Cancer - LEARNING
1/103
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
104 Terms
how do tumours or neoplasms form?
arise from normal tissues but are unorganised
what are the types of tumours?
benign and malignant
what does benign mean?
non-invasive, localised and can maintain normal function
what does malignant mean?
invasive and metastatic and tissue shows loss of form and function
what are the four major groups of cell type?
epithelial, mesenchymal, haematopoietic, and neuroendodermal
where are epithelial cells found?
line walls/cavities or form outer coverings
what is the function of epithelial cells?
secrete proteins to form basement membranes
what tumours are derived from epithelial cells?
squamous cell carcinomas and adenocarcinomas
where are mesenchymal cells found?
derived from connective tissue, originally from embryonic mesoderm
what tumours occur in mesenchymal cells?
sarcomas
where are haematopoietic cells found?
derived from cell types in blood-forming tissues and the immune system
what tumours occur in haematopoietic cells?
leukaemia and lymphoma
where are neuroendodermal cells found?
derived from components of the CNS and PNS
what tumours occur in neuroendodermal cells?
glioblastomas, neuroblastomas, schwannomas, and astrocytomas
describe the development of cancer?
develops progressively with tumours demonstrating different gradations of abnormality
what are the types of benign tumours?
hyperplastic and metaplastic
what does a hyperplastic tumour consist of?
excessive cell number, cells are normal and form structures
what does a metaplastic tumour consist of?
displacement of normal cells with other normal cells not found in tissue, frequently found in an epithelial transition zone, can be premalignant
give an example of a premalignant metaplastic tumour?
Barrett’s oesophagus
what is dysplastic tissue?
cytologically abnormal cells, premalignant state, may cause malignant tumours
what are cytologically abnormal cells?
variable shape and crowded, hyperchromatic nuclei, lack differentiation markers
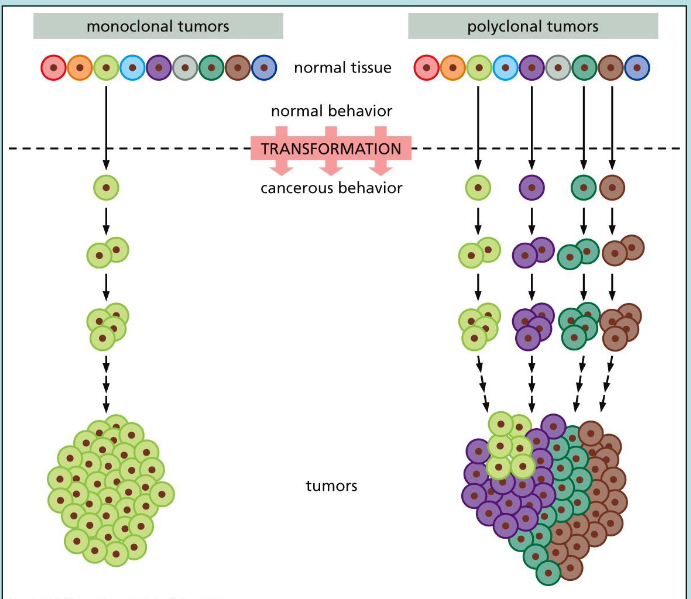
what are monoclonal tumours?
transformation of one type of normal cell to cancerous cells
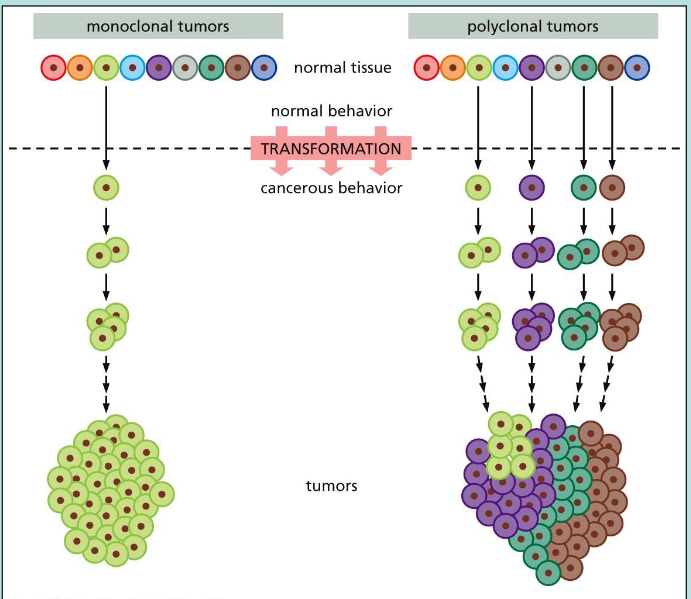
what are polyclonal tumours?
