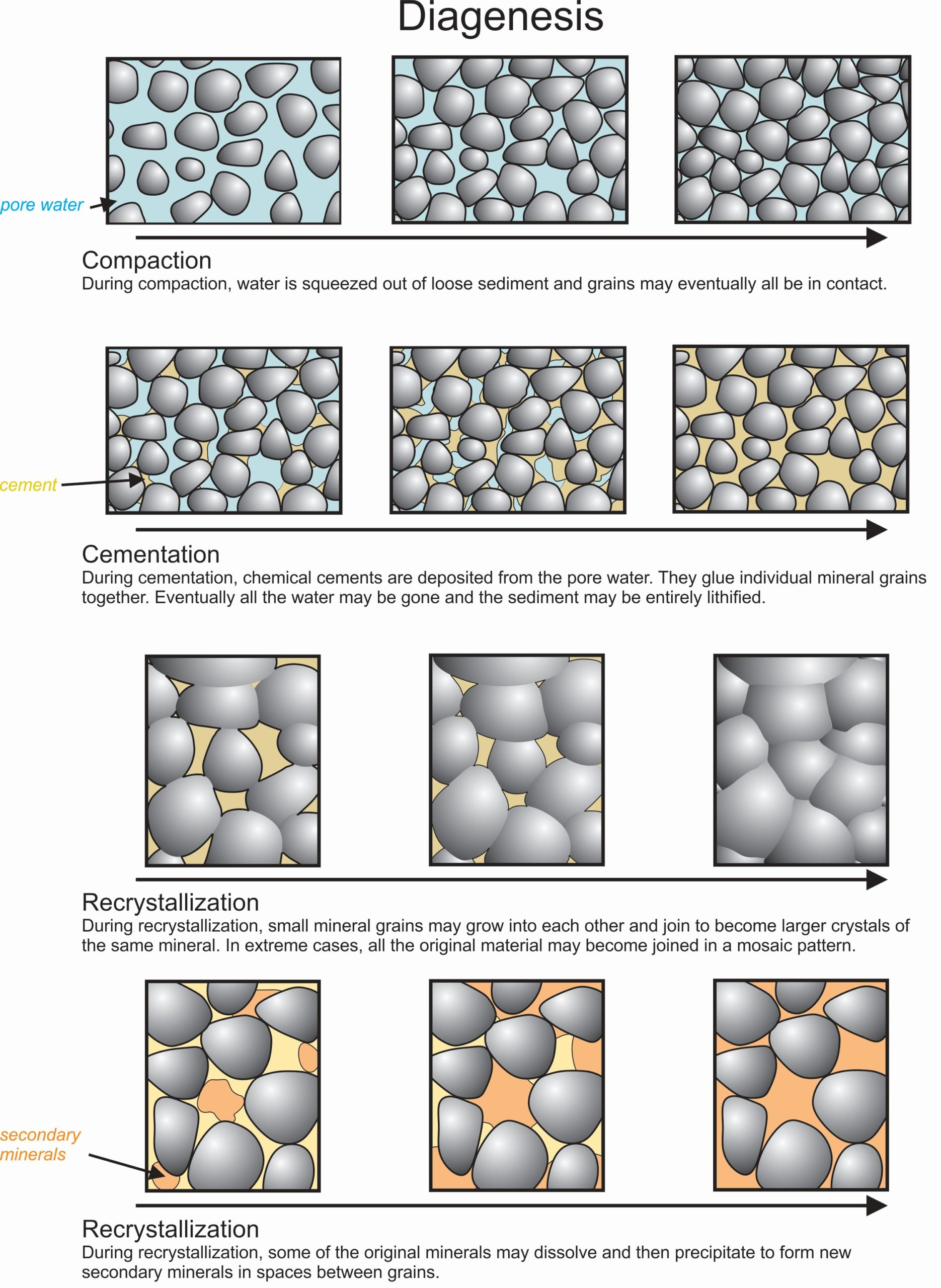
Authigenic minerals
Products of diagenesis (Zeolites, clays, feldspar, pyrite, and quartz)
Mechanical weathering
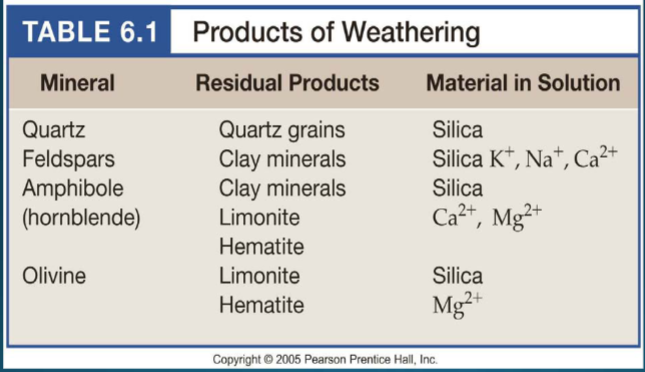
Weathering by physical processes (erosion); produces detrital sediment. Minerals that survive the weathering process (quartz in sandstone)
Chemical weathering
Weathering by chemical reactions (acid rain). produces chemical sediment (clay minerals, oxides, hydroxides)
Precipitation
Minerals that form as a chemical precipitates (halides, carbonates, sulfates)
Why are some minerals more common as detrital minerals?
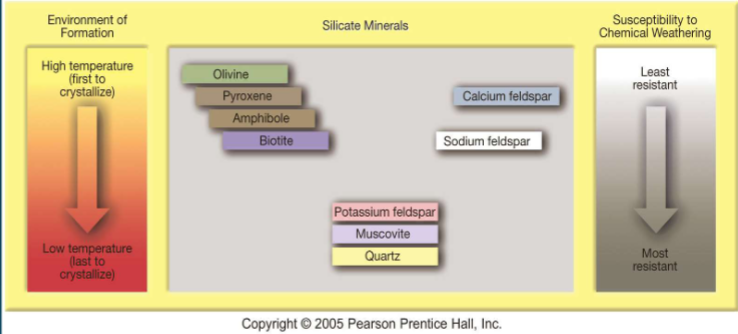
Because of their weathering resistance. Rocks formed further below the earth will be more unstable at the surface due to the high pressures/temps they form at and their unstable nature (olivine)
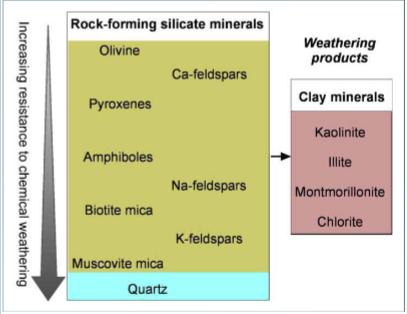
The last minerals to crystallize in Bowen’s reaction series are
the most stable at the surface due to low Gibbs energy. They are most resistant to weathering
Goldich weathering series
inversion of Bowen’s series to describe weathering