transformation of many normal cells to cancerous cells
why is screening for cancer important?
to detect cancer in early stages, before spread beyond local site, and before symptoms appear, so that it is easier to treat, and that the treatment is more likely to be successful if detected at early stage
what are the criteria for cancer screening?
the condition must be a major health problem, with known natural history and risk factors.
the test must be safe and reliable, specific and sensitive.
the treatment must be effective.
the screening programme must be shown to reduce morbidity, have benefits that outweigh physical and psychological harm, patients must be fully informed, and it must be economically effective and viable.
what are the UK’s cancer screening programmes?
colorectal cancer faecal occult blood test.
cervical cancer HPV test
breast cancer mammography
what are the distinct cellular changes that distinguish a cancer cell from a normal cell?
sustaining proliferative signalling, evading growth suppressors,
avoiding immune destruction, enabling replicative immortality,
activating invasion and metastasis,
inducing angiogenesis,
resisting cell death,
deregulating cellular energetics
what factors influence human carcinogenicity?
environment, reproductive life, lifestyle choices
how do cancer cells sustain proliferation via growth signal autonomy?
normal cells require external stimulation from growth factors to promote entry to the cell cycle, cancer cells may have enhanced external stimulation, or lost dependency due to activating mutations in signal transduction or mutations in cell cycle components
how do cancer cells evade growth inhibitory or suppressive signals?
in order to maintain homeostasis, normal cells respond to inhibitory signals which regulate growth, differentiation, and apoptosis, cancer cells loose this ability via loss of activity in developmental signalling pathways.
how do cancer cells resist cell death?
normal cells with DNA damage are removed via apoptosis, cancer cells evade apoptotic signals via mutations that affect the intrinsic pathway of apoptosis, or mutations that affect the extrinsic pathway
what is angiogenesis?
the formation of new blood vessels
how can cancer cells perform angiogenesis?
occurs via sprouting from pre-existing blood vessels, regulated by a balance between the levels of angiogenic inducers and angiogenic inhibitors, modulated by vascular endothelial growth factor which is regulated by hypoxia and additionally in tumours by oncogenic signalling pathways.
how can cancer cells maintain replicative immorality?
normal cells shorten the ends of their chromosomes during every round of DNA replication which defines a finite number of cell doublings, cancer cells produce telomerase, maintain telomere length and immortality, excessive telomere length in cancer cells causes instability and promotes mutations
how can cancer cells invade the body and produce metastases?
normal cells maintain their location in the body and generally do not migrate, cancer cells gained the ability to invade and spread to distant sites by breaking cell-cell adhesion and cell-extracellular matrix attachment, changing shape and becoming more motile, degrading the ECM via proteases to allow invasion, and inducing angiogenesis to support “secondary” tumour growth
explain metastatic cell preferential spread to particular sites.
close proximity to primary tumour allows for direction of blood flow, and tumour cells get trapped in capillary bed.
distal/non-obvious spread supports seed and soil hypotheses, and premetastatic niche theories
what is the seed and soil hypothesis?
cells need to match with optimum environment
what is the premetastatic niche theory?
distant site is prepared via circulating tumour derived factors
what are the steps involved in invasion and metastasis?
invasion, intravasation, transport, extravasation, metastatic colonisation, and angiogenesis
how is invasion facilitated by epithelial-mesenchymal-transition?
activation of transcription factors SNAIL and TWIST
loss of cell polarity and loss of cell adhesion molecules
changes in cell shape
increase in mesenchymal markers
protease production
promotion of stemness abilities
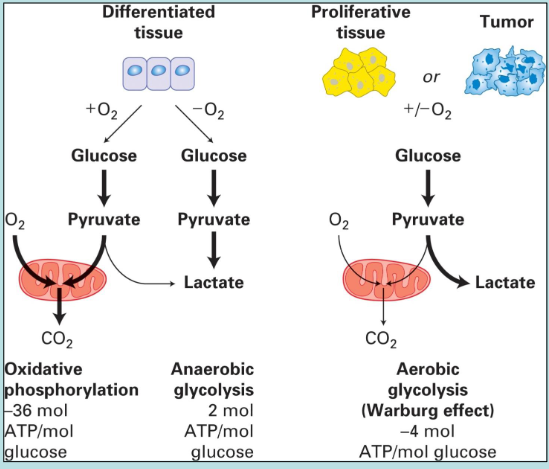
how can cancer cells deregulate cellular energetics?
use glycolysis for generation of intermediates for biosynthetic pathways such as growth
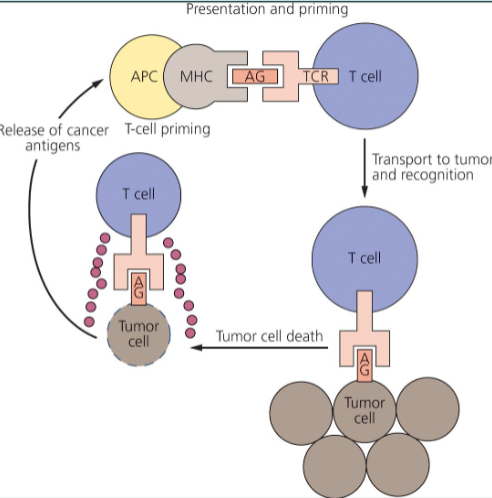
how can cancer cells avoid immune destruction?
loss of tumour antigens, downregulation of antigen-presenting molecules, over-expression of immune checkpoint proteins and anti-apoptotic proteins
what is the function of stem cells?
ability to self-renew and simultaneously form differentiated progeny
what are the types of stem cell?
embryonic and adult
what are embryonic stem cells?
from inner mass of early embryo that can differentiate into different cell types during development
what are adult stem cells?
in regenerative tissue, replaces cells that mature and die
what are cancer stem cells?
subpopulations within a tumour that are thought to initiate and maintain cancer
what are cancer stem cells responsible for?
heterogeneity of tumours, tumour plasticity, and migratory abilities
how can cancer stem cells be identified?
tissue specific stem cell markers, e.g. CD44 for breast and pancreas, and CD133 for colon, prostate, and brain
what causes genomic instability?
physical changes to chromosomes, genomic alterations, faulty DNA repair mechanisms
what does genomic instability promote?
mutation formation and accumulation
explain inflammation in cancer?
tumour cells secrete cytokines that attract tumour-associated macrophages, which produce cytokines and growth factors which can promote cell proliferation, invasion, and angiogenesis, and generate reactive oxygen/nitrogen species which can damage DNA.
what is the multi-hit model of carcinogenesis supported by?
genetic homogeneity in cells from a given tumour, cancer incidence increasing with age, and evidence of cooperative effects of mutations to drive cancer.
what is the effect of a driver mutation?
confers growth/survival advantage
what is the effect of a passenger mutation?
does not confer any advantage
how is ionising radiation a carcinogenic risk?
gamma rays and alpha and beta particles cause direct and indirect DNA damage
describe the direct DNA damage caused by ionising radiation.
ionisation of atoms comprising DNA
describe the indirect DNA damage of ionising radiation.
radiolysis of H2O and ROS generation
what are the cancer risk factors of ionising radiation?
exposure to X-rays, living at altitude, plane travel
how is UV radiation a carcinogenic risk?
mutations induced cause bends in the DNA which are misread by DNA polymerase
describe the effects of UVB?