Chemical reactions can alter
both the mineralogical and chemical composition of a rock
Hydrolysis
Minerals that react with water, causing new minerals to form. Happens in subsurface ad surface

Oxidation
reaction with oxygen (rusting). Can affect any Fe-bearing mineral. Reactions can form hematite and limonite.

Acid mine drainage
An example of oxidation. The oxidation of pyrite and other sulfide minerals forms sulfuric acid, which acidifies surface water

Dissolution
dissolving of minerals in a solution
Clay minerals further decompose to…
Aluminum hydroxides (bauxite) and dissolved silica
Controls on weathering
Resistance of primary minerals to chemical weathering
Climate
availability of water for chemical weathering
temperature (affects reaction rates)
vegetation (increases availability of carbonic acid)
Goethite
FeO(OH); Iron ore. Formed by oxidation of Fe-rich
minerals. Typically massive, fibrous; except bog ore

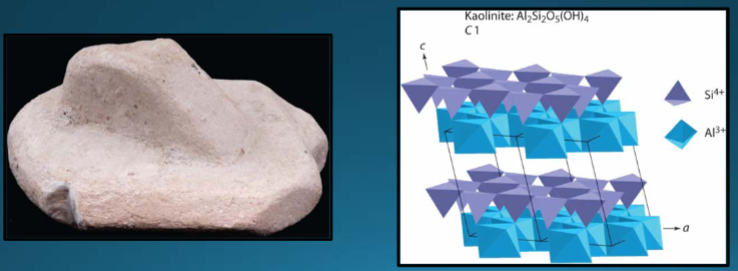
Kaolinite
Al2Si2O5(OH)4. Authigenic (forms in place). Hydrolysis of Al-silicates, such as feldspars. Clay - needs XRD for ID. Ceramics, cosmetics, Kaopectate
Carbonates
Have (CO3)2- as anionic complex
Calcite group (carbonates)
Hexagonal. Effervescence. Common as precipitates or replacement minerals. Dolomite is part of this group but is not isostructural with calcite; forms through replacement after calcite

Aragonite group (carbonates)
Orthorhombic; Aragonite and calcite are polymorphs. No rhombohedral cleavage. Less stable at surface conditions (forms at low T, high P)

Halides
Cations bonded to Cl- or F-. Isometric, common in evaporites

Sulfates
Built with the (SO4)2- anionic complex. Gypsum and anhydrite are common in evaporates

Chert/agate
Microcrystalline to cryptocrystalline quartz.
Chert: found in marine deposits (beds, nodules); BIF
Agate: fills vugs and other voids