P53 tumour suppressor affected, causes 80% of skin cancer
what are the risk factors of UV radiation?
over exposure to the sun
describe the general mechanism of action for chemical carcinogenic agents?
an electrophilic form reacts with the nucleophilic sites in the purine and pyrimidine rings of nucleic acids.
what are the direct acting chemical carcinogens?
reactive electrophiles, interaction with nitrogen and oxygen atoms in DNA
what are the indirect acting chemical carcinogens?
unreactive and water soluble, electrophilic centre produced by enzyme modification, interact with bases in DNA forming adducts
how are oncogenic viruses carcinogenic agents?
encode viral proteins that block tumour suppressor action
how are bacteria carcinogenic agents?
cause chronic inflammation that helps promote cancer, bacterial proteins affect cell signalling proteins that regulate cell proliferation, survival, and invasion
how can endogenous reactions have carcinogenic risks?
pathogen killing by immune cells, and enzyme reactions, and carcinogens form from natural endogenous molecules
what are the classes of genes that are implicated in the onset of cancer?
proto-oncogenes, tumour suppressor genes, and caretaker genes
what is the normal role of proto-oncogenes?
involved in positively promoting cell proliferation or cell survival.
what is the effect of mutation of proto-oncogenes?
dominant mutation converts proto-oncogenes into oncogenes
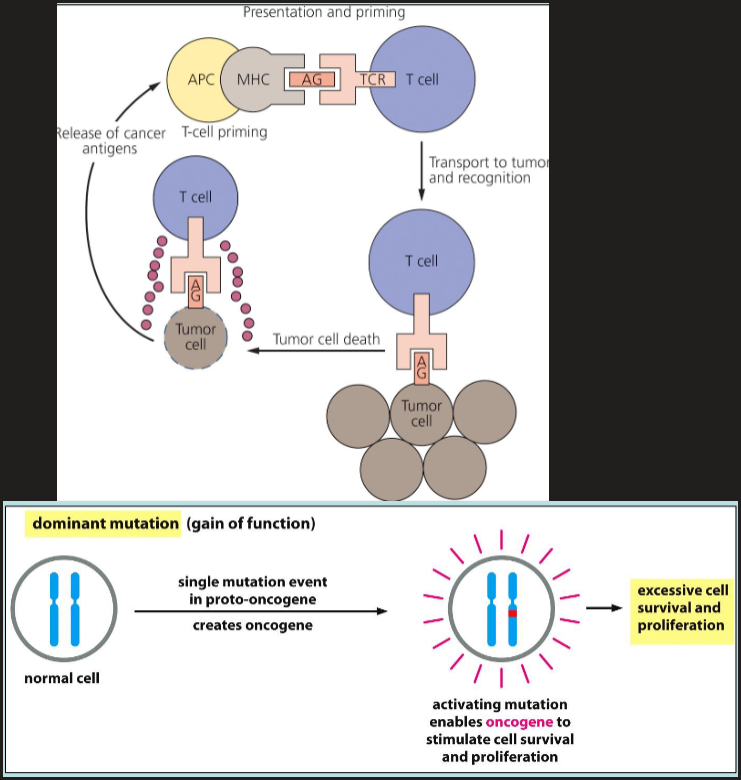
what are the genetic changes that convert a proto-oncogene into an oncogene?
mutation in coding sequence causes protein to become highly active
gene amplification causes normal protein to be overproduced
chromosome rearrangement causes normal protein to be overproduced
what is the normal role of tumour suppressors?
inhibition of cell survival and negatively regulating cell proliferation
what is the effect of mutation on tumour suppressors?
recessive mutation causing excessive cell survival and proliferation
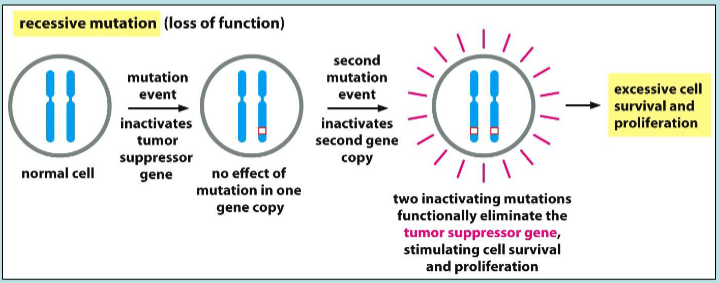
what genetic events can cause loss of function in tumour suppressor genes?
loss of entire chromosome, chromosomal rearrangements, mutations in coding sequence resulting in non-functional proteins, gene activity silenced by epigenetic changes
what is hereditary retinoblastoma?
loss of RB tumour suppressor gene, resulting in childhood retinal tumours and other tumours later in life
what is familial adenomatous polyposis?
loss of APC gene, causing formation of colon polyps which if untreated could develop into malignant colon adenocarcinoma
what is the function of caretaker genes?
repair or prevent DNA damage
what is the effect of mutation on caretaker genes?
recessive mutation affecting DNA mismatch repair, nucleotide excision repair, double stranded DNA breaks
what are the phases of the cell cycle?
M phase (mitosis and cytokinesis) → interphase (G1 phase, S phase, G2 phase)
what is passage through the cell cycle regulated by?
cyclins and cyclin-dependent kinases
what are the classes of cyclins?
G1 cyclins
G1/S cyclins
S phase cyclins
M phase cyclins
what influences activity of CDKs?
cyclin binding, phosphorylation and dephosphorylation, and binding to CDK inhibitors
describe the start checkpoint in interphase?
end of gap 1 phase before entering S phase, questions whether the environment is favourable
describe the G2 checkpoint in interphase?
has all the DNA been replicated successfully, and has any damage been repaired effectively?
describe the checkpoint in mitosis?
just before duplicated chromosomes are pulled apart, to check whether chromosomes are correctly attached to the mitotic spindle to be pulled apart
how do cancer cells deregulate cell division and checkpoint regulators?
overexpression of the proto-oncogene for cyclin D, loss of tumour suppressor genes for p16 CDK inhibitor and RB
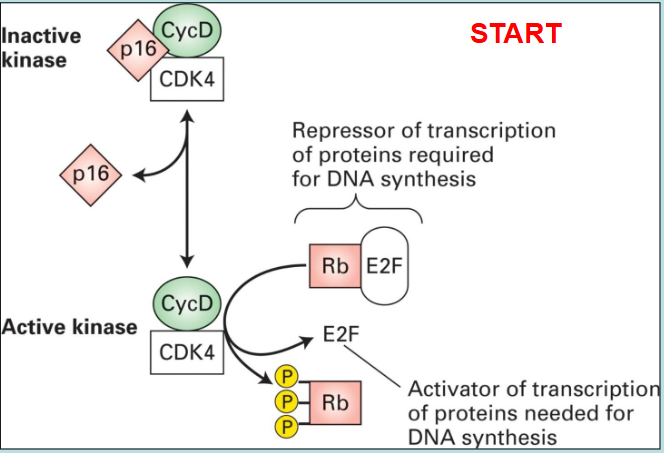
what is p53?
transcription factor, that binds DNA as a tetramer, and binds promoter regions in 300 target genes
what does p53 promote?
cell cycle arrest via production of CDK inhibitors, facilitation of activation of DNA repair mechanisms, and apoptosis
how do mutations in p53 cause cancer?
promote the formation of the malignant phenotype
describe the activation of receptor tyrosine kinase.
ligand induces dimerization interface of inactive monomers, causing receptor-mediated dimerization into active dimers, which then undergo trans-autophosphorylation causing signal transmission of 5H2 or PTB domains
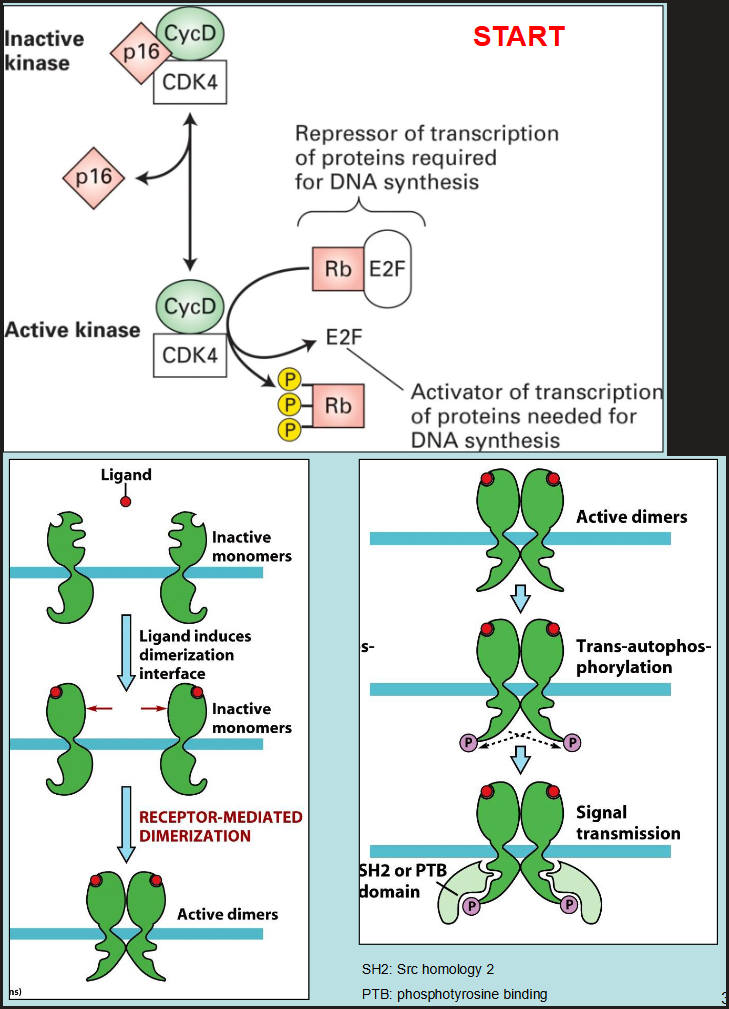
what are the mechanisms of oncogenic activation of growth factor induced pathways?
hyperactive mutant GF, elevated levels of normal GF, increased levels of RTK, mutation in RTK, activation of RTK by a viral protein, and loss of RTK regulatory elements
how is EGFR signalling regulated?
through receptor degradation. activated EGFR recruits the ubiquitin ligase CBL, receptor becomes ubiquitinylated and internalised, targeted by the endocytic pathway, receptor is degraded by hydrolytic enzymes in the lysosome
what mechanisms lead to abnormalities in EGFR signalling?
increased ligand production, increased EGFR levels on cell surface, mutations giving rise to constitutively active variant receptors
how does increased ligand production lead to abnormalities in EGFR signalling?
EGFR ligands particularly EGF are frequently over-expressed in cancer, occurs via autocrine stimulation
what causes increased EGFR levels on cell surface?
gene amplification, defective gene promoter activity, and defective receptor downregulation
how do mutations giving rise to constitutively active variant receptors lead to abnormalities in EGFR signalling?
a variety of mutations are known to exist and can be found in all areas of the receptor, causing loss of extracellular binding domain, constitutive activity, activation of the P13 kinase pathway, increased cell survival and resistance to treatment
what signal transduction molecules drive gain of function mutations in oncogenesis?
RAS, P13-kinase, and RAF
what growth inhibitory molecules drive loss of function mutations in oncogenesis?
p53, retinoblastoma, PTEN
what are the two routes triggering apoptosis?
intrinsic/mitochondrial route, and extrinsic route